Contents
- What is Aripiprazole?
- How aripiprazole looks like?
- Aripirazole molecular formula, structure, weight, chemical name, drug class
- Why is this medicine prescribed?
- How does Aripiprazole works in the body?
- Aripiprazole brand names
- What is Aripiprazole used for?
- How must Aripiprazole be taken?
- Aripiprazole dosage for different indications
- Aripirazole dosage for different forms
- Ariprazole drug abuse and dependence
- What happens if a dose of aripiprazole is missed?
- What happens if I overdose aripiprazole?
- Aripiprazole side effects
- For how long aripiprazole stays in the body?
- Warnings and precautions of aripiprazole
- Use of aripiprazole in pregnancy
- Use of Aripiprazole in nursing mothers
- Use of aripiprazole in pediatrics
- Use of aripiprazole in geriatric use
- Use of aripiprazole in renal Impairment
- Use of aripiprazole in hepatic impairment
- Use of aripiprazole and different genders
- Use of aripiprazole and different race
- Can I take aripiprazole with alcohol?
- Can I take aripiprazole with aspirin?
- Can I take aripiprazole with bupropion?
- Can I take aripiprazole with acetaminophen?
- Can I take aripiprazole with codeine?
- Can I take aripiprazole with clozapine?
- Can I take aripiprazole with hydrocodone?
- Can I take aripiprazole with fentanyl?
- Can I take aripiprazole with methadone?
- Can I take aripiprazole with oxycodone?
- Can I take aripiprazole with tramadol?
- Can I take aripiprazole with topiramate?
- Can patients with diabetes use aripiprazole safely?
- Can patients with high cholesterol levels use aripiprazole safely?
- Can you get weight gain after aripiprazole use?
What is Aripiprazole?
Aripiprazole is an antipsychotic medicine that is used for the treatment of schizophrenia. It is also prescribed to treat acute mania and episodes of bipolar disorder. It shows its antipsychotic effect at the D2 receptor by partial agonism. It also shows partial agonist activity at 5-HT1A receptor. It show the antagonist effect at 5-HT2A receptor. It show moderate affinity towards histamine and alpha adrenergic receptors.
How aripiprazole looks like?
It is colorless and is like a flake crystals that is derived from ethanol. The boiling point is 139-139.5 degree Celsius. It is soluble in water. Its solubility in water is 0.045 mg/L at 25 degree C. Usually its solubility level is <0.3 [ug/mL]. Vapor pressure is 3.15X10-13 mm Hg at 25 degree Celsius. It is stable under standard storage conditions.
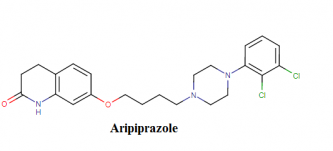
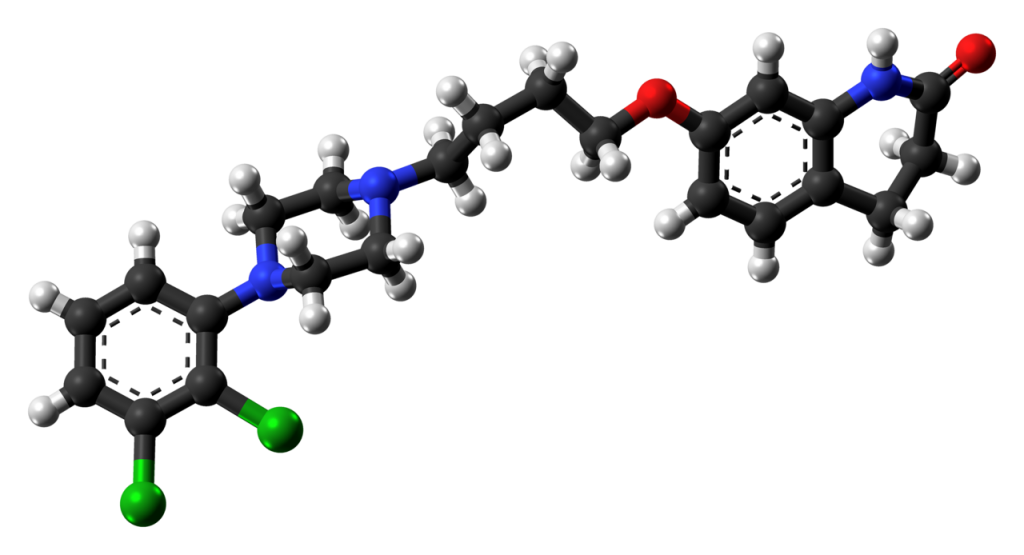
Aripirazole molecular formula, structure, weight, chemical name, drug class
Molecular weight- Average: 448.385 g/mol; Monoisotopic: 447.148032537
Aripirazole molecular structure:
Chemical (IUPAC) name- 7-{4-[4-(2,3-dichlorophenyl)piperazin-1-yl]butoxy}-1,2,3,4-tetrahydroquinolin-2-one
Chemical or Molecular Formula- C23H27Cl2N3O2
Aripiprazole drug class- Antipsychotic
This compound belongs to the class of organic compounds known as phenylpiperazines. These are compounds containing a phenylpiperazine skeleton, which consists of a piperazine bound to a phenyl group.
Why is this medicine prescribed?
Aripirazole is available in following dosage forms: oral tablets, oral solution and orally-disintegrating tablets. Oral forms are used for the treatment of:
- Schizophrenia
- Adjunctive Treatment of depression
- Treatment of Tourette’s Disorder
- Irritability that is associated with Autistic.
- Acute manic and mixed episodes with bipolar I disorder
Injection is prescribes for treating:
- Agitation with schizophrenia and bipolar mania
How does Aripiprazole works in the body?
Its activity is the result of the combination of two kinds of receptors- 1. Antagonism at D2 receptors in mesolimbic pathway. 2. 5HT2A receptors in the frontal cortex. Antagonism at D2 receptors has positive symptoms while antagonism at 5HT2A receptors results in negative symptoms of schizophrenia. It is a psychotropic agent that belong to the class of benzisoxazole derivatives. It is indicated for the treatment of schizophrenia.
It is a selective monoaminergic antagonist that have high affinity for the serotonin Type 2, 1 and 2 adrenergic dopamine Type 2 (D2), and H1 receptors. It also acts as an antagonist at other sites of receptors with lower potency. Aripiprazole antagonism of histamine H1 receptors explains somnolence. Aripiprazole antagonism of adrenergic receptors explains the orthostatic hypotension.
Aripiprazole brand names
Popular brand names for aripirazole are: Abilify Discmelt, Abilify.
What is Aripiprazole used for?
It is used to treat psychotic disorders like schizophrenia and bipolar I disorder. The enough information is not present that states that if Aripiprazole is effective in children of age 13 with schizophrenia and bipolar disorder.
It is also used with other medicines for the treatment of the major depressive disorder in adults. It is used in children of age 6 years or older suffering from Tourette’s disorder and autistic disorder. It is used to treat the symptoms of irritability, mood swings, aggression, and self-injury.
How must Aripiprazole be taken?
You must take this drug as prescribed by the doctor. You must follow all the instructions that are mentioned on your prescription. You are advised to not to take this medicine without the advice of the doctor. Do not take this medicine longer than 6 weeks. If the doctor has prescribed to take, then you can have it. It can be taken without or with food. In the case of liquid medicine and measure it with the syringe.
Do not stop using this medicine suddenly. If you stop this medicine that can make your condition worse. The doctor needs to check the progress while using this medicine. Store it at room temperature. It must be stored away from heat and moisture.
For taking the orally disintegrating tablet, the following instructions must be followed-
- Keep the tablet in the blister pack until you are not ready to take. Open the packet and peel foil. Do not push the tablet from the foil else it can result in the damaging of the tablet.
- Wipe your wet hands and use dry hands for removing the tablet. Place it in your mouth.
- Do not swallow the whole tablet. Keep the tablet on your tongue. Let it dissolve in your mouth. If it is require, you can drink water to swallow the tablet.
- Use Aripiprazole on the regular basis.
Aripiprazole dosage for different indications
Schizophrenia treatment
- Adults
The recommended is 10 or 15 mg/day to be administered once in a day. It is shown to be effective in a range of 10 – 30 mg/day when given in the form of a tablet. The doses higher than 10 or 15 mg/day were not effective.
The Maintenance Dose in schizophrenia was shown in a trial that involves patients with schizophrenia. They were stable with other antipsychotic medicines for the period of 3 months or longer. The patients were discontinued from these medications and were given Aripiprazole 15 mg/day. Patients should be reassessed from time to time to determine the maintenance dose.
- Adolescents
The recommended is 10 mg/day. It was studied in adolescent patients of age 13 – 17 years with schizophrenia at doses of 10 mg and 30 mg. The starting dose of the tablet was 2 mg that was titrated to 5 mg after 2 days. It was targeted to the dose of 10 mg after 2 days. The Subsequent dose increases to 5 mg. The 30 mg/day dose has not shown efficacy than 10 mg/day dose.
- When you are switching from other Antipsychotic drugs
There are no available data that specify switching patients suffering with schizophrenia from other antipsychotics to this drug. The immediate discontinuation of the previous antipsychotic drugs can be acceptable for some patients. But gradual discontinuation is recommended for others.
Bipolar I disorder: treatment of mixed episodes & acute mania
- Adults
The recommended starting dose in adults is 15 mg that is given once in a day. The recommended dose for mono therapy is 10 mg- 15 mg once in a day. It can be given with lithium or valproate. The target dose is 15 mg/day. The dose can be increased to 30 mg/day. The safety of dose above 30 mg/day is not been analyzed in clinical trials.
- Pediatrics
The recommended initial dose for age 10 to 17 years as mono therapy is 2 mg/day. It can be titrated with titration to 5 mg/day after 2 days. It can further be titrated to a target dose of 10 mg/day after 2 days. The recommended dose as adjunctive therapy for lithium or valproate is same. The dose can be increased to 5 mg/day.
Adjunctive treatment in depression
- Adults
The recommended initial dose for the patients that are already taking an antidepressant is 2 to 5 mg/day. The recommended dose is 2 to 15 mg/day. The dose can be adjusted to 5 mg/day. The patients must be periodically analyzed to determine the need of maintenance treatment.
Irritability that is associated with autistic disorder
- Pediatric Patients of age 6 to 17 years
The recommended dose for pediatric patients suffering from irritability with autistic disorder is 5 to 15 mg/day. The dose must be initiated at 2 mg/day that can be increased to 5 mg/day. If required the doses can be increased to 10 or 15 mg/day. The dose can be adjusted to 5 mg/day. The patients must be periodically analyzed.
Tourette’s Disorder
- Pediatric Patients of age 6 to 18 years
The recommended dosage is 5 to 20 mg/day. The patients that weigh less than 50 kg. The dosing should be given at 2 mg/day. The target dose is 5 mg/day after 2 days. The dose may be increased to 10 mg/day. The dose adjustments can occur at less than a week.
The patients that are weighing 50 kg or more, dosing can be initiated with 2 mg/day for 2 days. It can be increased to 5 mg/day for 5 days. The target dose is 10 mg/day. The dose may be increased to 20 mg/day. The dose adjustments can be increases to 5 mg/day at intervals of less than a week.
Agitation that is associated with schizophrenia and Bipolar disorder (Intramuscular Injection)
- Adults
The recommended dose is 9.75 mg. The recommended range is 5.25 -15 mg. A lower dose of 5.25 mg can be considered. If required a second dose can be increased to 30 mg/day. However, the efficacy of repeated doses of this injection in the agitated patients have not been evaluated.
The safety of daily doses greater than 30 mg has not been evaluated in the clinical trial. If with the continuous therapy still the agitation is recommended then oral drug must be given. The dose is 10 to 30 mg/day.
Aripirazole dosage for different forms
- Dosing administration of the injection
It must be given through the route of intramuscular only. Do not administer this drug subcutaneously and intravenously. Inject slowly and deep into the muscle mass. The drug product must be inspected thoroughly. It must be looked visually for the presence of any particulate matter or discoloration before administration.
- Dose of Oral Solution
The oral solution may be substituted in the place of tablets. It can be given to 25 mg. The patients that are receiving 30 mg tablets must take 25 mg of the solution.
- Dose of Oral Disintegrating Tablets
The dosing for an Oral disintegrating tablet is same as the oral dose of tablet
Ariprazole drug abuse and dependence
Abuse
Aripiprazole has not been studied in humans for abuse, physical dependence and tolerance. Patients must be analyzed for the history of drug abuse. Patients must be observed closely for signs of misuse or abuse.
Dependence
The clinical studies were conducted on monkeys for physical dependence studies. Withdrawal symptoms were observed on cessation of dose. The clinical trials have not revealed the tendency for drug-seeking behavior. However, these observations are not systematic. It is not possible to predict with this limited experience. It is very difficult to predict that for how much extent that this drug will be misused or abused.
What happens if a dose of aripiprazole is missed?
Take the missed dose as you remember. You must skip the missed dose if it the time for your next dose. You are required not to take the medicine to make up for the missed dose.
What happens if I overdose aripiprazole?
In the case, if you have taken an overdose of the medicine, then contact your doctor immediately.
The symptoms of overdose are
- Drowsiness, aggression, vomiting, confusion, fluctuations in heart rate, confusion, convulsions, trouble in breathing and fainting.
Aripiprazole side effects
The following list is the possible side-effects due to this drug. They are as follows.
- Agitation and feeling of restlessness.
- Uncontrollable movements of eyes, tongue, lips, arms, face and legs
- Mask-like look of the face,
- Trouble in swallowing and problems in speech
- Convulsions or seizures
- Thoughts of suicide
- The Reaction of Nervous system. The rigid muscles, sweating, fever, confusion, and fluctuations in the heart rate
- Low count of blood cells, Weakness ,chills, sore throat, mouth sores , swollen gums, skin sores, cold symptoms, and trouble in breathing
- High blood sugar that can lead to increase in thirst, increase urination, hunger, dry mouth, drowsiness, weight loss , and dry skin.
- Gain in weight
- Blurred vision
- Vomiting, constipation and changes in appetite.
- Dizziness, headache, drowsiness, and feeling tired
- Insomnia
For how long aripiprazole stays in the body?
The volume of distribution is 4.9 L/ Kg. It has the protein binding of >99%. It is metabolized by the liver. It gets eliminated through urine and excreta. Less than 1 percent of the unchanged drug is excreted in urine and about eighteen percent of the oral drug gets eliminated unchanged in the feces. It has a half -life of 75-146 hours.
Warnings and precautions of aripiprazole
- The USFDA has issued a warning for the use of Aripiprazole in elderly people with dementia and psychosis. Dementia is a condition that involves confusion, judgment, disorientation, intellect and loss of memory. Old people with dementia or Alzheimer disease if are treated with Aripiprazole can die in comparison of those who are not treated with these medicines. USFDA does not approve to treat dementia in the old patients. The caution must be taken before using Aripiprazole in the old patients with dementia.
- This drug may cause a life-threatening condition called NMS. It is known as a neuroleptic malignant syndrome. Symptoms of NMS are high fever, confusion, fluctuation in heart rate, blood pressure or pulse. The symptoms like stiff muscles, sweating and irregular heart rhythms can also result. If you have any symptom of NMS tell your doctor immediately.
- It can cause tardive dyskinesia. It is a condition that involves uncontrollable body movements. This condition may become permanent. The best way to avoid it is to tell your doctor. You must tell him if you notice any abnormal movements while taking Aripiprazole.
- The risk of stroke in older people can increase who take this drug for dementia.
- It causes an increase in the level of blood sugar and increases the risk of diabetes. Consult your doctor if you have the following signs of diabetes while taking Aripiprazole. The signs of diabetes are increased thirst, hunger and increased urination. Your blood sugar must be monitored carefully while taking Aripiprazole.
- It can cause a decrease in blood pressure, a condition called orthostatic hypotension. This can cause dizziness, or fainting. Consult and tell your doctor if you have any of symptoms while standing. It is dangerous in people with heart disease or CHF.
- Aripiprazole can increase the risk of epilepsy or seizures. Consult your doctor if you have epilepsy or seizures.
- It may cause difficulty swallowing that can result in inhalation of food that can cause pneumonia. Tell your doctor in the case of this problem.
- This drug can result in low white blood cells. Tell your doctor know if you have persistent infections. If you already have a low white blood cell count.
- Avoid taking alcohol with this drug.
Use of aripiprazole in pregnancy
AU TGA pregnancy category C: Drugs which, owing to their pharmacological effects, have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible.
Accompanying texts should be consulted for further details. US FDA pregnancy category C: Animal reproduction studies have shown an adverse effect on the fetus and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
In the animal studies, this drug has shown the developmental toxicity that includes the possibility of teratogenic effects in rabbits and rats.
Teratogenic effect
Pregnant rats were treated with the doses of 30, 10, and 3mg/kg/day for 10 times, 3 times, and 1 time respectively. Gestation was prolonged at 30 mg/kg. The treatment caused a delay in fetal development. It was proved by decreased fetal weight, undescended testes, and delayed skeletal ossification.
There were no adverse effects on embryo fetal. Delivered offspring weight was decreased with increased incidences of diaphragmatic hernia. Postnatal, delayed vaginal opening was also seen with impaired reproductive performance.
Some level of maternal toxicity was also seen. The pregnant rats that were receiving Aripiprazole injection of 3, 9, and 27 mg/kg/day during organogenesis were found to have decreased fetal weight and delayed skeletal ossification. It also caused some amount of maternal toxicity.
Pregnant rabbits were also treated orally with 10, 30, and 100 mg/kg/day with the frequency of 2, 3, and 11 times more than human exposure. It was found to have decreased food consumption in mother and increased abortions.
The treatment has resulted in increased fetal mortality, decreased fetal weight and increased risk of skeletal abnormality. In pregnant rabbits that received Aripiprazole injection in the dose of 3, 10, and 30 mg/kg/day at the time of organogenesis. It resulted in the decrease in fetal weight, decreased fetal skeletal ossification and increased in fetal abnormalities.
Non-teratogenic Effects
No adequate information and controlled studies are been done in pregnant women. It is not known that Aripiprazole can cause fetal harm when given to an expecting lady. The neonates exposed to antipsychotic drugs during the third trimester of pregnancy are at the risk of extrapyramidal or withdrawal symptoms after delivery.
There are many reports of agitation, hypertonia, tremor, respiratory distress, somnolence, and feeding disorder in the neonates. These complications vary in severity. Aripiprazole must be used during pregnancy only if the potential benefit can justify the potential risk to the fetus.
Labor and Delivery
The effect of Aripiprazole in the case of delivery and labor in humans is unknown.
Use of Aripiprazole in nursing mothers
Limited information indicates that maternal doses of aripiprazole up to 15 mg daily produce low levels in milk, but until more data become available, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. Aripiprazole can lower serum prolactin in a dose-related manner and can affect the milk supply variably.
Use of aripiprazole in pediatrics
The effectiveness and safety in pediatric patients with agitation or depression with schizophrenia or bipolar mania are not been established. The safety and effectiveness of Aripiprazole was established in 6-week, 202 pediatric patients that were 13 – 17 years of age. It were established in the placebo-controlled trial. The efficacy of maintenance drug has not been evaluated.
The safety and effectiveness of this drug for the kids suffering from bipolar mania were established. The placebo-controlled clinical trial was carried out in 197 pediatric patients of age between 10 to 17 years in four week duration. The efficacy of maintenance has not been evaluated.
The efficacy of this drug simultaneously with lithium or valproate for the treatment of mania in pediatric patients has not been evaluated.
The safety and effectiveness of this drug for the kids suffering from irritability with autistic disorder were established. It was established in the placebo-controlled clinical trial that was performed in 212 pediatric patients of the age group of 6 to 17 years in an 8 week study.
A maintenance trial was conducted in the age group of 6 to 17 years of age with irritability and autistic disorder. The pharmacokinetic of Aripiprazole in 10 -17 years of age is similar to those in adults.
Use of aripiprazole in geriatric use
During pharmacokinetic studies Aripiprazole clearance was found to be 20 percent lower in the elderly patients of age ≥65 years as compared to an adult of 18 – 64 years. After multiple doses, the pharmacokinetics of this drug in elderly patients is same as in young, healthy subjects.
In the clinical trials, 13,543 patients were treated with orally with Aripiprazole. 1073 were ≥65 years old and 799 were ≥75 years old. The 1073 patients were diagnosed with Dementia.
In the clinical trial where 749 patients were treated with Aripiprazole injection. 99 were ≥65 years and 78 were ≥75 years old. This drug was given to the person suffering from agitation with schizophrenia or bipolar mania. This placebo-controlled study was not having sufficient subjects of 65 years old. The number of older subjects were not sufficient to reach a particular conclusion.
Use of aripiprazole in renal Impairment
In the patients that are suffering from severe renal impairment, C max of dehydro-aripiprazole and Aripiprazole was increased by 36% and 53%. The renal excretion of both unchanged drug is less than 1% of the dose. The dose of 15 mg was given to these patients.
Use of aripiprazole in hepatic impairment
In a single-dose study of 15 mg of Aripiprazole in a patient suffering from liver cirrhosis, AUC of Aripiprazole decreased 20% in severe HI, increased by 31% in mild HI, and 8% in moderate HI.
Use of aripiprazole and different genders
The AUC and C max of Aripiprazole and its active metabolite are 40% – 30% higher in women than in men. The apparent oral clearance of Aripiprazole is lower in women. These differences are due to differences in body weight of men and women.
Use of aripiprazole and different race
However, no study was conducted to investigate the effects of Aripiprazole on the different races. The pharmacokinetic study has not revealed any race-related differences.
Can I take aripiprazole with alcohol?
This drug can make you drowsy. If you consume alcohol with this drug it can make you drowsier. You must not take alcohol with this medicine.
Can I take aripiprazole with aspirin?
Simultaneous administration of Aripiprazole with aspirin must not be taken. It can cause sedation, death, coma and respiratory depression. The patient must be closely monitored for the symptom of sedation and respiratory depression. You must tell your doctor if you are taking aspirin before starting the treatment of Aripiprazole.
Can I take aripiprazole with bupropion?
Combining these two medicines together can increase the risk of seizures. Bupropion can increase the level of Aripiprazole in the blood that can increase other side-effects.
Can I take aripiprazole with acetaminophen?
Using both these drugs together can increase the risk of dry mouth, blurred vision, and drowsiness, difficulty in urination, flushing, abdominal cramping, irregular heartbeat, and constipation, confusion, and memory problems. The side effects may increase with the age, use of alcohol. It is important to tell your doctor about the medicines that you are taking.
Can I take aripiprazole with codeine?
The simultaneous use of aripirazole with codeine must be avoided. It can result in respiratory depression, sedation, death and coma. The patients must be closely observed for the symptoms of respiratory depression. You must avoid driving the car or operating any kind of machinery.
You can suffer from more seizures in the case of old age, or taking alcohol. The condition can worsen, if you have the history of seizures, head trauma and brain tumor. Frequent medicines is required to monitor the situations.
Can I take aripiprazole with clozapine?
Simultaneous administration of both these drugs can cause side effects like respiratory or cardiac arrest and low blood pressure. These side effects occur before the initiation of the therapy. Other side effects are blurred vision, dizziness, dry mouth, low white blood cell count, constipation and irregular heart rhythm. Contact your doctor in the case you are taking clozapine. Your doctor can prescribe other alternatives or can do the dose titration.
Can I take aripiprazole with hydrocodone?
Both these drug together can result in respiratory depression, sedation, and death. You must tell your doctor so that he can monitor you.
Can I take aripiprazole with fentanyl?
You must avoid taking these drug together as it can result in the respiratory depression and coma. This combination can be fatal. You must ask your doctor to give you alternative medicines or dose titrations. In the case, if this medicine has to be given, then doctor must closely monitor the condition of the patient.
Can I take aripiprazole with methadone?
This combination if taken together increases sedation and drowsiness. You must avoid driving or operating any kind of machinery.
Can I take aripiprazole with oxycodone?
The combination must be avoided. Tell your doctor if you are feeling drowsiness or sedation. The patient must be closely monitored for any serious complications.
Can I take aripiprazole with tramadol?
This combination may be avoided as it can cause respiratory depression and sedation. In the more serious condition the patient may go into coma or death can also occur.
Can I take aripiprazole with topiramate?
This combination can cause increase in body temperature and reduce sweating. The person can suffer from heat stroke. You must talk to your doctor before starting this medicine with topiramate. The doctor can give you the alternate medicine or drug titration can be done.
Can patients with diabetes use aripiprazole safely?
The patients suffering from diabetes given the treatment of anti-psychotic like aripirazole can result in ketoacidosis, coma or death. Diabetic patients must be monitored with worsening of blood control when treated with aripirazole. You must go fasting blood sugar test when starting the treatment. You must also keep the check on your blood sugar level during the treatment.
The patient must be monitored for the following symptoms in the case of hyperglycemia. They are polyuria, polydipsia, weakness and polyphagia. In some cases, hyperglycemia gets resolved after discontinuation of the therapy. In other cases, anti-diabetic treatment is required after discontinuation of the therapy of aripirazole.
Can patients with high cholesterol levels use aripiprazole safely?
The therapy with aripirazole has been associated with changes in lipid levels. Before the initiation of the drug, you can go for fasting lipid profile test. Also, you are required to take the test on the regular basis to monitor fluctuation in the lipid level.
Can you get weight gain after aripiprazole use?
The cases of weight gain have been seen in the patients that are taking the therapy of aripirazole. Weight gain must be carefully monitored. You must check the weight before starting the therapy and after the therapy.