Contents
- What is Warfarin
- What is the generic and brand name of the drug
- What is the source of the drug (natural or synthetic)
- What does Warfarin do
- Pharmacophore structure: Information about the chemical structure of the drug
- Chemical information of the drug
- Warfarin available strength
- How does Warfarin work?
- Warfarin recommended doses
- When should I discontinue, withhold or modify the dose of Warfarin
- What are the pharmacokinetic properties of the drug
- Which pregnancy category (A; B; C; D; X) has been assigned to Warfarin
- How to take Warfarin
- How to store Warfarin
- How to dispose Warfarin
- Does Warfarin has approval from government / FDA /or any other related agencies
- Other uses of the drug
- What special dietary precautions should I follow
- What special precautions should I follow / What should I avoid while using Warfarin
- Warfarin side effects
- If you experience any of these symptoms, call your health care provider immediately
- Warfarin Overdose
- Warfarin missed dose
- Warfarin interactions with other drugs
- Does Warfarin have any interaction with Diseases
- Where can I get more information
- Clinical research and current scenario of the drug
- References from chemical, biological and toxicological databases
What is Warfarin
- Warfarin is a drug that is used for the treatment of existing blood clots and to prevent new blood clots formation inside the body.
What is the generic and brand name of the drug
- The drug is available under generic name Warfarin or Warfarin sodium and brand names Jantoven, Coumadin, Uniwarfin,
What is the source of the drug (natural or synthetic)
- Warfarin is a synthetic (man-made) pharmaceutical anticoagulant that acts by inhibiting vitamin K-dependent coagulation factors.
What does Warfarin do
- Warfarin is used to treat or prevent venous thrombosis (swelling and blood clot in a vein) to exclude chances of further spreading of the clot
- The drug is also prescribed for patients suffering from pulmonary embolism (a blood clot in the lung) to lower the risk of further embolism.
- Warfarin is also recommended for people with irregular and rapid heartbeat (atrial fibrillation) or artificial heart valves to minimize the risk factor of strokes, and also for people who have had a heart attack.
- The drug also is helpful in some of the orthopedic surgeries like replacement of hip or knee to prevent formation of blood clots.
- During the treatment of coronary heart disease, the drug find application in preventing closure of coronary artery stents ( tube-shaped device placed in the coronary arteries supplying blood to the heart, to keep the arteries open) due to clotting.
Pharmacophore structure: Information about the chemical structure of the drug
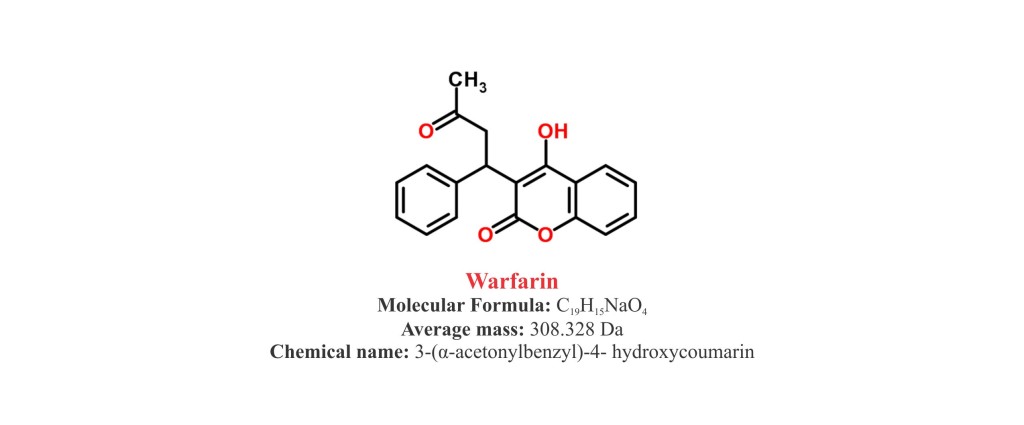
Warfarin chemically belongs to the class of organic synthetic compounds known as 4-hydroxycoumarins that contain one or more hydroxyl groups at C4-position in the coumarin skeleton. The detailed chemical classification of Warfarin is as follows.
| Kingdom | Organic compounds |
| Super Class | Phenylpropanoids and polyketides |
| Class | Coumarins and derivatives |
| Sub Class | Hydroxycoumarins |
| Direct Parent | 4-hydroxycoumarins |
Chemical information of the drug
- Warfarin sodium (Coumadin) is a synthetic pharmaceutical class of organic compound known as 4-hydroxycoumarins.
- It is chemically known as 3-(α-acetonylbenzyl)-4- hydroxycoumarin.
- It is available as sodium salt with a molecular formula C19H15NaO
- Molecular weight of Warfarin sodium is 330.31 g/mol and melting point is 161°C.
- Warfarin sodium occurs as a white, odorless, crystalline powder
- It is very soluble in water, freely soluble in alcohol, and very slightly soluble in chloroform and ether.
- It has a pKa (the number that denotes the pH) of 5. 08.
Warfarin available strength
- Warfarin is available for oral administration in tablet form or in the form of injections.
- Warfarin tablets are available in different dosage strength which contain different dosage of Warfarin sodium (1 mg, 2 mg, 2.5 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7.5 mg and 10 mg) respectively.
- The Warfarin tablets contain a very small amount of colour additives for product identification.
- Each tablet shows strength imprinted on one side, and is scored so that it can be broken easily in two halves to adjust the dose as directed by the doctor.
- Each strength of tablet is depicted by different colour to differentiate between them: 0.5 mg tablets are white, 1 mg tablets are brown, 3 mg tablets are blue and 5 mg tablets are pink.
- The formulation contains Warfarin Sodium as the active ingredient and Lactose, starch, and magnesium stearate as the inactive ingredients.
- Powder for injection is also available as 5 mg/vial.
How does Warfarin work?
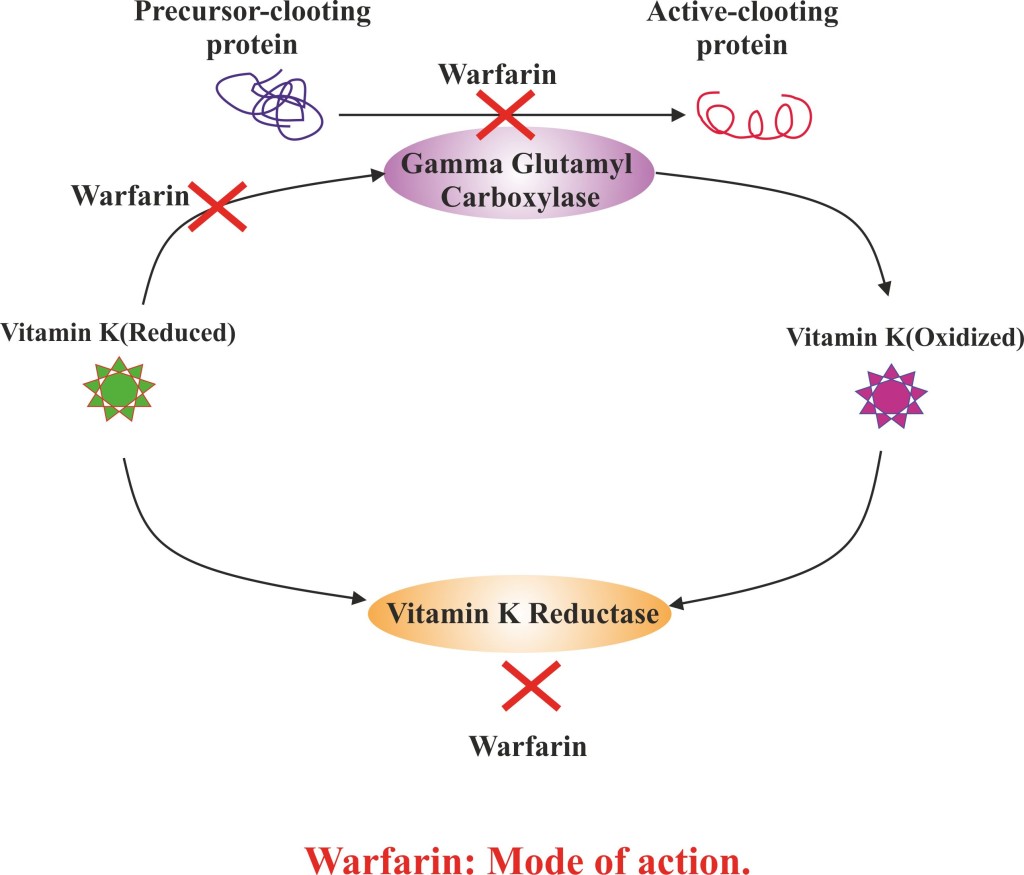
- Warfarin acts by antagonizing vitamin K and reduction of the factors involved directly or indirectly in blood clotting.
- Warfarin brings about the inhibition of vitamin K reductase enzyme, which results in decrease pool of reduced form of vitamin K.
- Reduction in vitamin K (reduced) lowers down the glutamate residues carboxylation in the N-terminal regions of coagulant proteins.
- This inhibits the synthesis of vitamin K-dependent coagulation factors (factors II, VII, IX, and X) and anticoagulant proteins (regulatory factors) C and S by the liver.
- Decrease in the coagulation factors causes lowering in the prothrombin levels.
- Reduce prothrombin affects production of thrombin and fibrin bound thrombin,which ultimately reduces blood clotting.
Warfarin recommended doses
- Warfarin may be taken along with or without food.
- Warfarin dosage in patients suffering from liver and kidney dysfunction need to be lowered as it may affect its metabolism by the liver and excretion by the kidneys.
- Regular blood tests (International Normalized Ratio (INR) or prothrombin time (PT)) have to be performed to determine the effect of Warfarin and to adjust dosing.
- The dosage and administration of the drug has to be individualized for each patient according to the patient’s INR response to the drug.
- The drug treatment initially is started at 2 to 5 mg dosage per day and then adjusted on the basis of INR tests and the diseased state.
Congestive Heart Failure:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Myocardial Infarction:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Pulmonary Embolism — First Event:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Pulmonary Embolism — Recurrent Event:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Atrial Fibrillation:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Deep Vein Thrombosis — First Event:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Deep Vein Thrombosis — Recurrent Event:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Deep Vein Thrombosis Prophylaxis after Hip Replacement Surgery:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day. - Maintenance: 2 to 10 mg orally or intravenously once a day.
Deep Vein Thrombosis Prophylaxis after Knee Replacement Surgery:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Thromboembolic Stroke Prophylaxis:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
Maintenance: 2 to 10 mg orally or intravenously once a day.
Chronic Central Venous Catheterization:
- 1 mg orally or intravenously per day with initiation of therapy three days prior to the insertion of the catheter.
Prosthetic Heart Valves — Mechanical Valves:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
- Maintenance: 2 to 10 mg orally or intravenously once a day.
Prosthetic Heart Valves – Tissue Valves:
- Initial: 2 to 5 mg orally or intravenously per day for 1 to 2 days, then adjusted according to results of the INR or PT.
- Maintenance: 2 to 10 mg orally or intravenously once a day.
- The duration of therapy to be continued depends on individual patient. However, the drug therapy should be continued until the risk of thrombosis and embolism has passed.
When should I discontinue, withhold or modify the dose of Warfarin
- Warfarin therapy should be discontinued or modified in some of the dental and surgical procedures.
- Warfarin is contraindicated in pregnant women except in cases of pregnant women who have mechanical heart valves and are prone to thromboembolism.
- Avoid Warfarin as initial therapy in patients with heparin-induced thrombocytopenia or thrombocytopenia.
- Avoid Warfarin under following health conditions, including:
- High blood pressure
- Hypersensitivity
- Bleeding disorder, such ashaemophilia
- Increased risk of bleeding inside the body such as in peptic ulcer, CNS haemorrhage, dissecting aorta, or over bleeding of the gastrointestinal, genitourinary or respiratory tracts;
- Threatened abortion
- Patients with conditions such as dementia, alcoholism, psychosis
- Hemorrhagic tendencies
- Spinal puncture and other diagnostic or therapeutic procedures with potential for uncontrollable bleeding
What are the pharmacokinetic properties of the drug
- Pharmacokinetic studies suggested that after oral administration, Warfarin is rapidly absorbed with considerable inter-individual variations.
- The elimination of Warfarin is almost entirely by metabolism. Very little Warfarin is excreted unchanged in urine. The metabolites are principally excreted into the urine; and to a lesser extent into the bile.
- Warfarin is stereo-selectively metabolized by hepatic cytochrome P-450 (CYP450) microsomal enzymes to inactive hydroxylated metabolites (predominant route) and by reductases to reduced metabolites (Warfarin alcohols)
- Warfarin distributes into a relatively small apparent volume of distribution of about 0.14 L/kg.
- The bioavailability of the drug ranges from 79-100% (oral).
- It has been observed that following a dose of Warfarin maximum (or peak) serum concentration is achieved within the first 4 hours.
- Following absorption the majority (99%) of the drug is bound to plasma proteins (primarily to albumin).
- The average median plasma half-life of Warfarin is approximately 40 hours.
Which pregnancy category (A; B; C; D; X) has been assigned to Warfarin
- The Warfarin is classified by US FDA pregnancy category: D for women with mechanical heart valves and prone to thromboembolism; category X in case of other pregnant populations
- Warfarin intake during pregnancy results in congenital malformations such as fetal haemorrhage, Warfarin embryopathy, spontaneous abortion and fetal mortality.
- No studies have been done to evaluate the reproductive or developmental effects of Warfarin in animals.
- No studies indicate the excretion of Warfarin in human breast milk.
How to take Warfarin
- Warfarin is available in tablet form for oral administration by mouth with or without food usually once a day. .
- Follow the instructions carefully as directed on prescription leaflet and take Warfarin exactly as directed. Take the medication regularly, even if you feel well until the time prescribed by your doctor.
- Dose should not be changed even in case of skipping the dose two or more days continuously.
- Do not change the dose as prescribed by your doctor, or stop abruptly as the dosage is based on your health condition, laboratory tests (such as INR), and treatment response.
- A regular and balanced diet is recommended while consuming Warfarin.
- Avoid any modification in the food habits especially related to vitamin k as it may affect the working of the drug as well as treatment and dose.
- Consult with the doctor in case you plan to diet.
- It is also recommended to take drug at almost the same time every day.
The dose and schedule of Warfarin depends on various factors, including health conditions, medical background and associated diseases.
How to store Warfarin
- Warfarin is stored at room temperature between 59 and 86 ° F (15 and 30 ° C).
- The container should be tightly closed and away from excess heat, direct sun light and dampness.
- Do not freeze or store the medicine at extreme cold too.
- The medication should be kept out of the reach of children.
How to dispose Warfarin
- Throw away unused and opened, outdated or no longer used container.
- Also dispose the old medicine after the expiration date.
- Talk to your pharmacist or waste disposal company about the proper disposal of your medication.
- Do not pour the drug in the drain or flush it.
- Warfarin has received its official approval from US Food and Drug Administration (FDA) in June 1954.
Other uses of the drug
- Warfarin may also be used for other uses not listed here. It is advisable to ask your doctor or pharmacist for more information.
What special dietary precautions should I follow
- It is generally recommended to eat green leafy vegetables while you are on Warfarin medication.
- However, enormous consumption of green leafy vegetables and certain vegetable oils which are rich source of vitamin K should be avoided as it interferes with the action of Warfarin.
- Foods rich in vitamin K should not be removed entirely from the diet.
- Follow a normal routine diet with similar levels of food every week to maintain the INR constant.
- Consult your doctor about eating grapefruit and drinking grapefruit juice while taking Warfarin.
- Do not make any change in the food habit without bringing it focus to your dietician.
What special precautions should I follow / What should I avoid while using Warfarin
It has been observed that person to person response or Warfarin is different and primarily dependent on their heredity or genetic make-up. Therefore, it is advisable that Warfarin prescription/administration should be taken under the guidance of your health care provider.Some common precautions which should be followed before taking Warfarin are as follows.
- Do not use the medicine if you are hypersensitive to any of the ingredients. Ask your pharmacist or check the Medication leaflet or Guidance book for complete list of the ingredients.
- It is advisable to discuss with your doctor and pharmacist about what prescription and nonprescription medications, vitamins, and nutritional and natural supplements you are taking or plan to take.
- Talk with your doctor or pharmacist about what herbal or botanical products you are taking, especially St. John’s wort, coenzyme Q10 (Ubidecarenone), Ginkgo biloba, goldenseal, garlic, ginseng, and),
- Tell your doctor if you have or have ever had history of stomach or intestinal bleeding , low blood count or cancer ,diabetes, history of stroke, or “mini-stroke”, serious heart disease and kidney problems.
- Tell your doctor if you are taking or plan to take any other medicines that increase your risk of bleeding, other medicines to prevent or treat blood clots and NSAIDs.
- Tell your doctor if you are being treated or have recently been treated with chemotherapy or radiation therapy.
- Inform your doctor if you are breastfeeding or pregnant or plan to become pregnant. Warfarin do not recommended for pregnant women (may harm the fetus).unless they have a mechanical heart valve.
- Inform your doctor if you are taking birth control pills while taking Warfarin.
- Inform your doctor having surgery, including dental surgery.
- Avoid using tobacco products; smoking can significantly reduce the Warfarin effect.
- Keep regular and timely visit with your doctor and routinely undergo for blood test like prothrombin test (reported as international normalized ratio) to check your body’s response to Warfarin.
When you start taking Warfarin, regularly refill your prescription , read the information carefully and discuss all your queries with your doctor or health care provider. For more details you can also visit Food and Drug Administration (FDA) website (http://www.fda.gov/downloads/Drugs/DrugSafety/ucm088578.pdf).
Warfarin side effects
The common side effects of Warfarin are as follows.
- Abdominal or stomach pain, cramping and gas
- Taste change
- Dizziness
- Chills or cold
- Hair loss
- Bloating etc.
If symptoms persist and become more severe consult your doctor immediately.
If you experience any of these symptoms, call your health care provider immediately
Some of the common signs that need to be addressed immediately to your health care provider comprise:
- Allergy i.e. hives, rashes, itching.
- Breathing/ swallowing difficulty
- Severe chest pain or pressure
- Swelling of the face, the hands, lower legs, throat, tongue, lips, ankles and eyes
- Lack of energy/extreme tiredness
- Nausea/vomiting
- Flu like symptom and fever
- Hoarseness
- Infections
- Yellowing of the skin or eyes
- Loss of appetite
- Ulcer etc
Warfarin Overdose
What should I do in case of overdose?
Some common symptoms of Warfarin overdose are as follows:
- Spitting or coughing up blood. Coffee color stool
- Continued oozing or bleeding from minor cuts or injuries
- Yellowish eyes, pink, red, or dark brown urine.
- Bloody or red, or tarry bowel movements
- Excessive bleeding during menstrual period
- Small, flat, round red spots under the skin
- Unusual large bruising or bleeding
- Try to avoid taking the overdose of the drug. If you overdose the drug contact with your doctor or pharmacist for symptomatic and supportive measures.
- In case you or some other person has taken overdose of this medication contact your local poison control centre at 1-800-222-1222 or emergency room immediately.
- Call local emergency services at 911 in case a patient collapses or faces difficulty in breathing.
Warfarin missed dose
What should I do in case of missed a dose?
- In case of missed dosage, take it as soon as you remember and maintain a regular dosing schedule.
- Skip the missed dose if it is almost time for your next scheduled dose. Keep in mind to not use a double dose to make up a missed dose
Warfarin interactions with other drugs
The Warfarin may interact with one of the following drugs. Care should be taken when you are taking these medications together.
- Medications for asthma such as zafirlukast, montelukast and zileuton .
- Medications for cholesterol such as atorvastatin and fluvastatin
- Medications for narcolepsy such as armodafinil and modafinil
- Medications for seizures such as phenobarbital, carbamazepine , phenytoin and rufinamide Medications to treat tuberculosis such as rifampin and isoniazid
- Medications used to treat cancer such as imatinib, capecitabine , , and nilotinib
- Serotonin reuptake inhibitors (SSRIs) or selective serotonin and norepinephrine reuptake inhibitors (SNRIs).
- Medications for digestive disorders such as famotidine, cimetidine , and ranitidine.
- Medications for human immunodeficiency virus (HIV) infection such as fosamprenavir , amprenavir, atazanavir , efavirenz , etravirine, lopinavir/ritonavir, ,indinavir , nelfinavir etc.
- Acyclovir , allopurinol , alprazolam etc
- Antibiotics such as ciprofloxacin, norfloxacin sulfinpyrazone, erythromycin , clarithromycin , nafcillin, and tigecycline
- Anticoagulants such as argatroban , bivalirudin , dabigatran , heparin, desirudin
- Antifungals such as fluconazole , ketoconazole , posaconazole , terbinafine , miconazole , voriconazole
- Antiplatelet medications such as cilostazol , , prasugrel, clopidogrel , dipyridamole and ticlopidine
- Aspirin or aspirin-containing products and other nonsteroidal anti-inflammatory drugs such as diclofenac, celecoxib , diflunisal, fenoprofen , ibuprofen , indomethacin, mefenamic acid oxaprozin and sulindac
- Antiarrhythmic medications such as amiodarone , propafenone and mexiletine.
- Calcium channel blocking medications such as amlodipine, verapamil and diltiazem
This list do not comprises all the drugs that interact with Warfarin and there are many other drugs that can interact with Warfarin. If you start any new medication, first consult your doctor or pharmacist
Does Warfarin have any interaction with Diseases
It has been observed that some medical conditions (disease state) may also interact with Warfarin such as:
Diabetes: Diabetes predisposes the patient to increased hemorrhage in presence of Warfarin.

- Bleeding: Patients with hemorrhagic diathesis (an inherited predisposition to abnormalities characterized by excessive bleeding), risks for bleeding, active ulceration, active bleeding, threatened abortion, cerebrovascular hemorrhage, deficiency in vitamin c and k, malnutrition, etc. are more prone to uncontrollable hemorrhage or bleeding complications during Warfarin therapy.
- Liver diseases: Patients with hepatic impairment shows heightened response to Warfarin owing to decreased metabolism and defective hemostasis by impaired clotting factors synthesis via liver.
- Hypertension: Patients of hypertension (malignant or severe, uncontrolled) are more prone to cerebral hemorrhage.
- Protein C Deficiency: It has been observed that Warfarin consumption may interfere with familial, hereditary,or clinical deficiencies of protein C . Since protein C deficiency may be associated with a hyper-coagulable state and an increased risk of the complication.
- Renal dysfunction: It has been suggested that person suffered from renal impairment may demonstrate platelet defects and may be at increased risk for bleeding.
- Decreased Response: Patients with edema, hypothyroidism, hereditary coumarin resistance, hyperlipidemia, or nephrotic syndrome may exhibit lower than expected hypo-prothrombinemic response to Warfarin.
- Increased Response: Patients with a collagen vascular disease (e.g., systemic lupus erythematosus, rheumatoid arthritis, and scleroderma), congestive heart failure (especially decompensated disease), severe or prolonged diarrhea, fever, hyperthyroidism, malabsorption, or steatorrhea may exhibit greater than expected hypoprothrombinemic response to Warfarin.
Where can I get more information
Your pharmacist or health care provider can provide more information about Warfarin.
Clinical research and current scenario of the drug
- Efficacy of Warfarin in reducing the risk of systemic thromboembolism has been shown in some randomized controlled clinical trials in patients of non-rheumatic Atrial Fibrillation (AF).
- Clinical trials in patients suffering with both mitral stenosis and atrial fibrillation have shown them to be benefited from the use of Warfarin.
- Randomized, open-label clinical trial of Warfarin in providing the thromboembolic-free interval in patients with mechanical prosthetic heart valves was more significant as compared with aspirin/ pentoxifylline and aspirin / dipyridamole -treated patients.
- Open label clinical study has shown similar frequency of thromboembolism in patients with mechanical prosthetic heart valves receiving moderate or high Warfarin therapies with major bleeding in case of high intensity.
- Randomized study with higher or lower intensities of Warfarin therapy has shown same frequency of thromboembolism in different groups of test patients with major haemorrhages in the higher intensity group compared to zero in the lower intensity group.
- The Warfarin Re-Infarction Study has shown major bleeding episodes in patients receiving Warfarin than those receiving aspirin alone. Frequency of major bleeding reduced drastically in patients receiving aspirin and Warfarin while frequency of minor bleeding episodes was higher in the combined therapy group.
References from chemical, biological and toxicological databases
- Warfarin | C19H16O4 – PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Warfarin.
- DrugBank: Warfarin (DB00682). drugbank.ca/drugs/DB00682.
- Warfarin | C19H16O4 | ChemSpider. chemspider.com/Chemical-Structure.10442445.html.
- Warfarin: MedlinePlus Drug Information. nlm.nih.gov/medlineplus/druginfo/meds/a682277.html.
- Novel oral anticoagulants: a review of the literature and considerations in special clinical situations. http://www.ncbi.nlm.nih.gov/pubmed/23466963.
- A review of Warfarin dosing and monitoring. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1305837/.
- Warfarin – National Library of Medicine – PubMed Health . http://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0001928/.
- Comparison of the efficacy and safety of new oral anticoagulants with Warfarin in patients with atrial fibrillation: a meta-analysis of randomized trials. http://www.ncbi.nlm.nih.gov/pubmed/24315724.
- Warfarin and atrial fibrillation: from ideal to real the Warfarin affaire. http://www.thrombosisjournal.com/content/12/1/5.
Other medicines,
“Fenofibrate, drug class, uses, strength, side effects, mechanism of action“
“Filgrastim, drug class, uses, strength, side effects, mechanism of action“
