Contents
- What is G352 pill?
- Active ingredients of G352 pill and their identification
- G352 chemistry
- G352 pill uses
- Contraindications for G352 pill use
- G352 pill legal status
- What are different brand names for fenofibrate?
- G352 price and prescription
- G352 pill mechanism of action
- G352 pill side effects
- Can G352 pill cause dependence?
- G352 pill pharmacokinetics
- How long G352 pill stays in the system?
What is G352 pill?
G352 is an imprint on a pill identified as fenofibrate 160 mg. It is a white, capsule-shaped pill which is used a fibric acid derivative and is used to treat different types of hyperlipidemias. This pill is not a controlled substance. It is supplied by Global Pharmaceuticals.
- Imprint: G352
- Strength: 160 mg
- Color: White
- Size: 15.00 mm
- Shape: Capsule shaped
- Availability: Prescription only
- Drug Class: Fibric acid derivative
- Pregnancy Category: C (Risk cannot be ruled out)
- CSA Schedule: This is not a controlled drug
- Labeler / Supplier: Global pharmaceuticals
- Inactive Ingredients: croscarmellose sodium, hypromelloses, hypromellose 2910 (3 mPa.s), hypromellose 2910 (6 mPa.s), hypromellose 2910 (50 mPa.s), titanium dioxide, polyethylene glycol, polydextrose, triacetin, magnesium stearate, microcrystalline cellulose
Active ingredients of G352 pill and their identification
G352 pill contains only a single active ingredient, fenofibrate, in 160 mg strenght. Under ordinary conditions, Fenofibrate is a white crystalline powder which is stable.
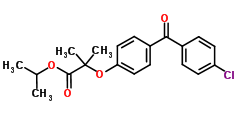
G352 chemistry
Fenofibrate:
IUPAC name: propan-2-yl 2-[4-(4-chlorobenzoyl)phenoxy]-2-methylpropanoate
Molecular formula: C20H21ClO4
Molecular weight: 360.834 g/mol
Molecular structure:
Drug class: Fenofibrate is an organic compound belonging to the group of benzophenones. These compounds are organic compounds contain a ketone which is attached to 2 phenyl groups.
G352 pill uses
Fenofibrate is used in the treatment of hyperlipidemias as a single drug or in combination with other drugs. The various type of hyperlipidemias it can treat are:
- hypertriglyceridemia
- Hyperlipoproteinemia: hyperlipoproteinemia type IIa, hyperlipoproteinemia type IIb, hyperlipoproteinemia type IV
- elevated LDL
- elevated VLDL
Although reduced levels of circulating lipids have known to reduce the risk for coronary artery disease, fenofibrate has not been shown to reduce the risk of CAD in the presence of diabetes mellitus type 2. So if an individual suffers for DM type 2, it is preferred to use another lipid-lowering agent.
Contraindications for G352 pill use
The use of G352 pill is contraindicated in the following conditions:
- Acute renal failure
- Chronic renal failure
- Acute hepatitis
- Chronic active hepatitis
- Liver cirrhosis
- Gallbladder disease
- Known hypersensitivity to fenofibrate
- Breastfeeding mothers
G352 pill legal status
G352 pill is not a controlled drug under the controlled substances act of 1970 because neither fenofibrate nor any of its inactive ingredients are implicated to cause addiction.
However its use is still regulated by your community or hospital pharmacist as this is a prescription only drug and not an over the counter drug.
What are different brand names for fenofibrate?
Fenofibrate is marketed by several different companies in the USA. Different brand names of fenofibrate 160 mg are: Fenoglide, Lofibra, Lipofen, Tricor. It is also available in generic form.
G352 price and prescription
Fenofibrate 160 mg is a moderately priced drug used to lower circulating lipids in the blood in individuals who are at risk of getting inflammation of the pancreas from having very high amounts of fat in their blood.
It is prescribed to only those patients who are unable to control their blood fats by dietary restrictions. This drug is more popular than comparable drugs.
Generic fenofibrate is covered by most insurance plans as well as the government funded Medicare. The average retail price of G352 pill is $72.55 but some pharmacies offer a discount.
On GoodRx, the lowest price for the most common version of fenofibrate is around $22.36 which is approximately 69% cheaper than the average retail price.
G352 pill mechanism of action
Fenofibrate causes the activation of peroxisome proliferator activated receptor a (PPARa). The activation of this receptor is linked to an increase activity of enzyme lipoprotein lipase.
This enzyme increases lipolysis and elimination of triglyceride-rich particles from plasma and production of apoprotein C-III is also decreased.
The fall in the level of triglycerides will result in an alteration in the composition and size of LDL from small, dense particles, to large buoyant particles. These large particles are catabolized rapidly as they have a higher affinity for cholesterol receptors.
Fenofibrate also increases HDL-cholesterol by reducing cholesteryl ester transfer protein (CETP) -dependent transfer of cholesterol from HDL to (V)LDL.
G352 pill side effects
Fenofibrate may cause following side effects:
Common side effects:
- Mild headache
- Nausea
- Constipation
- Runny nose
Serious side effects (require immediate treatment):
- Shortness of breath
- Chest pain and/or discomfort in jaw, neck, upper back, or arms
- Swelling of tongue or other mouth and throat problems
- Yellowish discolouration of the skin, nails, or sclera of the eyes due to jaundice (due to hepatotoxicity)
- Indigestion, heart burn, bloating, flatulence, vomiting
- Cold or clammy skin
- Tachycardia
- Swelling of the limbs
- Weakness, or pain of the muscles
- Red discolouration of urine
- Allergic reactions: from a simple rash to anaphylaxis to Steven Johnson syndrome
- Blisters of lips, mouth, eyes, ears, nose, or genital areas
- This will cause easy bruising, red pin-prick spots on the skin, gums bleeding when you brush your teeth
Can G352 pill cause dependence?
G352 pill does not cause dependence. As a result, it is not a controlled substance.
G352 pill pharmacokinetics
Absorption: The bioavailability of fenofibrate after oral intake is above 80%.
Distribution: The plasma protein binding of fenofibrate is very high and is around 99%. The volume of distribution of the drug is found to be 0.89 l/kg.
Metabolism: The drug undergoes hepatic metabolism in two steps. First, the entire fenofibrate is hydrolyzed by esterases to the active metabolite, fenofibric acid. As a result, no unchanged fenofibrate is detected in plasma.
Seond, Fenofibric acid then undergoes either one of the two reactions. Most of the drug undergoes conjugation with glucuronic acid.
A small amount of fenofibric acid is reduced to a benzhydrol metabolite which then undergoes conjugation with glucuronic acid.
Excretion: The glucoronidated metabolites are eliminated by both urinary and fecal routes. 60% to 70% of the drug is eliminated in urine while the remaining is excreted in feces.
How long G352 pill stays in the system?
The half-life of fenofibrate is recorded to be 20 hours. This means that after complete cessation of drug, 110 hours are needed to completely eliminate the drug from an individual’s body.
However, in patients with renal and hepatic problems, this time will be increased.
“U17 pill – Drug class, ingredients, uses, dosage and side effects“