Contents
- What is cefuroxime?
- Cefuroxime IUPAC name, molecular formula, weight, structure, and drug class
- Cefuroxime mechanism of action
- Cefuroxime indications
- Against which bacteria is cefuroxime effective?
- Cefuroxime pharmacokinetics
- What are the contraindications of cefuroxime?
- Warning and precautions associated with cefuroxime
- Use of cefuroxime in special population
- Adverse effects associated with cefuroxime use
- How Cefuroxime is administered?
- What are the possible drug-drug interactions with cefuroxime?
- Resistance to cefuroxime
- Information for patient during cefuroxime therapy
What is cefuroxime?
Cefuroxime is a generic name for second generation, semi-synthetic cephalosporin antibiotic. Cephalosporins are antibiotics that are closely related to penicillin antibiotics in structure as well as in function and are chemically known as 7-aminocephalosporic acid derivatives of penicillin.
All cephalosporins contain a six-membered ring with a sulfur atom and a ß lactam ring.
Cefuroxime is antibiotic drug resistant to beta-lactamase. It is a broad spectrum antibiotic effective against a wide range of gram-positive and gram-negative bacteria, gonorrhea, and haemophilus.
It was first discovered by Glaxo, now GlaxoSmithKline, and first marketed in 1978 with the name of Zinacef. Later it was approved by USA FDA in October 1983.
Cefuroxime becomes orally active when it is converted into is an acetoxyethyl-ester-prodrug of Cefuroxime which is effective orally.
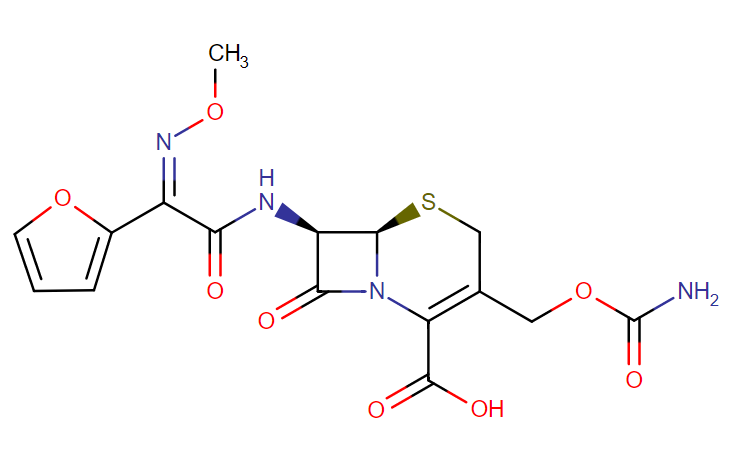
Cefuroxime IUPAC name, molecular formula, weight, structure, and drug class
IUPAC name: (6R,7R)-3-[(carbamoyloxy)methyl]-7-[(2Z)-2-(furan-2-yl)-2-(methoxyimino)acetamido]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid
Molecular formula: C16H16N4O8S
Molecular weight: 424.385 g/mol
Molecular structure:
Drug class: Cefuroxime belongs to the class of organic compounds known as cephalosporin 3′-carbamates which are are cephalosporins that are substituted at the 3′-position by a carbamate group.
Cefuroxime mechanism of action
Like other Cephalosporins, cefuroxime has the similar mode of action as other β-lactam antibiotics (such as penicillins), but are less affected by the β-lactamases enzyme.
β-lactamase enzyme tends to destroy β-lactam ring of β-lactam antibiotics (like penicillin) and thus bacteria become resistant to that antibiotic.
Cephalosporin destroys the cell wall formation of bacteria by disrupting the synthesis of the peptidoglycan layer that forms the bacterial cell wall. This peptidoglycan layer is essential for cell wall structural integrity.
The last step of transpeptidation in the synthesis of the peptidoglycan is facilitated by penicillin-binding proteins (PBPs).
PBPs proteins bind to the D-Ala-D-Ala at the end of muropeptides (peptidoglycan precursors) to crosslink the peptidoglycan.
Cefuroxime mimics the D-Ala-D-Ala site, irreversibly inhibiting PBP crosslinking of peptidoglycan resulting in a cell lysis that may occur, either through osmotic pressure or through the activation of autolysins. That’s why this group of drugs is considered as bactericidal.
The success of a penicillin antibiotic in causing cell death is related to the antibiotic’s size, charge, and hydrophobicity.
Penicillins are only effective against rapidly growing organisms that synthesize a peptidoglycan cell wall.
Consequently, they are inactive against organisms devoid of this structure, such as mycobacteria, protozoa, fungi, and viruses.
Cefuroxime indications
As it is mentioned that cefuroxime has very good efficacy against rapidly producing bacterias so it is usually indicated in acute cases of infection. Cefuroxime is primarily indicated in conditions like
- Acute bronchitis
- Bone and joint infection
- Chronic bronchitis
- Cystitis
- Early Lyme disease
- Genitourinary tract infections
- Gonococcal infections
- Gonorrhea
- Impetigo
- Infected wounds
- Late Lyme disease prevention
- Meningitis
- Muscle relaxation (long duration) for surgery and intubation
- NSAID-associated gastric or duodenal ulcers
- Otitis media
- Peritonitis
- Pharyngitis
- Pneumonia
- Post-operative infections
- Prophylaxis of acute rejection in allogeneic renal transplantation
- Pyelonephritis
- Respiratory tract infections
- Sinusitis
- Skin infections
- Soft tissue infections
- Surgical prophylaxis
- Urinary tract infection
Against which bacteria is cefuroxime effective?
Cefuroxime is active against following microorganisms:
Aerobic gram-positive:
- Pneumococci
- Streptococcus pyogenes
- Staphylococcus aureus
Aerobic gram-negative:
- Escherichia coli
- H. influenzae
- Moraxellacatarrhalis
- Haemophilusparainfluenzae
- Klebsiellapneumoniae
- Neisseriagonorrhoeae which produce beta lactamase
Spirochetel infection: Borreliaburgdorferi
Cefuroxime shows in-vitro activity against the following microorganisms:
Aerobic gram-positive:
- Staphylococcus epidermidis
- Staphylococcus saprophyticus
- Streptococcus agalactiae
Listeria monocytogenes, Enterococcus faecalis (strain of enterococci) and MRSA are not sensitive to cefuroxime.
Aerobic gram-negative:
- Proteus mirabilis
- Morganellamorganii
- Proteus inconstans
- Providencia rettgeri
Anaerobic microorganisms:
- Peptococcusniger
Cefuroxime pharmacokinetics
Absorption: After oral administration cefuroxime is well absorbed. Esterases enzymes, which are not present in the intestine and blood, hydrolyse it to the active form cefuroxime. Cefuroxime sodium is administered by the IM or IV route.
Cefuroxime has extensive tissue distribution. Inflammation of meninges allows it to pass into the CSF. It penetrates the placental barrier and is secreted during lactation.
Bioavailability of cefuroxime is 37% when taken with empty stomach. It increases up to 52% when taken with after food.
Distribution: Cefuroxime is readily distributed right through the extracellular fluids. Serum protein biding of cefuroxime is approximately 50%
Metabolism: The biological half-life of cefuroxime is about 80 minutes.
Excretion: Cefuroxime is excreted in unchanged through urine via renal route. In adults, approximately 50% of the administered dose is recovered in the urine within 12 hours.
As cefuroxime is eliminated via kidneys, the half-life is prolonged in patients with decreased renal function.
What are the contraindications of cefuroxime?
Cefuroxime is contraindicated in those patients in which there is possible chance of hypersensitivity due to cefuroxime.
Warning and precautions associated with cefuroxime
- Allergy: If some patients is allergic to medications that are similar to cefuroxime, then such patients should not take cefuroxime. An allergic reaction can be serious, and in some cases, it can be fatal (cause death) due to prophylactic shock. Always, discuss with your doctor to find out if you’re at risk of an allergic reaction.
- Clostridium difficile-associated diarrhea: usually it is advised not to use high doses of Cefuroxime, or not to use this drug for longer than 14 days, because this can lead to diarrhea. Clostridium difficile is the causative organism of such type of diarrhea.Most often, the diarrhea is mild to moderate. In rare cases, it can lead to fatal inflammation of the colon, the part of large intestine.
- Phenylketonuria: The oral suspension form of cefuroxime contains phenylalanine. This amino acid naturally occurs in many daily foods, such as eggs and meat. Cefuroxime should be avoided if some is suffering from phenylketonuria. Because in this condition phenylalanine is breakdown by the body thus phenylalanine got accumulated in body, it is then converted into phenylpyruvate, and the individual usually develops seizures, brain damage, and mental retardation.
Use of cefuroxime in special population
- For people with kidney problems: Cefuroxime is majorly eliminated from a renal route. If a patient is renal impair then Cefuroxime got accumulate in body and dose-related toxicity may occur. To avoid this dose adjustment of Cefuroxime in renal impaired patient is necessary.
- For pregnant women: Cefuroxime lies in pregnancy category B drug. Pregnancy category B means two things:
- Research in animals has not shown a risk to the fetus when the mother takes the drug.
- There aren’t enough studies done in humans to show if the drug poses a risk to the fetus.
- Discuss with doctor if you’re pregnant or planning to become pregnant. Animal studies do not always predict the way humans would respond. Therefore, this drug should only be used in pregnancy if clearly needed.
- For women who are breastfeeding: Cefuroxime passes into breast milk and may cause side effects in a child who is breastfed. Discuss with doctor if you breastfeed your child. You may need to decide whether to stop breastfeeding or stop taking this medication.
- If mother is suffering from phenylketonuria, use of cefuroxime should be avoided because mother milk is enriched with phenylalanin.
- Eldelry: As in older ages, functioning of kidneys slows down. This can cause a body to process drugs more slowly. As a result, more of a drug stays in your body for a longer time. This raises your risk of side effects and drug toxicity.
- Children: Cefuroxime should not be used in children younger than 3 months of age
Adverse effects associated with cefuroxime use
- watery or bloody diarrhea
- fever, chills, body aches, flu symptoms
- chest pain, fast or pounding heartbeats
- unusual bleeding
- blood in urine
- seizure (convulsions)
- pale or yellowed skin, dark colored urine, fever, confusion or weakness
- jaundice (yellowing of the skin or eyes)
- fever, sore throat, and headache with a severe blistering, peeling, and red skin rash
- skin rash, bruising, severe tingling, numbness, pain, muscle weakness
- increased thirst, loss of appetite, swelling, weight gain, feeling short of breath
- Painful or difficult urination, urinating less than usual or not at all.
Less serious side effects may include:
- nausea, vomiting, stomach pain, mild diarrhea, gas, upset stomach
- cough, stuffy nose
- stiff or tight muscles, muscle pain
- joint pain or swelling
- headache, drowsiness
- feeling restless, irritable, or hyperactive
- white patches or sores inside your mouth or on your lips
- unusual or unpleasant taste in your mouth
- diaper rash in an infant taking liquid cefuroxime
- mild itching or skin rash
- vaginal itching or discharge
How Cefuroxime is administered?
Cefuroxime is available in multiple strengths and dosage forms. It is available both as single ingredient as well as in combination. As single ingredient it is available in
Inj: 1 g, 1.5 g, 125 mg, 250 mg, 500 mg, 750 mg,
Inj IM/IV: 250 mg, 750 mg,
Susp: 125 mg/5ml, 250 mg/5ml,
Tabs: 125 mg, 250 mg,
Caps: 125 mg, 250 mg, 500 mg,
Adult dosage (ages 18 years and older):
The typical dosage is 250 mg every 12 hours for 10 days.
Child dosage (ages 13 to 17 years):
The typical dosage is 250 mg every 12 hours for 10 days.
Child dosage (ages 3 months to 12 years who can swallow tablets whole):
The typical dosage is 250 mg every 12 hours for 10 days.
Child dosage (ages 0 to 2 months):
Cefuroxime should not be used in children younger than 3 months of age.
What are the possible drug-drug interactions with cefuroxime?
There are some possible drug interactions when cefuroxime oral tablet is taken with other medications, vitamins, or herbs. An interaction is when a substance changes the way a drug works. This changes the overall working and effect of a drug.
Examples of drugs that can cause interactions with cefuroxime are listed below:
Oral contraceptives
Oral contraceptives (birth control pills) may not be absorbed well by the body after concomitant use with cefurozime. This means they may not work as well and pregnancy may occur. Other birth control management are required to avoid pregnancy during the use of cefuroxime.
Stomach acid drugs
Absorption of cefuroxime is reduced when after concomitant use with any antacids, due to the decrease in stomach acids. This means it may not work as well. Examples of these drugs include:
- antacids, such as:
- calcium carbonate
- magnesium hydroxide
- aluminum hydroxide
- H2-antagonists, such as:
- famotidine
- cimetidine
- ranitidine
- proton pump inhibitors, such as:
- lansoprazole
- omeprazole
- pantoprazole
Cefuroxime should be taken at least 1 hour before antacids are taken, or 2 hours afterward. H2 antagonists and proton pump inhibitors should be avoided during treatment with cefuroxime.
Probenecid is used to treat several conditions, including gout and kidney stones. Taking probenecid with cefuroxime increases the amount of cefuroxime in your body. This raises your risk of side effects. Your doctor will likely monitor you for side effects of cefuroxime if you take these drugs together.
Diuretic (water pill) such as bumetanide, furosemide, indapamide, hydrochlorothiazide, Lopressor, metolazone, spironolactone, torsemide, and others may effects the absorption of cefuroxime.
Resistance to cefuroxime
Resistance to cefuroxime may be caused by:
- Failure of the antibiotic to reach its site of action
- Alterations in the penicillin-binding proteins (PBPs)
This can lead to the binding of the cephalosporin to beta-lactamases which hydrolyzes the beta-lactam ring and inactivates the cephalosporin.
Destruction by hydrolysis of beta-lactam structure is the most predominant way of resistance to cefuroxime like other cephalosporins. Enormous amounts of Beta-lactamase are released by many gram-positive organisms.
The gram-negative bacteria produce decreased amounts of the beta-lactamase enzyme but its periplasmic position makes them more capable of destructing the cephalosporins.
Cefuroxime is more resistant to hydrolysis by the beta-lactamases formed by gram-negative bacteria than first-generation cephalosporins.
Information for patient during cefuroxime therapy
Before taking cefuroxime, inform the doctor if you are allergic to any drugs especially penicillins and other Cephalosporins, or if you have:
- kidney disease;
- liver disease;
- a history of intestinal problems, such as colitis;
- diabetes; or
- malnourished.
Any of the above conditions need dose adjustment for the safe use of cefuroxime.
The oral suspension (liquid) form of cefuroxime may contain phenylalanine. It is advised to take cefuroxime with caution if a patient is suffering from phenylketonuria (PKU).
FDA classified cefuroxime in pregnancy category B. This medication is not expected to be harmful to an unborn baby.
Cefuroxime can make birth control pills less effective, which may result in pregnancy. If you are taking contraceptive pills then inform the doctor about this prior to the use of Cefuroxime. Another form of birth control management is required in such cases.
Cefuroxime can bypass into breast milk and may harm a feeding baby. Do not use this medication without the advice of doctor if you are breast-feeding a baby.
“Can you take amoxicillin for chlamydia?”
“What is the drug terazosin used for? When should you take terazosin?“