Contents
- What is Filgrastim
- Generic and brand name of Filgrastim
- What is the source of drug (natural or synthetic)
- What does Filgrastim do
- Pharmacophore structure: Information about the chemical structure of the drug
- Chemical information of the drug
- Filgrastim strengths
- How does Filgrastim work
- Recommended doses of Filgrastim
- When should I discontinue, withhold or modify the dose of Filgrastim
- What are the pharmacokinetic properties of the drug
- Which pregnancy category (A; B; C; D; X) has been assigned to Filgrastim
- How to take Filgrastim
- How to store the drug
- How to dispose the medicine
- Does Filgrastim has approval from government / FDA /or any other related agencies
- Other uses of the drug
- What special dietary precautions should I follow
- What special precautions should I follow?/ What should I avoid while using Filgrastim
- Filgrastim side effects
- Filgrastim overdose
- Filgrastim missed dose
- Does Filgrastim have any interaction with drugs
- Does Filgrastim have any interaction with Diseases
- Where can I get more information
- Clinical research and current scenario of the drug
What is Filgrastim
- Filgrastim is a synthetic pharmaceutical analogue of naturally occurring granulocyte colony-stimulating factor(G-CSF).
Generic and brand name of Filgrastim
- The drug is available under generic name Filgrastim and brand names Filcad, Imumax, Neukine, Neupogen, Grafeel, Emgrast, Religrast, Zarzio or Zarxio and Nufil.
- The Filgrastim is marketed in by Cadila Pharmaceuticals, Abbott Laboratories, Dr. Reddys Laboratories, Intas pharmaceuticals, Novartis, Biocon etc.
What is the source of drug (natural or synthetic)
- Filgrastim is a synthetic (man-made) pharmaceutical antineutropenic and hematopoietic agents.
What does Filgrastim do
Why is this medication prescribed?
- Filgrastim is a granulocyte colony-stimulating factor (G-CSF) analogue, which is principally used to enhance the growth, differentiation and proliferation of granulocytes by stem cells.
- Granulocytes (neutophils, PMN, mast cells, eosinophils and basophils) are key cells of immune system and help to protect body against diseases and foreign invaders.
- Filgrastim is generally used in cancer patients (acute myeloid leukaemia) who are receiving chemotherapy, or in patients undergoing bone marrow transplants or in patients under condition of severe chronic neutropenia (low number of neutrophils in the blood).
- Since chemotherapy weakens the patient immune system and decrease the number of neutrophils thus increases the chances of infections. Filgrastim works by helping the body by stimulating the proliferation and differentiation of granulocytes (including neutrophils), thus protect the body from infections.
- The drug is also used to increase the number of stem cells as well as to treat low white blood cells count in case of patients undergoing bone marrow transplants or under condition of severe chronic neutropenia.
- The drug is also used for blood preparation for leukapheresis, (a process whereby white blood cells are removed from the blood in the body and returned back following chemotherapy).
- Filgrastim is also used to enhance the survival in patients undergoing or receiving radiation therapy.
Pharmacophore structure: Information about the chemical structure of the drug
- Filgrastim chemically belongs to the class of organic compounds known as carboxylic acid derivative. The detailed chemical classification of Filgrastim is as follows.
| Kingdom | Organic compounds |
| Super Class | Organic Acids |
| Class | Carboxylic Acids and Derivatives |
| Sub Class | Amino Acids, Proteins , Peptides, and Analogues |
| Direct Parent | Peptides/Proteins |
Chemical information of the drug
- Filgrastim is a synthetic pharmaceutical recombinant protein of 175 amino acid (non-pegylated human granulocyte colony stimulating factor analogue).
- The sequence of Filgrastim is identical to its natural sequence as predicted from DNA, except N-terminal methionine which is needed for its expression in bacterial host.
- It is synthesized using recombinantDNA technology in bacterial strain of E. coli.
- The compound has molecular formula C845H1343N223O243S9 and the molecular weight of 18.8 Da.
- The melting point of Filgrastim is 60°C.
- The hydrophobicity and isoelectric point of Filgrastim is 209 and 5.65, respectively.
Filgrastim strengths
What is the available strength of the drug?
- Filgrastim is available in solution (liquid) form in vials and pre filled syringes for intravenous and subcutaneous administration.
- The drug can be injected directly into the skin or into a vein.
- Filgrastim is available in different dosage strength of 300 µg/ 0.5 ml or ml and 480 µg/0.8 ml or 1.6 ml.
How does Filgrastim work
How the medicine works (mode of action)?
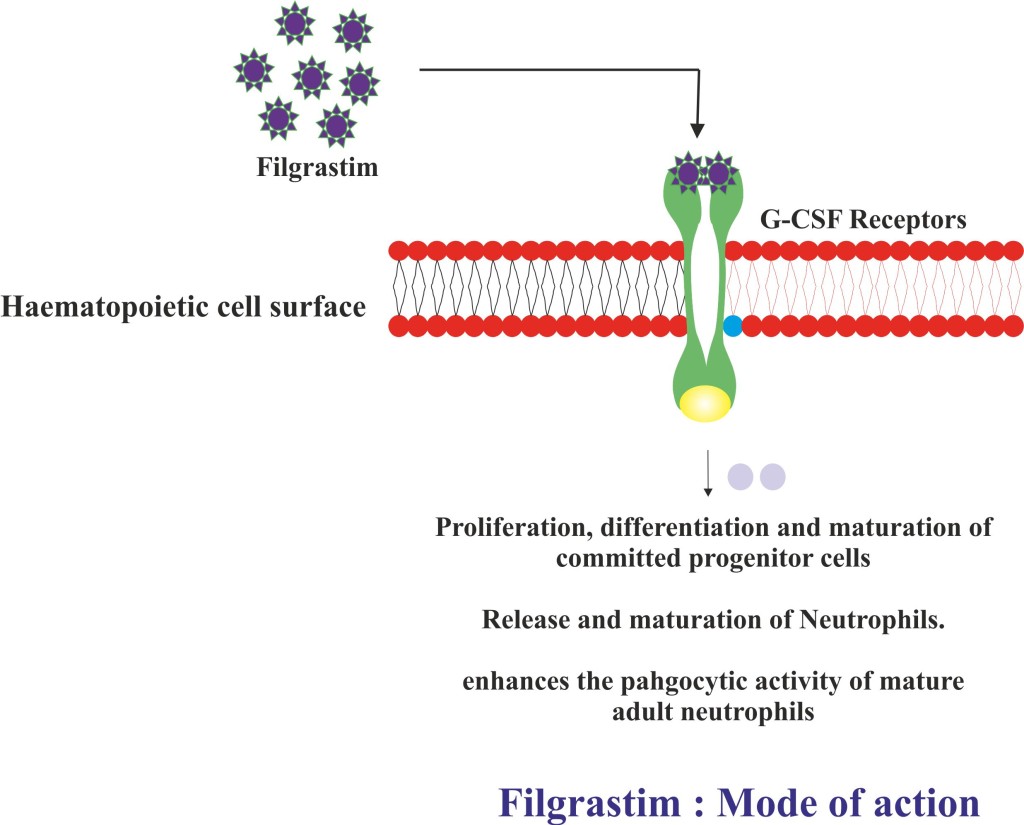
- Filgrastim is an antineutropenic and hematopoietic agent that enhances the production of granulocytes.
- It acts on hematopoietic cells as an analogue of granulocyte colony-stimulating factor(G-CSF) and binds to specific cell surface receptors.
- Following binding Filgrastim stimulates the proliferation, differentiation and maturation of committed progenitor cells.
- It also stimulates the release and maturation of neutrophils from storage pool of bone marrow cells.
- Apart from this, Filgrastim also enhances the phagocytic activity of mature adult neutrophils.
- In patients receiving chemotherapy or radiation therapy Filgrastim reduces the duration of neutropenic state by enhancing the neutrophils recovery.
Recommended doses of Filgrastim
The Filgrastim is available in ready to use solution form and the dosage varies depending upon the diseases status.
- The initial usual dose of Filgrastim for adult patient with Neutropenia Associated with Chemotherapy is 5 mcg/kg/day either as subcutaneous infusion / or short intravenous infusion (15 to 30 minutes)/or subcutaneous or intravenous infusion.
- The initial usual dose of Filgrastim for adult patient with Bone Marrow Transplantation is10 mcg/kg/day either as an intravenous infusion of 4 or 24 hours, or continuous 24 hour subcutaneous infusion.
- The initial usual dose of Filgrastim for adult patient with Peripheral Progenitor Cell Transplantation is 10 mcg/kg/day either as subcutaneous infusion or as a bolus or a continuous infusion, for a minimum period of 4 days. The treatment begins before the first leukapheresis procedure and scheduled until the last leukapheresis.
- The initial usual doses of Filgrastim for adult patient with Congenital Neutropenia are 6 mcg/kg subcutaneously (twice a day), while 5 mcg/kg subcutaneously once a day in case of Idiopathic or Cyclic Neutropenia.
- The initial usual dose of Filgrastim for pediatric use in patients with Congenital Neutropenia are 6 mcg/kg subcutaneously (twice a day), while 5mcg/kg subcutaneously once a day in case of Idiopathic or Cyclic Neutropenia.
- The initial usual dose of Filgrastim for pediatric use for Neutropenia Associated with Chemotherapy is 5 mcg/kg/day either as subcutaneous bolus injection, or short intravenous infusion (15 to 30 minutes) or by continuous subcutaneous or intravenous infusion.
It is recommended that Filgrastim should not be administered earlier than 24 hours following cytotoxic chemotherapy.
A complete blood count (CBC) and platelet count should be monitored before instituting therapy, during the therapy.
Bone marrow and cytogenetic evaluations should also be performed throughout the duration of treatment, particularly in case of congenital neutropenia.
However, the represented dose schedule can vary according to patient response and can change on the basis of CBC count or other routine tests. Treatment should continue until disease progression or unacceptable toxicity occurs.
When should I discontinue, withhold or modify the dose of Filgrastim
- The dose of Filgrastim should be adjusted based on the patient’s clinical status, course and absolute neutrophil count (ANC).
- The data for use of Filgrastim is contraindicated in case of severe renal impairment as well as patient undergoing dialysis.
- The drug is also not recommended in patients with active liver disease or persistent liver dysfunction or abnormal function.
- The use of the drug is also restricted in patients who are hypersensitive to the Filgrastim.
- In case of pre-existing gallbladder disease, the Filgrastim use is not recommended.
- The use of Filgrastim has not been evaluated in case of hepatic impairment; however it is necessary that the condition should be monitored very cautiously while using higher dosage.
What are the pharmacokinetic properties of the drug
- Following administration, Filgrastim does not accumulate and follows first order absorption pharmacokinetic without apparent concentration dependence.
- It has been observed that following subcutaneous injection of 3.45 mcg/kg and 11.5 mcg/kg of Filgrastim the maximum serum concentration is 4 and 49 ng/mL.
- The peak plasma concentration of Filgrastim is achieved within 2 to 8 hours.
- The absolute bioavailability of Filgrastim is approximately 62% and 71% for 375 mcg and 750 mcg doses respectively.
- Pharmacokinetic studies with Filgrastim have shown that following administration is primarily eliminated by the kidney.
- Besides kidney, the drug is also eliminated by neutrophils/neutrophils precursors. This route of elimination involves binding of the growth factor receptor, internalization, and subsequent degradation inside the cells.
- The peak plasma level of drug is appeared within 6-8 hours following administration of the drug.
- The bioavailability of Filgrastim is increased significantly to 35% under fed conditions as compared to fasting.
- The details regarding protein binding, metabolism and toxicity of drug was not reported.
- Filgrastim is primarily eliminated by the kidney and neutrophils/neutrophils precursors. The latter presumably involves binding of the growth factor to the G-CSF receptor on the cell surface, internalization of the growth factor-receptor complexes via endocytosis, and subsequent degradation inside the cells.
- Following absorption the majority (~90%) of the drug is protein bound to plasma.
- The distribution volume of the drug in healthy people and cancer patients is 150 mL/kg.
- The average elimination half-life of Filgrastim in healthy subjects and cancer patients is near 3.5 hours.
- No significant relationships were observed between the drug absorbance and clearance to patient age, body weight or gender.
Which pregnancy category (A; B; C; D; X) has been assigned to Filgrastim
- The Filgrastim is classified by US FDA pregnancy category: C
- Due to lack of adequate and well-controlled studies the use and safety of Filgrastim in pregnant women is contraindicated and recommended only when potential benefit justifies the risk.
- No adequate data is available on excretion of Filgrastim into human breast milk. However, the use of drug is not recommended in nursing mothers.
Due these facts caution should be exercised when taking Filgrastim during pregnancy.
How to take Filgrastim
- Follow the instructions carefully as directed on prescription leaflet and take Filgrastim exactly as directed or exactly directed by your health care professional.
- Do not change the dose of the drug as prescribed by your doctor.
- It is also recommended to take drug at almost the same time every day.
- If you have any queries about the drug immediately consult to your doctor to explain any part you do not understand.
- If you use Filgrastim at home, ask your doctor beforehand about the technique of using it.
- Filgrastim should be removed from the refrigerator and kept at room temperature for 30 minutes prior to its use.
- Shaking the vial or syringe of Filgrastim is not recommended due to the formation of foam.
- Discoloured, particulate or cloudy solution of Filgrastim should not be used.
- Damaged or cracked vials/syringes of Filgrastim should be disposed off.
- Only disposable syringes and needles should be used for the injection of Filgrastim.
How to store the drug
- Filgrastim is stored in the refrigerator at 2° to 8°C (36° to 46°F).
- Do not freeze or store the medicine at extreme cold too.
- The drug should be thawed in the refrigerator if it gets frozen. Repeated freezing should not be allowed.
- Discard Filgrastim if exposed to room temperature for more than 24 hours.
How to dispose the medicine
- Throw away outdated or no longer used medicine.
- Also dispose the old medicine after the expiration date.
- Talk to your pharmacist about the proper disposal of your medication.
- Filgrastim has received its official approval from US Food and Drug Administration (FDA) in February 1991 to treat neutropenia (low white blood cells).
Other uses of the drug
- Filgrastim may also be used for reducing the chances of infection in HIV infected individuals.
- It also decreases infection in the people who are on medications that lowers the neutrophils count.
- Filgrastim is also prescribed for the treatment of cancer of white blood cells (acute myeloid leukemia).
- Filgrastim is also sometimes recommended for the treatment of myelodysplastic syndrome wherein blood cells are either poorly formed or are dysfunctional.
- Filgrastim may also be used for other cases not mentioned here. It is advisable to ask your doctor or pharmacist for more information.
What special dietary precautions should I follow
- It is generally recommended to continue with the normal diet unless and until asked by your doctor.
What special precautions should I follow?/ What should I avoid while using Filgrastim
- First of all inform your doctor if you are allergic to Filgrastim products or any of the ingredients present in the Filgrastim product or any medication made from coli bacteria. Ask your pharmacist or check the prescription leaflet carefully for a list of the ingredients.
- It is advisable to discuss with your doctor and pharmacist about what prescription and non-prescription medications, vitamins, and nutritional supplements you are taking or plan to take.
- Tell your doctor if you are being treated or have recently been treated with radiation therapy.
- Inform your doctor if you are pregnant or plan to become pregnant. Tell your doctor if you are breastfeeding.
- Inform your doctor if you are having surgery, including dental surgery.
- It is generally recommended to avoid vaccination while you are on Filgrastim medication.
Filgrastim side effects
- Filgrastim may be associated with a number of side effects. Some of the effects can be more serious and others less severe. A person may not show all the side effects but may be one or a few.
- Some people may develop allergic reactions on taking Filgrastim.
- A person should get the emergency help immediately if he shows any sign of allergic reaction. These may include
- Swelling on the body parts including throat, lips, tongue and face
- Hives
- Difficulty in breathing
In case of serious side effects, the use of drug should be withdrawn instantaneously. Side effects that may be serious are as follows:
- Fever
- Sore throat
- Chill
- Bleeding in nose and gums
- Abdominal or stomach pain
- Nausea
- Vomiting
- Extreme weakness
- Rapid breathing
- Sore in the mouth
- Loss of appetite
- Difficulty in breathing
- Headache
- Signs of flu
- Dizziness
- Paralysis
- Prolonged bleeding in the cuts or wounds
- Difficulty in urination
- Blood during cough
Some of the effects may be less serious and include:
- Swelling or redness at the place of injection
- >Headache
- Constipation
- Itching
- Skin rashes
- Feeling of tiredness
- Diarrhea
- Pain in bones
- Hair loss
- Body or muscle ache
This list of side effects is not complete. Other side effects may also occur. In some cases side effects vanish during treatment and need no medical care. Your doctor would suggest you the ways to lessen the side effects. Consult your doctor in case of any query or if the side effects continues.
Filgrastim overdose
What should I do in case of overdose?
- Overdose usually occurs when someone by mistake or deliberately takes more than the prescribed limit of this medication.
- There is no specific treatment for Filgrastim overdose.
- In case of overdose, contact with your doctor or emergency room immediately.
- Seek emergency medical attention or call the Poison Help line at 1-800-222-1222.
Filgrastim missed dose
What should I do in case of missed a dose?
- In case of missed dosage, take it as soon as you remember and maintain a regular dosing schedule.
- Skip the missed dose if it is almost time for your next scheduled dose. Keep in mind not to use a double dose to make up a missed dose.
Does Filgrastim have any interaction with drugs
- Due to lack of controlled trial studies of Filgrastim interactions, no formal data is available. However, it has been suggested that precautions should be taken while taking any of the drug that can stimulate release, activation or proliferation of neutrophils. Therefore, caution should be taken when co administrating the Filgrastim with one of the following drugs.
- It has been observed that enhanced hematopoietic activity of bone marrow may be associated with transient positive bone imaging changes with transient positive bone imaging changes. This fact should be kept in mind when interpreting bone imaging results of patients taking Filgrastim.
- This information is not complete and if you start any new medication, first consult your doctor or pharmacist.
Does Filgrastim have any interaction with Diseases
It has been observed that Filgrastim consumption may potentiate or initiate the following medical complications (disease):
- Acute Respiratory Distress Syndrome (ARDS): It has been observed that continuous use of Filgrastim can cause Acute respiratory distress syndrome (ARDS). It is advisable to discontinue Filgrastim in patients with ARDS or if patient develop respiratory distress, fever and lung infiltrates should be monitored for ARDS.
- Splenic Rupture: Filgrastim consumption may also cause splenic rupture and fatal cases can occur. Patients treated with Filgrastim should be discontinue and diagnose properly if feeling upper abdominal or shoulder pain.
- Tumor Growth Stimulatory Effects on Malignant Cells: Clinical studies with Filgrastim suggested that Filgrastim acts on tumor cell lines, thus can acts as a growth factor for any tumor type i.e. Myeloid malignancies and myelodysplasia.
- Allergic Reactions: It has been observed that Filgrastim administration can cause serious allergic reactions including anaphylaxis. It can occur on either initial exposure or during subsequent treatment. It bis generally recommended to permanently discontinuing Filgrastim in patients with serious allergic reactions.
- Use in Patients with Sickle Cell Disease: Filgrastim use in Sickle Cell Disease may cause severe and sometimes fatal sickle cell crises. It is advisable to keep the potential risks and benefits into consideration prior to the administration of Filgrastim.
Where can I get more information
Your pharmacist can provide more information about Filgrastim.
Clinical research and current scenario of the drug
- Randomized, placebo-controlled, double-blind experiment in patients suffering from nonmyeloid malignancies has shown the efficacy and safety of Filgrastim in lowering the chances of infection.
- Randomized, multi-center, placebo-controlled, double-blind experiments in patients of acute myeloid leukaemia(AML) have revealed role of Filgrastim in decreasing the fever, neutrophils recovery time after chemotherapy of patients.
- The effectiveness of Filgrastim in diminishing the extent of neutropenia in patients inflicted with nonmyeloid malignancies has been established in randomized control experiments.
- Studies have also indicated the role of Filgrastim in mobilization of peripheral blood progenitor cells that can be collected in a procedure wherein white blood cells are separated from the sample of blood.
References from chemical, biological and toxicological database
- http://www.nlm.nih.gov/medlineplus/druginfo/meds/a692033.html
- Filgrastim | C20H21ClO4 | ChemSpider. chemspider.com/Chemical-Structure.3222.html
- DrugBank: Filgrastim (DB00099). http://www.drugbank.ca/drugs/DB00099
- Tbo-filgrastim – Food and Drug Administration.
- Filgrastim – Drug Information – Chemocare. http://chemocare.com/chemotherapy/drug-info/filgrastim.aspx
- Filgrastim and antibiotics treatment reduces neutropenia severity in solid cancer patients. http://www.ncbi.nlm.nih.gov/pubmed/19827886
- A review of its pharmacological properties and therapeutic efficacy in neutropenia. http://www.ncbi.nlm.nih.gov/pubmed/7530630
- Randomized trial of filgrastim, sargramostim, or sequential sargramostim and filgrastim after myelosuppressive chemotherapy for the harvesting of peripheral-blood stem cells. http://www.ncbi.nlm.nih.gov/pubmed/10623692
- Filgrastim | C20H21ClO4 – PubChem. http://pubchem.ncbi.nlm.nih.gov/compound/Filgrastim
- The role of Filgrastim in clinical practice. http://www.ncbi.nlm.nih.gov/pubmed/17935056
- Filgrastim : a review of its use in primary dyslipidaemia, the metabolic syndrome and type 2 diabetes mellitus. http://www.ncbi.nlm.nih.gov/pubmed/17209672
- Filgrastim: treatment of hyperlipidemia and beyond. http://www.ncbi.nlm.nih.gov/pubmed/19018684
- Filgrastim is a recombinant, non-pegylated human granulocyte colony stimulating factor (G-CSF) analogue manufactured by recombinantDNA technology using a strain of E. coli. It is marketed as the brand name Neupogen by Amgen. Chemically, it consists of 175 amino acid residues. The protein has an amino acid sequence that is identical to the natural sequence predicted from human DNA sequence analysis, except for the addition of an N-terminal methionine necessary for expression in E coli. Tbo-filgrastim, which is marketed by Sicor Biotech and FDA approved on August 29, 2012, contains the same active ingredient as Neupogen and is biologically similar, but it is formulated to be short-acting. On March 6, 2015, the FDA approved the biosimilar Zarxio (filgrastim-sndz) and is indicated for use in the same conditions as Neupogen. Zarxio is marketed by Sandoz.
“Warfarin, uses, strength, side effects, mechanism of action“
“Vardenafil, uses, strength, side effects, mechanism of action“
