Contents
- What is Esomeprazole
- What is the generic and brand name of the drug
- What is the source of the drug (natural or synthetic)
- Waht does Esomeprazole do
- Pharmacophore structure: Information about the chemical structure of the drug
- Chemical information of the drug.
- Esomeprazole strength
- How does Esomeprazole work
- What are the recommended doses of Esomeprazole
- When should I discontinue, withhold or modify the dose of Esomeprazole
- What are the pharmacokinetic properties of the drug
- Which pregnancy category (A; B; C; D; X) has been assigned to Esomeprazole
- How to take Esomeprazole
- How to store the drug
- How to dispose the medicine
- Does Esomeprazole has approval from government / FDA /or any other related agencies
- Other uses of the drug
- What special dietary precautions should I follow
- Esomeprazole precautions
- Esomeprazole side effects
- Esomeprazole overdose
- Esomeprazole missed dose
- Esomeprazole drug interactions
- Does Esomeprazole have any interaction with Diseases
- Where can I get more information
What is Esomeprazole
- Esomeprazole is a drug that is used for the treatment of gastroesophageal reflux disease (GERD).
What is the generic and brand name of the drug
- The drug is available under generic name Esomeprazole and brand names Nexium® and many others worldwide.
- The drug is marketed in by Astra Zeneca Lp. and STAT Rx USA
What is the source of the drug (natural or synthetic)
- Esomeprazole is a synthetic (man-made) pharmaceutical proton pump inhibitor of gastric parietal cells.
Waht does Esomeprazole do
- Esomeprazole is used to inhibit gastric acid secretion and formation of excessive stomach acid.
- The drug is generally used to treat symptoms of gastroesophageal reflux disease (GERD). The disease is characterized by backward flow of stomach acid (gastric reflux) into oesophagus (tube lining the throat and stomach). It results in heart burn, mucosal damage and chronic injury to esophagus.
- Besides GERD, Esomeprazole is also recommended to treat peptic ulcer disease, dyspepsia, and Zollinger-Ellison syndrome (condition in which stomach produces large amount of acid).
- Esomeprazole is used in combination with other drugs for treatment peptic ulcer caused by consumption of nonsteroidal anti-inflammatory drugs (NSAIDs) or by a certain type of bacteria ( pylori).
- The drug reduced formation of excessive stomach acid by inhibiting H+ / K+ ATPase (membrane transporter) in the parietal cells of the stomach. This results in prevention of further damage and allows healing of damage esophagus.

Pharmacophore structure: Information about the chemical structure of the drug
Esomeprazole chemically belongs to the class of organic compounds which are known as sulfinylbenzimidazoles. The sulfinylbenzimidazoles are chemically characterize as polycyclic aromatic compounds containing a sulfinyl group attached to benzimidazole moiety.The detailed chemical classification of Esomeprazole is described below:
| Kingdom | Organic compounds |
| Super Class | Organic heterocyclic compounds |
| Class | Benzimidazoles |
| Sub Class | Sulfinylbenzimidazoles |
| Direct Parent | Sulfinylbenzimidazoles |
Chemical information of the drug.
- Esomeprazole is a synthetic pharmaceutical organic compound named as 5-methoxy-2-[(S)-(4-methoxy-3,5-dimethylpyridin-2-yl)methanesulfinyl]-1H-1,3-benzodiazole.
- The compound has molecular formula C17H19N3O3S and the molecular weight of 461 Da.
- The melting point of Esomeprazole is 155 °C.
- It is slightly soluble in water with a maximum solubility of 0.353mg/ml.
Esomeprazole strength
- The medication is supplied as a delayed-release capsule or suspension for oral administration that means the drug is directly release in intestine rather than stomach to prevent its breakdown by stomach acids.
- Esomeprazole capsule is available in two strengths (20 mg and 40 mg) and as a delayed release oral suspension in three strength (2.5mg, 5mg, 10 mg, 20 mg and 40 mg).
- Each 20 mg and 40 mg capsule of Esomeprazole contains 22.3 mg, or 44.5 mg Esomeprazole magnesium trihydrate in the form ofenteric-coated
- Besides Esomeprazole, other ingredients of capsule includes hypromellose, magnesium stearate, glyceryl monostearate 40-55, hydroxypropyl cellulose, methacrylic acid copolymer type C, polysorbate 80, sugar spheres, talc, and triethyl citrate.
- The capsule is usually swallowed as a whole or open and mix with water or applesauce to take by mouth or or emptied into a syringe for administration through a feeding tube.
- The drug is usually recommended once a day and at least 1 hour prior to meal.
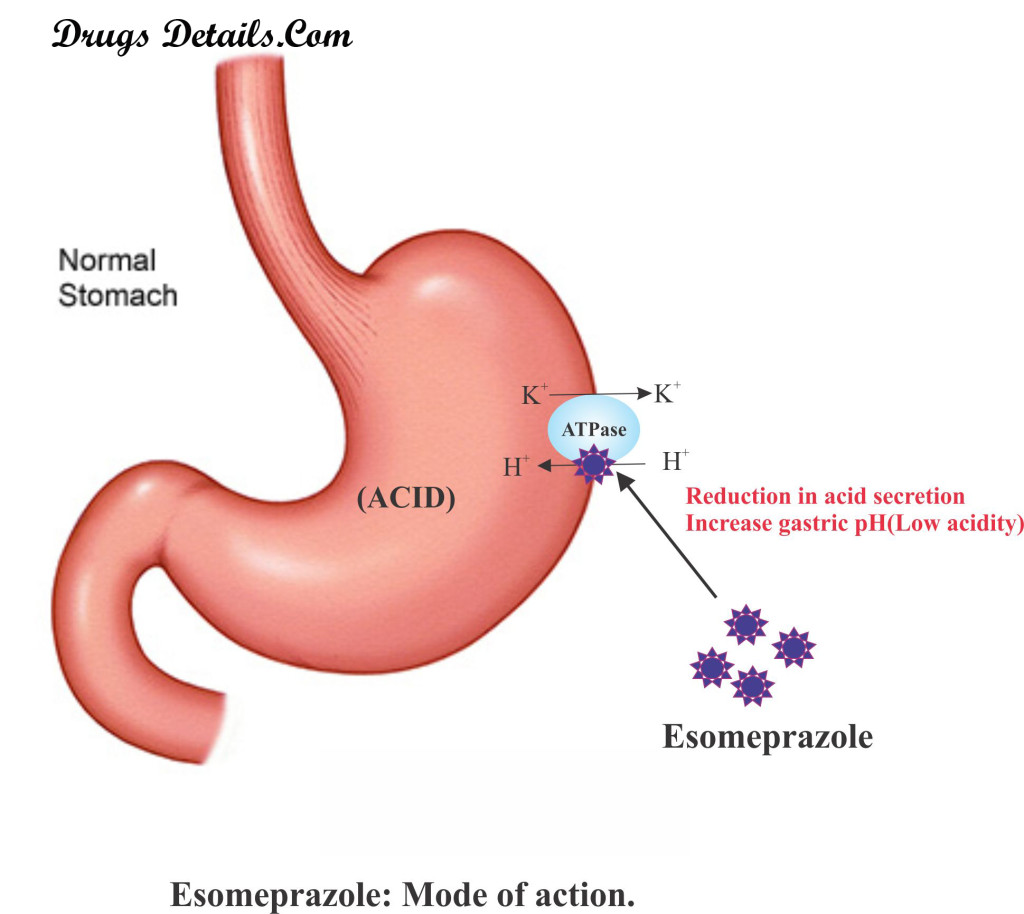
How does Esomeprazole work
- The mode of action of Esomeprazole is similar to the working of other proton pump inhibitors.
- Proton pump inhibitors bind their respective proton pump and blocks their function.
- Esomeprazole brings about inhibition of the H+/K+-ATPase in the stomach or gastric parietal cells responsible for the production of the acid.
- This interferes with the final step in the production of the acid, thereby decreasing the gastric acidity.
What are the recommended doses of Esomeprazole
The prescribed dose of Esomeprazole varies depending upon the age and the diseased state.
- For the treatment of gastroesophageal reflux disease (GERD) and reflux esophagitis:
- The recommended dose of Esomeprazole is 40 mg per day for a period of 4 to 8 weeks. The dose that follows this period is 20 mg per day.
- Treatment of reflux esophagitis in children:
- Age group of 12 months to 11 years: a daily dose of 10-20 mg as per the body weight for a period of 8 weeks.
- Age group of 11years to 17 years: a daily dose of 20-40 mg for a period of 4 to 8 weeks.
- Treatment of Non-erosive relux disease (NERD) comprising heartburn and reflux in children:
- Age group of 12 months to 11 years: a daily dose of 10 mg for a period of 8 weeks.
- Age group of 11years to 17 years: a daily dose of 20 mg for a period of 2 to 4 weeks.
- Treatment of Zollinger-Ellison syndrome characterized by the production of more acid as compared to normal requires a dose of 40 mg twice a day
- NERD or heartburn in case of adults requires the prescribed dosage of 20 mg once a day for the time duration ranging from 2 to 4 weeks.
- For the treatment of duodenal ulcers caused by H. pylori, a dosage of 20 mg twice or 40 mg once a day along with amoxicillin and clarithromycin for a period of 7-10 days.
- The prescribed dosage for prevention of NSAID (nonsteroidal anti-inflammatory drugs) induced ulcers is 20 to 40 mg per day for duration of 6 months. The treatment requires a daily dosage of 20 mg for a period of 4-8 weeks.
When should I discontinue, withhold or modify the dose of Esomeprazole
- The usual dosing of the drug (i.e. 10 mg per day) may vary depending upon the efficiency and side effects of the drug in a particular individual. The health care provider will determine dosage based on the patient’s age, weight and diagnosis.
- No dosage adjustment is necessary in mild to moderate hepatic impairment In case of severe cases, the drug dose should not be exceeded above 20 mg.
- The drug is contraindicated in case of pregnancy, breastfeeding or hypersensitive response to any component of the drug.
- Esomeprazole oral suspension is not prescribed in children less than one year old.
- It has been observed that patients taking higher doses of Esomeprazole are the risk for fractures, therefore it is advisable to take higher dose of medicine under surveillance of health care providers and try to recommend or prescribe a lower dose if possible.
- Dosage adjustments are required in case of geriatric population.
- Esomeprazole use is contraindicated in patients with known hypersensitivity to proton pump inhibitors e.g.,angioedema and anaphylactic shock.
What are the pharmacokinetic properties of the drug
- Pharmacokinetic studies suggest that after oral administration, Esomeprazole is rapidly absorbed and shows bioavailability ranging from 50 – 90 %.
- It has been observed that following a single 20mg to 40mg oral dose of Esomeprazole peak plasma concentrations of 0.5-1.0 mg/l is achieved within 1–4 hours.
- Following absorption the majority (97%) of the drug is bound to plasma proteins.
- The drug is mainly metabolized mainly by the hepatic system.
- The drug is eliminated mainly in urine as metabolites (about 80%) and the remaining 20% as feces.
- The average median half-life of Esomeprazole 1 to 1.5 hours.
- The average steady state volume of distribution of Esomeprazole is approximately 16 liters.
Which pregnancy category (A; B; C; D; X) has been assigned to Esomeprazole
- The Esomeprazole is classified by US FDA pregnancy category: C
- Due to lack of adequate and well-controlled studies the use of Esomeprazole in pregnant women is contraindicated and recommended only when benefit justifies the risk.
- Laboratory animal studies have shown adverse effects on the fetus toxicity.
- No adequate data is available on excretion of Esomeprazole into human breast milk.
- Despite these facts caution should be exercised when taking Esomeprazole.
How to take Esomeprazole
- Esomeprazole is available as delayed release capsule or in granule form for oral administration by mouth
- The capsules should be swallowed and not chewed or crushed.
- The drug is taken only once per day and should be taken at least 1 hour prior to food consumption.
- Try to take the medicine at the same time every day.
- Follow the instructions carefully as directed on prescription leaflet and take Esomeprazole exactly as directed.
- Antacid may be co-administered with the drug if necessary but requires consuming Esomeprazole at least 30 minutes prior to the antacid.
- Take the medication regularly, even if you feel well.
- Do not change the dose of the drug as prescribed by your doctor. Since, the dosage is based on patient medical condition and treatment responses.
How to store the drug
- Esomeprazole is stored at at room temperature between 68°F to77°F (20°C to 25°C).
- The container should be tightly closed and away from excess heat, direct sun light and reach of children.
- Do not freeze or store the medicine at extreme cold too.
How to dispose the medicine
- Throw away unused and opened, outdated or no longer used container.
- Also dispose the old medicine after the expiration date.
- Consult your pharmacist for the proper disposal of the drug.
- Esomeprazole has received its official approval from US Food and Drug Administration (FDA) in February 2001.
- The drug also received official approval from FDA to treat heartburn and other symptoms associated with gastroesophageal reflux disease (GERD).
- The FDA also approved Esomeprazole for treatment of healing of erosive esophagitis. The drug is also recommended to use in combination with amoxicillin and clarithromycin, for the treatment/eradication ofduodenal /peptidic ulcer disease caused by Helicobacter pylori
- Esomeprazole capsules are also approved to treat the gastric ulcers associated with use of nonsteroidal anti-inflammatory drugs (NSAIDs) and Zollinger-Ellison syndrome.
Other uses of the drug
- Esomeprazole may also be used for other uses not listed here. It is advisable to ask your doctor or pharmacist for more information.
What special dietary precautions should I follow
- It is generally recommended to continue with the normal diet unless and until asked by your doctor.
Esomeprazole precautions
- Do not use the medicine if you are hypersensitive or allergic to any of the ingredients.
- Before taking Esomeprazole, tell your doctor about your medical history preferentially if you have any kind of Liver disease or low levels of magnesium in the blood.
- During pregnancy this medication is recommended only when it is essential and under doctor or pharmacist supervision.
- Since the information about excretion of drug in milk is not explored consult your doctor before breast feeding to your child.
- Consult with your doctor and pharmacist if you are taking any prescription and nonprescription medications, nutritional supplements, vitamins and herbal products such as some antifungals like ketoconazole, mediators for HIV, anticoagulants like Warfarin, iron supplements, diuretics etc.
- Do not share this medication with other persons having the similar kind of problems. Consult your doctor for more details.
Esomeprazole side effects
Esomeprazole may provide relief from the acidic conditions but may be associated with a number of serious side effects which are as follows:
- Diarrhea: The chances of acquiring severe diarrhea caused due to intestinal infection by Clostridium difficile.
- Bone fractures: People who are on multiple doses of Esomeprazole for a longer duration pose increased risk of spine, hip or wrist fracture.
- Atrophic Gastritis: Prolonged uptake of Esomeprazole increases the chances of chronic inflammation of the stomach lining.
- Low body magnesium concentration: Magnesium level goes down in people who are on Esomeprazole medication for a period of at least 3 months and may show the following symptoms: seizures; tremors; muscle ache, cramps or weakness; fast heart beat; hands and feet spasm, and dizziness.
The most commonly occurring side effects associated with Esomeprazole may include:
- Gas
- Constipation
- Headache
- Abdominal pain
- Drowsiness
- Diarrhea
- Dry mouth
- Nausea
Serious allergic reactions: Some people may develop following allergic reactions on consuming proton pump inhibitors (such as Esomeprazole)
- Difficulty breathing
- Rash
- Face swelling
- Throat tightness
Other adverse side effects are also reported by the body system and include:
- Body as a Whole: Chest pain, fatigue, pain, enlarged abdomen, flu-like disorder, back pain, chest pain, facial and peripheral edema, leg edema, fever, asthenia, generalized edema, and rigors.
- Hearing: Tinnitus,
- Visual: Conjunctivitis, abnormal vision.
- Gastrointestinal: Constipation, duodenitis, eructation, bowel irregularity, gastroenteritis, esophageal stricture, esophagitis, dysphagia, epigastric pain, esophageal disorder, GI hemorrhage, dysplasia GI, frequent stools, ulcerative stomatitis, esophageal ulceration, vomiting, esophageal varices, gastric ulcer .
- Hematologic: Anemia hypochromic, epistaxis, anemia, cervical lymphadenopathy, leukopenia, leukocytosis, thrombocytopenia.
- Nervous System/Psychiatric: Confusion, nervousness, visual field defect, vertigo, tremor, sleep disorder, apathy, impotence, migraine, hypertonia, hypoesthesia, paresthesia, insomnia, increased appetite, dizziness, depression.
- Hepatic: Abnormal hepatic function, increased SGOT and SGPT, bilirubinemia.
- Cardiovascular: tachycardia, flushing, hypertension.
- Urogenital: Micturition frequency, moniliasis, fungal infection, genital moniliasis, albuminuria, hematuria, polyuria, abnormal urine, dysuria, cystitis.
- Respiratory: sinusitis, larynx edema, pharyngitis, dyspnea, asthma, coughing, rhinitis.
- Reproductive: Vaginitis, change in menstrual cycle, dysmenorrhea.
- Endocrine:
- Metabolic/Nutritional: Increased alkaline phosphatase, thirst, weight gain or loss, vitamin B12 deficiency, hyperuricemia, glycosuria, hyponatremia.
- Special Senses: Taste loss, parosmia, otitis media, taste perversion.
- Skin and Appendages: Dermatitis, increased sweating, urticaria, skin inflammation, acne, angioedema, rash, rash maculo-papular, rash erythematous.
- Musculoskeletal: Arthropathy, cramps, fibromyalgia syndrome, aggravated arthritis, polymyalgia rheumatica, hernia.
Esomeprazole overdose
What happens if you overdose on Nexium?
Try to avoid taking the overdose of the drug. Symptoms include extreme sweating, blurred vision, confusion, or extremely fast heartbeat.
- In case you or some other person has taken overdose of this medication contact your local poison control centre at 1-800-222-1222 or emergency room immediately.
- Consult your doctor or pharmacist for symptomatic and supportive measures in any case of overdose.
Esomeprazole missed dose
- In case of missed dosage, take it as soon as you remember and maintain a regular dosing schedule.
- Skip the missed dose if it is almost time for your next scheduled dose. Keep in mind to not use a double dose to make up a missed dose.
Esomeprazole drug interactions
Controlled clinical studies suggested that Esomeprazole may interact with one of the following drugs. Care should be taken when you are taking these medications together.
- Digoxin: Esomeprazole raises the serum concentration of digoxin but usually may not be clinically important
- Rifampin: Esomeprazole plasma concentration reduces along with its pharmacologic effects.
- Clopidogrel: Esomeprazole brings about an inhibition in the anti-platelet activity of clopidogrel.
- Antifungals (such as voriconazole, ketoconazole, and itraconazole): Co-administration with voriconazole increases the exposure of Esomeprazole and with itraconazole and ketoconazole lowers their bioavailability.
- Warfarin: Use of Esomeprazole enhances the chances of bleeding.
- Clarithromycin: This drug brings about an increase in the plasma levels of Esomeprazole and thereby its pharmacologic effects and side effects.
- Benzodiazepines (eg, diazepam): Coadministration with diazepam may increase its CNS effects (eg, sedation, ataxia).
- Nilotinib, dasatinib, erlotinib: Try avoiding these drugs along with Esomeprazole as their absorption is greatly hampered in its presence.
- Fluvoxamine: It elevates the plasma levels as well as adverse effects of Esomeprazole.
- Rilpivirine: Esomeprazole causes an elevation in the plasma levels and reduced pharmacologic effects and virologic resistance of rilpivirine.
- Cilostazol: Esomeprazole enhances the concentration of cilostazol along with its active metabolite (i.e. 3,4-dihydrocilostazol) and hence requires lowering of this drug dose when given in combination with Esomeprazole.
- Tacrolimus: Esomeprazole elevates the plasma levels of tacrolimus thereby affecting its pharmacologic and adverse effects.
- Calcium salts: The uptake of calcium by gastro-intestinal tract may be impeded by the use of drug to lower the acid secretion.
- Methotrexate: Esomeprazole causes an increase in the concentration and side effects of methotrexate by lowering its excretion by renal pathway.
- Clozapine: Esomeprazole causes an increase in the plasma concentration and hence pharmacologic and adverse effects of clozapine.
- Iron salts (such as ferrous sulfate): Esomeprazole consumption interferes with the uptake of iron salts.
- Protease inhibitors (like atazanavir, indinavir, nelfinavir, saquinavir): Plasma level of these inhibitors gets affected in presence of Esomeprazole. Saquinavir plasma levels get increased. Try to avoid coadministration of atazanavir or nelfinavir with Esomeprazole.
- Mycophenolate: The plasma levels of mycophenolate and its pharmacologic effects get decreased in presence of Esomeprazole.
- Tolterodine: Esomeprazole causes an elevation in the release of tolterodine from the ER and its pharmacologic and adverse effects.
Does Esomeprazole have any interaction with Diseases
- It has been suggested that Esomeprazole is primarily metabolized in liver, therefore, the exposure to Esomeprazole in patients with mild, moderate or severe hepatic impairment is significantly higher than the normal individuals.
- It is suggested that little amount of Esomeprazole can accumulate in Child Pugh Class A and B following once-daily, multiple-dose administration.
- Esomeprazole plasma concentration was increased substantially in patients with severe hepatic impairment (Child Pugh Class C) in comparison to healthy individuals. Thus, the drug is recommended a maximum dosage of 20 mg once daily in patients with severe liver disease.
Where can I get more information
Your pharmacist or health care provider can provide more information about Esomeprazole.
Clinical research and current scenario of the drug
- Esomeprazole reduces gastric acid secretion by inhibiting protom pump in the gastric parietal cells. It blocks the final step in the gastric acid production.
- In controlled clinical trials it has been observed that Esomeprazole administration elevated the serum gastrin (is a peptide hormone that stimulates secretion of gastric acid ) which returned to baseline levels following discontinuation of therapy.
- Clinical studies using oral doses of 20 mg and 40 mg Esomeprazole suggested no effects on thyroid and parathyroid functions.
- Esomeprazole reduces the gastric acidity which results increases gastric counts of bacteria normally present in the gastrointestinal tract. It has been observed that long term treatment with proton pump inhibitors may lead to increased risk of gastrointestinal infections such as Salmonella, Campylobacter and Clostridium difficile.
- Animal studies using RAT and rabbit model suggested that Esomeprazole at oral doses up to 280 mg/kg/day and 86 mg/kg/day showed no evidence of impaired fertility or harm to the fetus.
References from chemical, biological and toxicological databases
- DrugBank: Esomeprazole (DB00736). http://www.drugbank.ca/drugs/DB00736.
- Esomeprazole | C17H19N3O3S | ChemSpider. http://www.chemspider.com/Chemical-Structure.7843323.html.
- Medline plus information. http://www.nlm.nih.gov/medlineplus/druginfo/meds/a699054.html
- Esomeprazole: Indications, Side Effects, Warnings. http://www.drugs.com/cdi/esomeprazole.html.
- Nexium – Food and Drug Administration. http://www.fda.gov/downloads/Drugs/DrugSafety/UCM322355.pdf.
- The pharmacology of Esomeprazole and its role in gastric acid related diseases. http://www.ncbi.nlm.nih.gov/pubmed/19606942.
- Perils and pitfalls of long-term effects of proton pump inhibitors. http://www.ncbi.nlm.nih.gov/pubmed/23927671.
- Esomeprazole: a proton pump inhibitor. http://www.ncbi.nlm.nih.gov/pubmed/19210109.
- Esomeprazole: a review of its use in the management of gastric acid-related diseases in adults. http://www.ncbi.nlm.nih.gov/pubmed/18627213.
Read about,
“Vardenafil, uses, strength, side effects, mechanism of action“
“Fondaparilux, uses, strength, side effects, mechanism of action“
