Contents
- What is glimepiride?
- Glimepiride molecular structure, weight, formula, IUPAC name and drug class
- Mechanism of glimepiride action
- What are the indications and usage for glimepiride?
- Pharmacokinetics of glimepiride
- What are the side effects of glimepiride?
- Glimepiride overdosage
- Can glimepiride be given during pregnancy and breastfeeding?
- What are some drugs that can interact with glimepiride?
- Can glimepiride be taken with alcohol?
- Can glimepiride be taken with gatifloxacin?
- Drugs that can potentiate glimepiride hypoglicemic effects
- Can glimepiride be taken with aspirin?
- Can glimepiride be taken with atenolol?
- Can glimepiride be taken with ciprofloxacin?
- Can glimepiride be taken with warfarin?
- Can glimepiride be taken with esomeprazole?
- Can glimepiride be taken with furosemide?
- Can glimepiride be taken with ranitidine?
- Can glimepiride be taken with ibuprofen?
- Can glimepiride be taken with voriconazole?
- Can glimepiride be given to the patients with cardiovascular risk?
- Can glimepiride be taken with propranolol?
- Can glimepiride be given in renal or liver diseases?
- Can glimepiride be given to the patients with hypoglycemia?
- Can glimepiride be given to the patients with a G6Pd deficiency?
- Can glimepiride be given to the patients with hyponatremia?
What is glimepiride?
Glimepiride is first generation sulphonylurea with long duration of action. It is an oral diabetes medicine which helps in controlling the blood sugar levels. Along with diet and exercise, it is used to treat type-2 diabetes. Sometimes another medication such as insulin is also used in combination of glimepiride for the treatment of diabetes.
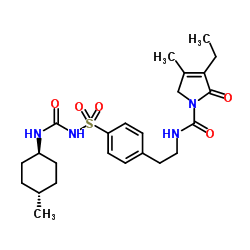
Glimepiride molecular structure, weight, formula, IUPAC name and drug class
Molecular structure:
Molecular formula: C24H34N4O5S
Molecular weight: 490.619g/mol
IUPAC name: 4-ethyl-3-methyl-N-[2-[4-[(4-methylcyclohexyl) carbamoylsulfamoyl] phenyl] ethyl]-5-oxo-2H-pyrrole-1-carboxamide
Drug Class: This compound belongs to the class of organic compounds known as benzenesulfonamides. These are organic compounds containing a sulfonamide group that is S-linked to a benzene ring.
Mechanism of glimepiride action
The mechanism of action of glimepiride in lowering blood glucose appears to be dependent on stimulating the release of insulin from functioning pancreatic beta cells, and increasing sensitivity of peripheral tissues to insulin.
Glimepiride likely binds to ATP-sensitive potassium channel receptors on the pancreatic cell surface, reducing potassium conductance and causing depolarization of the membrane. Membrane depolarization stimulates calcium ion influx through voltage-sensitive calcium channels. This increase in intracellular calcium ion concentration induces the secretion of insulin.
What are the indications and usage for glimepiride?
Glimepiride is indicated as an adjunct therapy with diet and exercise in order to improve the glycemic control in adults with type 2 diabetes mellitus.
Glimepiride should not be used for the treatment of type 1 diabetes mellitus or diabetic ketoacidosis, as it is not effective in the treatment of these indications.
Pharmacokinetics of glimepiride
In patients with type 2 diabetes mellitus, glimepiride showed peak plasma concentration in 2 to 3 hours after administration. The absorption of glimepiride reduces by 8% when given with meals. The pharmacokinetics of glimepiride does not differ between healthy subjects and in patients with type 2 diabetes.
Glimepiride possesses linear pharmacokinetics which is indicated by the fact that its clearance of Glimepiride after oral administration does not change over the 1 mg to 8 mg dose range. The volume of distribution of glimepiride was 8.8 L and total body clearance was 47.8 ml/min in healthy subjects, after intravenous administration. Protein binding was greater than 99.5%.
After intravenous or oral administration of glimepiride, the glimepiride is completely metabolized by oxidative biotransformation. The major metabolites are the cyclohexyl hydroxy methyl derivative (M1) and the carboxyl derivative (M2). Cytochrome P450 2C9 and other enzymes are responsible for the metabolism of glimepiride.
Following oral administration, approximately 60% of the total radioactivity was recovered in urine in a span of 7 days. The metabolites M1 and M2 are recovered in around 80% to 90% in urine. No parent drug was recovered from urine or feces. After intravenous dosing in patients, no significant biliary excretion of Glimepiride or its M1 metabolite was observed.
What are the side effects of glimepiride?
Glimepiride may cause changes in your blood sugar, which could cause blood sugar levels to fluctuate.
Symptoms of low blood sugar include:
- Sudden sweating
- Shaking
- Fast heartbeat
- Hunger
- Blurred vision
- Dizziness
- Tingling in the hands or feet
You may want to carry glucose tablets or gels with you to treat low blood sugar episodes. There are severe side effects of glimepiride. You should call your doctor immediately if you experience any of these side effects:
- Yellowing of the skin or eyes (Jaundice)
- Dark urine
- Light-colored stools
- Pain in the upper right part of the stomach
- Diarrhea
- Fever
- A Sore throat
- Unusual bruising or bleeding
Glimepiride overdosage
An overdosage of Glimepiride tablets, as with other sulfonylureas, can produce severe hypoglycemia. Mild episodes of hypoglycemia can be treated with oral glucose. Severe hypoglycemic reactions constitute medical emergencies requiring immediate treatment.
Severe hypoglycemia with coma, seizure, or neurological impairment can be treated with glucagon or intravenous glucose. Continued observation and additional carbohydrate intake may be necessary because hypoglycemia may recur after apparent clinical recovery
Can glimepiride be given during pregnancy and breastfeeding?
The use of glimepiride is not recommended during pregnancy. It comes under USFDA pregnancy category C which means that this drug is suspected to cause harmful effects on fetus without causing malformations. Instead of this medication, insulin is recommended during pregnancy in order to maintain blood glucose levels at normal.
The excretion of this drug in human milk is unknown still, breastfeeding is also not recommended during use of this drug.
What are some drugs that can interact with glimepiride?
There are certain drugs that affect the blood glucose level of your body and make them more difficult to control. If you are going to change, stop or even start a new medication, ask your pharmacist or doctor, prior to administration. You should strictly follow the instructions carefully if you are taking another anti-diabetic drug (like chlorpropamide) before stopping the previous medications.
The absorption can be reduced by the drug colesevelam. You will need to take glimepiride at least four hours before taking colesevelam. If you are taking any prescription, non-prescription, illegal, recreational, herbal, nutritional, or any other dietary drug, make sure that your doctor knows about it all.
There are many other medications that can interact with absorption and safety of glimepiride, therefore it is very important to let your doctor knows about all the other medications you are taking.
Can glimepiride be taken with alcohol?
There are certain side effects that can be caused by glimepiride which can worsen due to concurrent administration of alcohol with it. The combination of alcohol and glimepiride can cause severe symptoms such as flushing, headache, chest pain, nausea, vomiting, weakness, blurred vision, mental confusion, sweating, choking, difficulty breathing, and anxiety.
One should limit or avoid drinking alcohol while taking glimepiride. Glimepiride can make your skin more sensitive to sunlight, therefore, you should avoid unnecessary exposure to the sun while taking this medication. In patients with diabetes, alcohol may cause hypoglycemia or hyperglycemia.
During acute consumption of alcohol, the patient can suffer from hypoglycemia. When alcohol is administered on an empty stomach or after exercise, the blood pressure can be reduced even in small doses. The mechanism involves inhibition of both gluconeogenesis as well as the counter-regulatory response to hypoglycemia.
After ingestion of ethanol, episodes of hypoglycemia may last for even 8 to 12 hours. However, hyperglycemia and glucose tolerance can be impaired due to chronic alcohol abuse.
Moderate alcohol consumption generally does not affect blood glucose levels in patients with well-controlled diabetes. A disulfiram-like reaction (e.g., flushing, headache, and nausea) to alcohol has been reported frequently with the use of chlorpropamide and very rarely with other sulfonylurea.
In order to prevent serious consequences, patients with diabetes should avoid consuming alcohol if their blood glucose is not well controlled, or if they have hypertriglyceridemia, neuropathy, or pancreatitis. Patients with well-controlled diabetes should limit their alcohol intake to one drink daily for women and two drinks daily for men (1 drink = 5 oz wine, 12 oz beer, or 1.5 oz distilled spirits) in conjunction with their normal meal plan. Alcohol should not be consumed on an empty stomach or following exercise.
Can glimepiride be taken with gatifloxacin?
The co-administration of glimepiride and gatifloxacin is contraindicated as it may pose a major interaction. Gatifloxacin may interfere with the therapeutic effects of insulin and other antidiabetic agents.
The use of various quinolones has been associated with disturbances in blood glucose homeostasis possibly stemming from effects on pancreatic beta-cell ATP-sensitive potassium channels that regulate insulin secretion. However, hypoglycemia and hyperglycemia have been reported more frequently with gatifloxacin than with other quinolones.
Gatifloxacin-induced hypoglycemic episodes have generally occurred within the first 3 days of therapy and sometimes even after the first dose, while hyperglycemia usually occurred 4 to 10 days after initiation of therapy.
Death has been reported in severe cases. Severe refractory hypoglycemia and hypoglycemia coma can result from co-administration of gatifloxacin with sulfonylurea. Elderly patients and patients with reduced renal function are particularly susceptible.
The use of gatifloxacin is contraindicated in patients with diabetes mellitus. Other quinolones may be safer alternatives in such patients, although all quinolones should be used with caution.
Blood glucose should be closely monitored whenever quinolones are prescribed to patients receiving insulin or other antidiabetic agents, especially if they are elderly or have renal impairment.
One should make the patients learn to recognize the symptoms of hypoglycemia such as a headache, dizziness, drowsiness, nervousness, confusion, tremor, hunger, weakness, perspiration, palpitation, and tachycardia. Patients should initiate appropriate remedial therapy immediately, discontinue the antibiotic, and contact their physician if hypo- or hyperglycemia occurs during the quinolones therapy.
Drugs that can potentiate glimepiride hypoglicemic effects
Certain medications such as ACE inhibitors, amylin analogs, anabolic steroids, fibrates, monoamine oxidase inhibitors, nonsteroidal anti-inflammatory drugs, salicylates, selective serotonin reuptake inhibitors (SSRIs), disopyramide, propoxyphene, quinine, quinidine, and ginseng can potentiate the hypoglycemic effects of insulin secretagogues (eg. Sulfonylurea).
These drugs may increase the risk of hypoglycemia by enhancing insulin sensitivity (ACE inhibitors, fibrates, ginseng); stimulating insulin secretion (salicylates, NSAIDs, disopyramide, quinine, quinidine, MAOIs, ginseng); increasing peripheral glucose utilization (SSRIs, insulin-like growth factor); inhibiting gluconeogenesis (SSRIs, MAOIs, insulin-like growth factor); slowing the rate of gastric emptying (amylin analogs); and/or suppressing postprandial glucagon secretion (amylin analogs).
Or, they may increase the plasma concentration of insulin secretagogues by displacing them from plasma protein binding sites and/or inhibiting their metabolism (fibrates, NSAIDs, salicylates, sulfonamides). Clinical hypoglycemia has been reported during use of some of these agents alone or with insulin and/or sulfonylurea. Use of SSRIs has also been associated with loss of awareness of hypoglycemia in isolated cases.
Can glimepiride be taken with aspirin?
In order to manage the concurrent administration of glimepiride and aspirin, the close monitoring for the development of hypoglycemia is recommended if these drugs are co-administered with insulin secretagogues, particularly in patients with advanced age and/or renal impairment. The oral antidiabetic dosage(s) may require adjustment if an interaction is suspected.
Patients should be apprised of the signs and symptoms of hypoglycemia (e.g., headache, dizziness, drowsiness, nausea, hunger, tremor, weakness, sweating, palpitations), how to treat it, and to contact their doctor if it occurs. Patients should be observed for loss of glycemic control when these drugs are withdrawn.
Can glimepiride be taken with atenolol?
The psychologic response to hypoglycemia may be inhibited by the beta blockers. The patients who are on concurrent administration of these medications are usually cannot recognize the oncoming episode of hypoglycemia because the main symptoms such as tremor and tachycardia are absent.
In addition, usually with the noncardioselective beta-blockers (e.g., propranolol, pindolol, timolol), multiple effects on glucose metabolism have been reported, but occasionally also with relatively beta-1 selective agents (e.g., atenolol, metoprolol, nebivolol). In association with the beta blockade, inhibition of catecholamine-mediated glycogenolysis and glucose mobilization can potentiate insulin-induced hypoglycemia in diabetics.
It also delays the recovery of normal blood glucose levels. Prolonged and severe hypoglycemia may occur, although these events have rarely been reported. Significant increases in blood pressure and bradycardia can also occur during hypoglycemia in diabetics treated with insulin and beta-blockers due to antagonism of epinephrine’s effect on beta-2 adrenergic receptors, which leads to unopposed alpha-adrenergic effects including vasoconstriction. Other effects such as reduced glucose tolerance and decreased glucose-insulin secretion reported with the use of beta blockers.
Cardioselective beta blockers are considered safer as compared to noncardioselective antidiabetic agents. One should practice the precaution if such medications are prescribed to patients treated with insulin or oral antidiabetic agents that can cause hypoglycemia, for example, secretagogues.
Caution is advised because cardioselectivity is not absolute and higher doses of beta-1 selective agents can create the risk similar to nonselective agents. The patients should be advised to take care of the symptoms and should ask to monitor the regular blood glucose and they should be told about the hidden symptoms such as tremor and tachycardia and should be advised to keep an eye on such symptoms.
However, other symptoms such as a headache, dizziness, drowsiness, confusion, nausea, hunger, weakness, and perspiration may be unaffected. The same precautions are applicable in diabetic patients treated with ophthalmic beta-blockers
Can glimepiride be taken with ciprofloxacin?
The therapeutic effects of the insulin and other antidiabetic agents can be interfered due to concurrent administration of quinolone antibiotics. The use of certain quinolones such as clinafloxacin, gatifloxacin, temafloxacin, levofloxacin, and moxifloxacin has been associated with disturbances in blood glucose homeostasis possibly stemming from effects on pancreatic beta-cell ATP-sensitive potassium channels that regulate insulin secretion.
During the use of quinolones, hypoglycemia, as well as hyperglycemia (less frequently), has been reported. However, due to the underlying infections alone, a patient can suffer from hyperglycemia. Co-administration of sulfonylurea (most often glyburide) and/or other oral hypoglycemic agents with ciprofloxacin, norfloxacin, and especially gatifloxacin has occasionally resulted in severe, refractory hypoglycemia and hypoglycemic coma.
The more susceptible patients are old ones or the patients with impaired renal function. The hepatic metabolism of glyburide also gets inhibited by the ciprofloxacin. After one week of the ciprofloxacin therapy, the patient can suffer from the hypoglycemia that may be associated with elevated serum glyburide levels.
In order to manage the co-administration of quinolones and sulfonylurea, one should closely monitor the blood glucose level in patients (especially elderly ones or with impaired renal function) receiving insulin or other anti-diabetic agents.
The symptoms of hypoglycemia such as a headache, dizziness, drowsiness, nervousness, confusion, tremor, hunger, weakness, perspiration, palpitation, and tachycardia can occur if the patient is under concurrent therapy of ciprofloxacin (or other quinolones antibiotics) and sulfonylurea. The patient should be informed regarding such symptoms.
The patients should initiate appropriate remedial therapy immediately if hypo- or hyperglycemia occurs during quinolone therapy. Also, the patient should discontinue the antibiotic, and contact their physician
Can glimepiride be taken with warfarin?
Using warfarin together with glimepiride may increase the effects of glimepiride on lowering blood sugar. This could cause your blood sugar levels to get too low. Signs of low blood sugar include headache, hunger, weakness, dizziness, drowsiness, nervousness, sweating, confusion, and tremor.
Talk with your doctor before using warfarin together with glimepiride. You may need a dose adjustment or need to check your blood sugar more often to safely use both medications. It is important to tell your doctor about all other medications you use, including vitamins and herbs. Do not stop using any medications without first talking to your doctor.
Can glimepiride be taken with esomeprazole?
Esomeprazole is a benzimidazole proton pump inhibitor, which when administered with sulfonylurea (such as glimepiride), can enhance the sulfonylurea concentrations and also it can lead to enhanced hypoglycemic effects. The inhibition of cytochrome CYP 4502C19 enzyme or 3A4 hepatic metabolism may be the mechanism behind increased hypoglycemic effects.
Patients receiving this combination should be advised to regularly monitor their blood sugar, counseled on how to recognize and treat hypoglycemia. Also, a patient should notify their doctor if they notice any of these symptoms: a headache, dizziness, drowsiness, nausea, tremor, hunger, weakness, or palpitations). The sulfonylurea dosage may require a reduction in affected patients.
Can glimepiride be taken with furosemide?
Certain drugs such as atypical antipsychotics, corticosteroids, diuretics, estrogens, gonadotropin-releasing hormone agonists, human growth hormone, phenothiazines, progestins, protease inhibitors, sympathomimetic amines, thyroid hormones, asparaginase, copanlisib, danazol, diazoxide, isoniazid, megestrol, omacetaxine, pegaspargase, phenytoin, temsirolimus, as well as pharmacologic dosages of nicotinic acid and adrenocorticotropic agents are known to diminish the efficacy of insulin and other anti-diabetic agents.
Because these drugs can cause hyperglycemia, glucose intolerance, new-onset of diabetes mellitus and exacerbation of preexisting diabetes, they can also interfere with blood glucose control.
One should take precaution when these medications (which can interfere with glucose metabolism) are prescribed to patients suffering from diabetes. After initiation and discontinuation of these drugs are done, the close monitoring of glycemic control is advisable. One should adjust the dosage of the antidiabetic agents, if necessary.
If the blood glucose is consistently high in a patient, he/she should notify their physician. Symptoms such as excessive thirst and increase in the volume of the frequency of urination are mainly associated with hyperglycemia and should be notified immediately. Likewise, patients should be observed for hypoglycemia when these drugs are withdrawn from their therapeutic regimen.
Can glimepiride be taken with ranitidine?
H2 antagonists such as cimetidine and ranitidine may increase plasma concentrations of sulfonylureas and enhance hypoglycemic effects. The mechanism may be related to inhibition of liver cytochrome P450 enzymes responsible for sulfonylurea metabolism or increased absorption due to altered gastric pH. Cimetidine may also inhibit glucose metabolism. Other H2 antagonists may interact in a similar manner, particularly if increased pH is involved.
Can glimepiride be taken with ibuprofen?
Certain drugs such as ACE inhibitors, amylin analogs, anabolic steroids, fibrates, monoamine oxidase inhibitors (MAOIs, including linezolid), nonsteroidal anti-inflammatory drugs (NSAIDs), salicylates, selective serotonin reuptake inhibitors (SSRIs), sulfonamides, disopyramide, propoxyphene, quinine, quinidine, and ginseng potentiates the hypoglycemic effects of sulfonylurea (also called secretagogues). These different drugs act differently and increase the risk of hypoglycemia such as:
- Drugs like ACE inhibitors, fibrates, ginseng – enhance insulin sensitivity
- Drugs like salicylates, NSAIDs, disopyramide, quinine, quinidine, MAOIs, ginseng – stimulates insulin secretion
- Drugs like SSRIs, insulin-like growth factor- Increases peripheral glucose utilization
- Drugs like SSRIs, MAOIs, insulin-like growth factor: Inhibits gluconeogenesis
- Drugs like amylin analogs: slows the rate of gastric emptying and suppressing postprandial glucagon secretion
- Drugs like fibrates, NSAIDs, salicylates, sulfonamides: Increases the plasma concentration of insulin secretagogues (like glimepiride) by displacing them for plasma protein binding sites or inhibiting their metabolism.
Also when some of these agents used with sulfonylurea, clinical hypoglycemia has also been reported. Loss of awareness of hypoglycemia in isolates cases has also been associated with the use of SSRIs.
In order to manage the co-administration of sulfonylurea and NSAIDs, in patients with advanced age or in patients with impaired renal function, close monitoring for the development of hypoglycemia is advised. If an interaction is suspected, the adjustment of oral antidiabetic dose may be required.
The patients should be alerted for signs and symptoms of hypoglycemia (e.g., headache, dizziness, drowsiness, nausea, hunger, tremor, weakness, sweating, palpitations) in advance, They should also be informed about how to treat it and should contact their doctor immediately if any of these symptoms occur. In case of these drugs are to be withdrawn, the patient should be observed for loss of glycemic control.
Can glimepiride be taken with voriconazole?
CYP450 2C9 is the enzyme responsible for the metabolism of glimepiride, and azole antifungal agents such as fluconazole, voriconazole, and miconazole are the inhibitors of cytochrome P4502C9, therefore the coadministration of sulfonylurea hypoglycemic agents (glimepiride) with voriconazole can lead to increased plasma concentration of glimepiride.
There is sufficient pharmacokinetic data are available for chlorpropamide, tolbutamide, glipizide, glyburide, and glimepiride in combination with fluconazole. Another study, in which few postmenopausal women already receiving gliclazide or glyburide, were treated with low dose fluconazole for the treatment of vulvovaginal candidiasis, showed no effect on blood glucose control.
However, in this study, pharmacokinetics data were not involved. There have been rare reports of profound hypoglycemia and coma during treatment of a sulfonylurea with fluconazole or miconazole.
For the management of coadministration of azole antifungals (that inhibit CYP4502C9) and sulfonylurea, the caution is advised. The dose of the sulfonylurea should be adjusted as necessary and long with that, the blood glucose should be monitored.
There is increased the risk of hypoglycemia and patient should be informed regarding increased risk. Patients should also be alerted to potential signs such as a headache, dizziness, drowsiness, nervousness, confusion, tremor, hunger, weakness, perspiration, and palpitations.
Can glimepiride be given to the patients with cardiovascular risk?
The increased risk of cardiovascular mortality can be associated with the use of oral hypoglycemic agents. The risk of risk is high (with hypoglycemic agents) as compared with the treatment with diet alone or diet with insulin. A study was done on patients with non-insulin dependent diabetes, for checking the efficacy of glucose-lowering drugs in preventing or delaying cardiovascular complications.
They were treated with the sulfonylurea (such as tolbutamide or phenformin) for long-term (5 to 8 years). The patients were found to have more 2.5 times more mortality rate as compared to patients treated with diet alone. These findings result in discontinuation of both the treatments.
Despite controversy regarding interpretation of these results, particularly in the presence of underlying cardiovascular disease, the clinicians and patients should be aware of the potential risk when making treatment decisions for diabetes.
For hypoglycemic agents that belong to other classes, and other sulfonylurea or biguanides, sufficient data is not available. However, the caution should be applied due to similar structures or similar mode of action.
Can glimepiride be taken with propranolol?
Beta-blockers such as propranolol may inhibit some of the normal physiologic response to hypoglycemia caused by oral antidiabetic drug. Symptoms of hypoglycemia such as tremor and tachycardia may be absent, making it more difficult for patients to recognize an oncoming episode.
In addition, multiple effects on glucose metabolism have been reported, usually with the noncardioselective beta-blockers (e.g., propranolol, pindolol, timolol) but occasionally also with relatively beta-1 selective agents (e.g., atenolol, metoprolol, nebivolol).
Specifically, inhibition of catecholamine-mediated glycogenolysis and glucose mobilization in association with beta-blockade can potentiate insulin-induced hypoglycemia in diabetics and delay the recovery of normal blood glucose levels. Prolonged and severe hypoglycemia may occur, although these events have rarely been reported.
Significant increases in blood pressure and bradycardia can also occur during hypoglycemia in diabetics treated with insulin and beta-blockers due to antagonism of epinephrine’s effect on beta-2 adrenergic receptors, which leads to unopposed alpha-adrenergic effects including vasoconstriction. Other effects reported with various beta-blockers include decreased glucose tolerance and decreased glucose-induced insulin secretion.
Can glimepiride be given in renal or liver diseases?
The liver plays a major role in the metabolism of glimepiride as sulfonylurea are mainly metabolized by the liver into the metabolites which are excreted in urine and feces. Patients with impaired liver and/or renal function treated with sulfonylurea may be exposed to higher serum drug concentrations, which can increase the potential for severe hypoglycemic episodes induced by these agents.
In the presence of a hepatic impairment, gluconeogenic capacity may also be diminished, further compounding the risk. Therapy with sulfonylurea should be administered cautiously in patients with liver and/or renal disease.
Reduced dosages and longer intervals between dosage adjustments may be required. Hypoglycemia, if it occurs during treatment, may be prolonged in these patients because of slowed metabolism and/or excretion of the drugs.
Can glimepiride be given to the patients with hypoglycemia?
During the treatment with insulin or other oral hypoglycemic agents, there is increased the risk of hypoglycemia. In patients who are more susceptible to the development of hypoglycemic episodes while using these drugs, the extreme precaution is needed.
One should also practice the precaution in patients who are debilitated, malnourished, or those with a counter-regulatory mechanism (such as autonomic neuropathy, and adrenal or pituitary insufficiency) and also in those patients who are receiving beta-adrenergic blocking agents.
Can glimepiride be given to the patients with a G6Pd deficiency?
In patients with glucose 6-phosphate dehydrogenase deficiency, the sulfonylurea can cause hemolytic anemia. The glimepiride therapy should be used with precaution in a patient with G6PD deficiency, and one should look for another alternative of sulfonylurea.
However, in some patients receiving these drugs and did not have a known G6PD deficiency, the hemolytic anemia have been reported in post-marketing reports.
Can glimepiride be given to the patients with hyponatremia?
In patients who are on other medications or have medical conditions known to cause hyponatremia or increase release of antidiuretic hormone, the treatment with sulfonylurea may cause hyponatremia.
The syndrome of inappropriate antidiuretic hormone (ADH) secretion has been reported with certain sulfonylurea and these drugs may augment the peripheral (antidiuretic) action of ADH and/or increase the release of ADH. One should take precautions while treating patients with hyponatremia or at greater risk of developing hyponatremia such as elderly patients, patients taking diuretics or those who are volume-depleted.