Contents
- What is Cloderm topical?
- Cloderm molecular structure, weight, formula, class and IUPAC name
- Mechanism of action of Cloderm
- Pharmacokinetics of Cloderm
- What are the indications and usage of Cloderm?
- What are the contraindications of Cloderm?
- Can I use topical Cloderm for facial dermatoses?
- Can Cloderm be used during pregnancy?
- Can Cloderm be used in nursing mothers?
- How to use Cloderm in pediatric patients?
- What should I do in case of an overdose of Cloderm?
- What is the dosage and administration of Cloderm?
- Can topical Cloderm be used in diabetic patients?
- Can Cloderm be applied on diaper rash in children?
- Can Cloderm be used in ocular toxicities?
- What other drugs will affect the Cloderm topical?
- What are the side effects of the Cloderm?
- What are the precautions that should be taken while using Cloderm?
What is Cloderm topical?
Cloderm is a topical corticosteroid cream that contains clocortolone 0.1%. It is used to treat inflammation and itching associated with dermatoses and dermatitis by preventing the release of substances in the body responsible for inflammation. Clocortolone in topical form is beneficial in treating inflammation and itching caused by allergic reactions, eczema, and psoriasis.
You should not use clocortolone topical on a pediatric patient without consulting a doctor because it can cause severe side effects in children as large amount of medication get absorbed by the skin in children. The growth of children can also be get affected by the steroids. Consider talking to your doctor if you think your child is not growing at a normal rate while using this medicine.
Cloderm molecular structure, weight, formula, class and IUPAC name
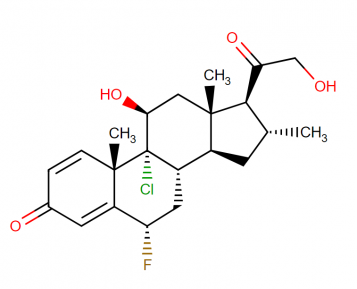
Molecular structure:
Molecular weight: 410.907 g/mol
Molecular formula: C22H28ClFO4
Drug class: Corticosteroids moderately potent group-II
This compound belongs to the class of organic compounds known as 21-hydroxysteroids. These are steroids carrying a hydroxyl group at the 21-position of the steroid backbone.
IUPAC name: (1R,2S,8S,10S,11S,13R,14S,15S,17S)-1-chloro-8-fluoro-17-hydroxy-14-(2-hydroxyacetyl)-2,13,15-trimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-3,6-dien-5-one
Mechanism of action of Cloderm
In general, the precise mechanism of the anti-inflammatory activity of topical steroids in the treatment of steroid-responsive dermatoses is uncertain. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins.
It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
These enzyme transcriptional changes are mediated by the drug binding first to the glucocorticoid receptor. This complex can migrate to the cell nucleus which then binds to DNA initiating genetic activation and repression of various genes.
Pharmacokinetics of Cloderm
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. Topical corticosteroids can be absorbed from the normal intact skin. The percutaneous absorption can be increased with inflammation or other disease processes.
The absorption from the percutaneous layer can also be increased by an occlusive dressing. Thus for the treatment of resistant dermatoses, occlusive dressing can be a valuable therapeutic adjunct.
Topical corticosteroids are handled through pharmacokinetic pathways, once absorbed through the skin, just like systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. And is metabolized by the liver and are excreted by the kidneys. Topical corticosteroids and its metabolites are also excreted into the bile.
What are the indications and usage of Cloderm?
Topical corticosteroids are indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses and dermatitis.
What are the contraindications of Cloderm?
Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
Can I use topical Cloderm for facial dermatoses?
- Cloderm topical can be used for the treatment of facial dermatoses. In a study, the efficacy and safety profile of Cloderm topical which contains clocortolone 0.1%, for the treatment of inflammatory facial dermatoses in young children, infants, and adults have been reported.
- Clocortolone is a mid-potency topical corticosteroid which is mainly indicated for the treatment of relief of inflammatory and pruritic conditions of dermatoses which is actively responsive to corticosteroids. Cloderm is also used extensively for the treatment of various corticosteroids responsive inflammatory dermatoses including facial skin in adults and children as well.
- As per a study, Cloderm was applied in patients with conditions like seborrheic dermatitis, contact dermatitis, scaling, pain, and pruritis of the facial skin.
- The Cloderm was applied for 21 days, three times daily.
- The treatment with Cloderm showed a reduction in erythema, edema, transudation, lichenification, scaling and pruritis, and pain in around 76% of patients.
- The excellent response to overall condition of the patients (68%) treated with Cloderm was noticed.
- Therefore the Cloderm cream proved effective in relieving the signs and symptoms of corticosteroid-responsive inflammatory dermatoses which involves the facial skin, including seborrheic dermatitis, contact dermatitis, atopic dermatitis, and psoriasis.
- Overall, the safety profile was favorable and devoid of any treatment-related serious adverse events.
Can Cloderm be used during pregnancy?
Cloderm is a topical corticosteroid preparation. Corticosteroids, when administered systemically at low doses, observed as teratogenic in animals tested in the laboratory. In animals, the dermal application in higher doses, corticosteroids have been shown to be teratogenic effects.
There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Cloderm comes under the pregnancy category C, therefore topical corticosteroids can be used in pregnancy but only if benefit outweighs the risk. However, it should not be used for long periods of time or in extensive amounts in pregnant patients.
Can Cloderm be used in nursing mothers?
The topical administration of corticosteroids that can result in detectable quantities in milk after sufficient systemic absorption has not known. If secreted in breast milk after followed by systemically administered corticosteroids does not cause any deleterious effect on the infants. However, the while administering this medication to a nursing mother, the caution should be exercised.
How to use Cloderm in pediatric patients?
As compared to mature or adult patients, pediatric patients can demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing’s syndrome due to large skin surface area and body weight ratio.
In children receiving topical corticosteroids such as Cloderm, hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported. Linear growth retardation delayed weight gain, and low plasma cortisol levels can occur as a result of manifestations of adrenal suppression in children.
In children, the least amount of topical corticosteroids which is compatible with an effective therapeutic regimen should be adopted. The growth and development of children could be altered due to continuing use of topical corticosteroids including Cloderm.
What should I do in case of an overdose of Cloderm?
Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects. Therefore there is the lesser chance of overdose in case of topical application of Cloderm, however, if you notice an unusual side effect of this medication, call your doctor right away.
What is the dosage and administration of Cloderm?
Cloderm cream that contains clocortolone 0.1%, should be applied to the affected areas for three times a day and rub it gently. In the management of psoriasis or recalcitrant conditions, occlusive dressings can be used. The use of occlusive dressings should be discontinued in case an infection develops. Instead, an appropriate antimicrobial therapy should be started.
Can topical Cloderm be used in diabetic patients?
Corticosteroid antagonizes and suppresses the action of insulin thereby increase the blood glucose level that results in inhibition of peripheral glucose uptake and increased gluconeogenesis. Topical corticosteroids rarely produce such effects still it should be used with extreme caution in patients with diabetes mellitus, glucose intolerance, or a predisposition to hyperglycemia.
Factors such as a vehicle, the concentration of the preparation, the size of the application area, the duration of administration, and the integrity of the skin, can alter the systemic absorption of the topical corticosteroids. In order to increase the percutaneous absorption, the occlusive dressing over the applied areas should be used.
Given equivalent doses, small children are generally at the greatest risk because of their larger skin surface to body mass ratios. In children, the use of highly potent agents such as betamethasone, clobetasol, diflorasone, and halobetasol should be avoided and also the use of Cloderm should be limited to small areas for 2 weeks only in adults.
Can Cloderm be applied on diaper rash in children?
For the treatment of diaper rash, the potent topical steroids such as clobetasol augmented betamethasone, diflorasone, and halobetasol should be not be used.
Because topical corticosteroids can be absorbed systematically that also depends upon the concentration of the preparation, vehicle, the size of application or affected area, and the duration of administration. Systemic absorption of topical steroids also depends upon whether or not occlusive dressing is used.
Due to the larger skin surface to body mass ratios in children, they are at greater risk for systemic toxicity such as adrenal suppression, Cushing’s syndrome and intracranial hypertension. It is advisable that medium to low potency corticosteroids should preferably be used if topical corticosteroids are necessary to treat a diaper rash.
The affected area should be kept open, and parents should be advised not to put tight-fitting clothes or plastic pants over the rash or affected area because occlusion of treated area can increase the drug absorption percutaneously.
Can Cloderm be used in ocular toxicities?
Posterior subcapsular cataracts and elevated intraocular pressure can be caused by prolonged use of topical steroids. The elevated intraocular pressure may lead to glaucoma or it can also damage the optic nerves. Topical corticosteroids should be used with extreme caution, however, the risk of such side effects are rare.
Therefore in patients with a history of cataracts, glaucoma, or increased intraocular pressure, the topical corticosteroids should be used cautiously, more potent agents such as augmented betamethasone, clobetasol, diflorasone and halobetasol should be used in the periorbital area.
Topical corticosteroids can be absorbed systematically that depends upon concentration of the preparation, vehicle, the size of application or affected area, and the duration of administration and whether or not occlusive dressings are used. Due to the larger skin surface to body mass ratios, the small children are generally at greater risk in given equivalent doses.
What other drugs will affect the Cloderm topical?
There are very less chances that other medications that you take orally or via other routes of administration will affect the topically applied Cloderm. However, there are many other medications which interact with each other.
You should always let your doctor know about other medications that you are already taking including your prescription medications, over-the-counter medications, vitamins, minerals or even herbal medications. While taking Cloderm, ask your doctor before starting a new medicine.
What are the side effects of the Cloderm?
- Most common reported and general side effects of Cloderm cream were burning, itching, dryness, and irritation.
- There were some endocrine side effects of Cloderm were also reported but the frequency of the same was not reported, such side effects are hypothalamic-pituitary-adrenal axis suppression, Cushing’s syndrome.
- Cloderm also can cause some side effects on nervous system, however, the frequency of the same has also been not reported, and such side effects include intracranial hypertension (including bulging fontanelles, headaches, and bilateral papilledema)
- Some metabolic side effects of Cloderm are hyperglycemia and glycosuria, and the frequency of the same has not been reported.
- Dermatological side effects of the Cloderm include folliculitis, : Folliculitis, hypertrichosis, acneform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, miliaria but the frequency of such side effects were also not reported.
- Some local side effects of Cloderm are burning, itching, dryness, and itching.
What are the precautions that should be taken while using Cloderm?
- If you are allergic to clocortolone, you should tell your doctor or pharmacist about it, also inform them about other corticosteroids (hydrocortisone, prednisone) if you are allergic to it. Cloderm may also contain some inactive ingredient which can cause severe allergic reactions, in case of any concern, ask your doctor or pharmacist directly. Talk to your doctor about your medical history especially if you have a history of poor blood circulation, diabetes, immune system problems before using this medication.
- If there is an infection or area which need to get treated is sore, avoid using this medication at such area.
- Rarely, using corticosteroid medication for a long time or over large areas of skin can make it more difficult for your body to respond to physical stress. Therefore you should tell your doctor or dentist, before undergoing any surgery, that you are using this medication or already have used this medication within the last few months.
- If you feel that height and growth of your child have not increased from long while using this medication, you should see a doctor without much delay, and the effect on final adult height is unknown.
- You should use this medication during pregnancy only if benefits outweigh the risk and should consult with your doctor before using this medicine. Similar is the case with nursing mothers, a nursing mother should not use this medication without prior consent with the doctor. However, this medicine does not observe to appear in the breast milk.