Contents
- What is Enoxaparin (Lovenox)?
- What is the generic and brand name of the drug?
- What is the source of the drug (natural or synthetic)?
- Why is this medication prescribed?
- Pharmacophore structure: Information about the chemical structure of the drug
- Chemical information of the drug.
- What is the available strength of the drug?
- How the medicine works (mode of action)?
- What are the recommended doses of Enoxaparin?
- When should I discontinue, withhold or modify the dose of Enoxaparin?
- What are the pharmacokinetic properties of the drug?
- Which pregnancy category (A; B; C; D; X) has been assigned to Enoxaparin?
- How to use the drug?
- How to store the drug?
- How to dispose the medicine?
- Does Enoxaparin has approval from government / FDA /or any other related agencies?
- Other uses of the drug.
- What special dietary precautions should I follow?
- What special precautions should I follow/ What should I avoid while using Enoxaparin?
- What are the possible side effects of this drug?
- What should I do in case of overdose?
- What should I do in case of missed a dose?
- Does Enoxaparin have any interaction with other drugs?
- Does Enoxaparin have any interaction with diseases?
- Where can I get more information?
- Clinical research and current scenario of the drug.
- References from chemical, biological and toxicological databases.
What is Enoxaparin (Lovenox)?
- Enoxaparin is a low molecular weight heparin (anticoagulant) that is used for the treatment of pulmonary embolism and deep vein thrombosis.
What is the generic and brand name of the drug?
- The drug is available under generic name Enoxaparin/Enoxaparin sodium and brand names Lovenox, Xaparin and Clexane.
- Enoxaparin is manufacture by Sanofi Aventis U.S. Llc, although a generic version of Enoxaparin is developed by Sandoz and Amphastar pharmaceuticals.
What is the source of the drug (natural or synthetic)?
- Enoxaparin is a semi- synthetic pharmaceutical antithrombotic agent (heparin benzyl ester), which is primarily derived from liver, lung, mast cells of vertebrates.
Why is this medication prescribed?
- Enoxaparin is an anticoagulant drug which has antithrombotic properties. It is commonly used to prevent the blood clotting in patients undergoing surgery.
- Enoxaparin substantially prevents Deep Vein Thrombosis (formation of blood clot within the deep vein usually in leg) in patients undergoing hip or knee replacement surgery and abdominal surgery.
- Enoxaparin is also used to reduce or prevent the occurrence of pulmonary embolism (medical complication characterized by blockage of the main artery or any branch of main artery of the lung) particularly in patients who are at higher risk for thromboembolic complications and undergoing surgery (hip or knee replacement or abdominal surgery).
- Enoxaparin also prevents the DVT in the patients who are confined to bed due to acute cardiac insufficiency, acute respiratory failure complicating chronic respiratory insufficiency and acute respiratory infections.
- Enoxaparin is also used along with aspirin to treat the complication of unstable Angina (pain in chest due to insufficient blood and oxygen supply in heart that may lead to heart attack) and non-Q wave Myocardial Infarction.
- The drug is also used as an anticoagulant in acute coronary syndromes (ACS).
- Clinical studies suggested that use of Enoxaparin decreases the risk of blood clot formation and maintains the blood flow to the heart in patients suffering from angina or heart attack. In this manner, Enoxaparin significantly improves the long-term mortality and morbidity.
Pharmacophore structure: Information about the chemical structure of the drug
- Enoxaparin chemically is a low molecular weight heparin substance that belongs to the class of organic highly acidic compounds, known as mucopolysaccharide. Enoxaparin is made up of sulfated D-glucosamine and D-glucuronic acid moiety joined together with sulfaminic bridges. Chemically, these are carbohydrate molecule, which contain a 2-O-sulfo-4-enepyranosuronic acid group at non-reducing end and 2-N,6-O-disulfo-D-glucosamine at reducing end of chain. The detailed chemical classification of Enoxaparin is described below:
| Kingdom | Organic compounds |
| Super Class | Organooxygen compounds |
| Class | Carbohydrates and carbohydrate conjugates |
| Sub Class | Oligosaccharides |
| Direct Parent | Polysaccharide derivatives |
Chemical information of the drug.
- The pharmaceutical available form of Enoxaparin is Enoxaparin sodium (sodium salt).
- The Enoxaparin sodium is obtained by alkaline depolymerization of heparin benzyl ester derived from vertebrate intestinal mucosa.
- It is a low molecular weight heparin, which is represented with a molecular formula (C26H40N2O37S5)n.
- The average molecular weight of the Enoxaparin sodium ranges between 3800 to 5000 daltons. Distribution of molecular weight is as follows:
<2000 Daltons ≤20%
2000 to 8000 Daltons ≥68%
>8000 Daltons ≤15%
- Chemically, Enoxaparin is 6-[6-[6-[5-acetamido-4,6-dihydroxy-2-(sulfooxymethyl)oxan-3-yl]oxy-2-carboxy-4-hydroxy-5-sulfooxyoxan-3-yl]oxy-2-(hydroxymethyl)-5-(sulfoamino)-4-sulfooxyoxan-3-yl]oxy-3,4-dihydroxy-5-sulfooxyoxane-2-carboxylic acid.
- Enoxaparin sodium is fine white powder, solid in texture and has a water solubility of approximately 200mg/mL.
- Enoxaparin sodium is practically insoluble in acetone, ethanol or chloroform.
- Enoxaparin aqueous solution (10%) has a pH of 6.2 to 7.7.
What is the available strength of the drug?
- Enoxaparin is available as a sterile aqueous solution (injection) for intravenous or subcutaneous administration.
- Enoxaparin should not be used for intramuscular administration.
- It is available in two different dosage strength of 100 and 150 mg/mL.
- The drug is available in the form of ampoules (30mg/0.3mL), prefilled syringes (30mg/0.3mL and 40mg/0.4mL), graduated prefilled syringes (60mg/0.6mL, 80mg/0.8mL and 100mg/mL) and multiple dose vials (300mg/3mL).
- Ampoules, prefilled syringes and graduated prefilled syringes contain no preservatives and for single dose injection only.
- Benzyl alcohol (15m/mL) is used in multiple dose vials as a preservative agent.
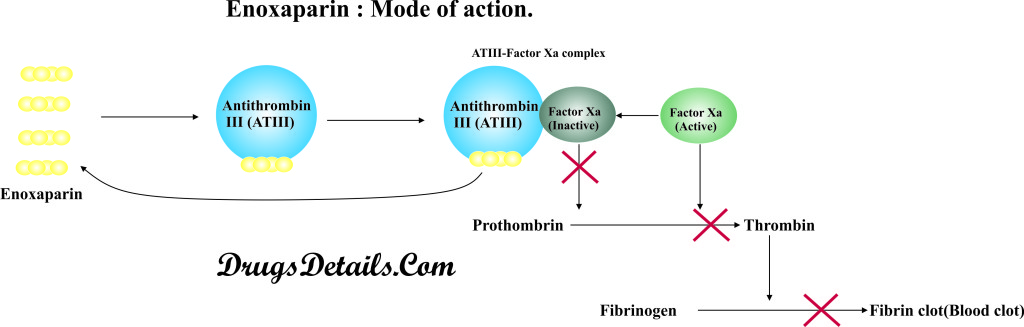
How the medicine works (mode of action)?
- The mode of action of Enoxaparin is similar to that of other drugs known as Factor Xa inhibitors.
- The active ingredient of the drug is Enoxaparin sodium that binds specifically to antithrombin III (ATIII).
- Binding of Enoxaparin to antithrombin III results in an increased activity (neutralization of Factor Xa) of antithrombin III.
- Following activation, antithrombin III (ATIII) brings about the inhibition of coagulation factor Xa and IIa.
- Neutralization of Factor Xa results in reduced conversion of prothrombin to thrombin, and fibrinogen to fibrin in a sequential manner, which ultimately inhibits the blood clot formation.
- Furthermore, prevention of activation of fibrin stabilizing factor inhibits the formation of stable fibrin clot.
- In this manner, Enoxaparin has no direct inhibitory effect on coagulation factor Xa and IIa, rather it potentiate the inhibition of coagulation factor Xa and IIa.
What are the recommended doses of Enoxaparin?
The prescribed dose of Enoxaparin varies depending upon the age and diseased state of the patient.
- In case of patients undergoing abdominal surgery.
- The prescribed dose of Enoxaparin is 40 mg per day to be injected subcutaneously.
- The initial dose should be administered 2 hours prior to surgery.
- The drug should be administered for a period of 7-10 days.
- Treatment of Acute ST-segment elevation myocardial infarction (STEMI).
- The prescribed Enoxaparin dose is single 30 mg intravenously + 1mg/kg subcutaneously followed by 1mg/kg every 12 hour.
- All patients should receive Aspirin (75-325 mg once a day) unless contraindicated.
- Treatment should be started at the earliest following diagnosis.
- The drug treatment should be continued for a period of 8 days or until hospital discharge.
- Geriatric dose (≥75):
- 75mg/kg every 12 hour subcutaneously.
- Treatment of Deep Vein Thrombosis (DVT) and Pulmonary Embolism(PE).
- The recommended dose of Enoxaparin to be administered subcutaneously twice per day at 12 hour interval:
- DVT without PE: 1mg/kg every 12 hour
- DVT with PE: 1mg/kg every 12 hour or 1.5mg/kg once a day at same time everyday
- The drug treatment should be continued for at least a period of 5 days or up to 17 days in controlled clinical conditions.
- Paediatric dose:
- Up to 2 months: 1.5mg/kg every 12 hour subcutaneously
- 2 months to 17 years: 1mg/kg every 12 hour subcutaneously
- In case of patients undergoing Hip or Knee Replacement Surgery:
- The recommended initial dose of Enoxaparin for hip or knee surgery is 30mg every 12 hour to be administered subcutaneously prior to 12-24 hour to surgery and for hip replacement surgery is 40 mg subcutaneously once a day, 12 hour prior to surgery.
- After initial dose, 40 mg once a day to be administrated subcutaneously.
- The drug treatment should continue for a period of 7 to 10 days or until clinical stabilization.
- Treatment of unstable angina/non- Q-Wave Myocardial Infarction:
- The prescribed Enoxaparin dose is 1mg/kg every 12 hour administered by subcutaneous injection with oral Aspirin therapy (100 to 325 mg once a day).
- Treatment should be started as early as possible following the disease diagnosis.
- The drug treatment should continue for a period of 2 to 8 days or until clinical stabilization.
When should I discontinue, withhold or modify the dose of Enoxaparin?
- The dosing of the drug may vary depending upon the diseased state and the body weight of the patient.
- No dosage adjustment is necessary in mild to moderate hepatic impairment patients.
- The drug is contraindicated in case of pregnancy, breastfeeding or hypersensitive response to any component of the drug.
- Enoxaparin multiple dose vial should not be used in case of newborns because it contains benzyl alcohol which is related with potentially lethal Gasping syndrome (characterized by central nervous system depression,gasping respirations, metabolic acidosis, and high levels of benzyl alcohol and its metabolites in the blood and urine).
- Consult to your doctor if you receive spinal anaesthesia or experience spinal tap because Enoxaparin can cause severe blood clot around spinal cord.
- Avoid the medication of Enoxaparin if you taking non steroidal anti-inflammatory drug (Diclofenac, Meloxicam, Naproxen or Ibuprofen) or blood thinners such as Coumadin (Warfarin).
- Avoid the use of Enoxaparin if you have creatinine clearance less than 20mL/min.
- Enoxaparin use is contraindicated in case of
- Acute bacterial endocarditis
- Major blood clotting disorders
- Gastric or duodenal ulcer
- Severe hypertension
- Diabetic or hemorrhagic retinotherapy
What are the pharmacokinetic properties of the drug?
- Pharmacokinetic studies suggested that after intravenous or subcutaneous administration, Enoxaparin is rapidly absorbed and has a bio-availability of approximately 92%.
- It has been observed that following a 20 mg and 40 mg dose of Enoxaparin, maximum (or peak) plasma concentration (1.58µg/mL and 3.83µg/mL respectively) is achieved in 3 to 5 hours in the fasted state.
- Following absorption the majority (80%) of the drug is bound to proteins (albumin).
- The average median half-life of Enoxaparin is 4.5 hours.
- The drug is mainly metabolized in the liver by the desulfation and depolymerisation.
- Enoxaparin is eliminated mainly in the urine as active (total 40%) and non-active(10%) fragments of the dose.
- The average steady state volume of distribution of the Enoxaparin is approximately 4.3 L.
Which pregnancy category (A; B; C; D; X) has been assigned to Enoxaparin?
- The Enoxaparin is classified by US FDA pregnancy category: B
- Due to lack of adequate and well-controlled studies the use of Enoxaparin in pregnant women is contraindicated and recommended only when benefit justifies the risk.
- Laboratory animal studies have shown no adverse effects on the fetus.
- No adequate data is available on excretion of Enoxaparin into human breast milk.
- Despite these facts caution should be exercised when taking Enoxaparin.
How to use the drug?
- Follow the instructions carefully as directed on prescription leaflet and take Enoxaparin exactly as directed.
- Enoxaparin is available as injectable solution. Learn proper technique from the doctor how to use it in case you use it at home.
- The drug should not be used if it turns cloudy, discolored or contains particle and syringe gets cracked or damaged.
- The drug is taken twice per day and should be injected subcutaneously (under the skin of the stomach) or intravenously (in veins) and not into muscle.
- The injection should be given at different sites every time to decrease the injury.
- Try to take the medicine at the same time every day.
- No other medication should be co-administered with the drug in the same injection.
- Take the medication regularly, even if you feel well.
- Do not change the dose of the drug as prescribed by your doctor. Since, the dosage is based on patient medical condition treatment, responses and weight.
How to store the drug?
- Enoxaparin is stored at room temperature 25°C (77°F).
- Brief excursion period is permitted to 15-30°C (59-86°F).
- Store the medicine away from direct sunlight, excess heat and moisture.
- The drug should be kept away from children and pets.
- Do not freeze or store the medicine at extreme cold too.
How to dispose the medicine?
- Throw away unused and opened, outdated or no longer used container.
- Also dispose the old medicine after the expiration date.
- The drug should neither be flushed down in the toilet nor poured in the drain.
- Enoxaparin should not be disposed in fire because it produces toxic fumes of nitrogen and sulphur dioxides, carbon dioxide and carbon mono-oxide.
- Consult your pharmacist or local waste disposal company for proper disposal.
- Enoxaparin had received the marketing permission (authorisation holder: Sanofi-Aventis U.S.) by US in 1993 as a blood thinning drug for intravenous and subcutaneous use.
- On 23 July, 2010, U.S. Food and Drug Administration (FDA) has approved the first generic version of Enoxaparin (Lovenox) as an anticoagulant drug used for multiple indications including prevention of Deep Vein Thrombosis (DVT).
Other uses of the drug.
- Enoxaparin in combination with aspirin is also used to treat the acute ST-segment Elevated Myocardial Infarction (STEMI) and patients of STEMI which managed medically or with subsequent Percutaneous Coronary Intervention (PCI).
- Enoxaparin is also used in combination with Warfarin (Coumadin) to treat blood clots in leg.
- Enoxaparin may also be used for other uses not listed here. It is advisable to ask your doctor or pharmacist for more information.
What special dietary precautions should I follow?
- It is generally recommended to continue with the normal diet, unless and until asked by your doctor.
What special precautions should I follow/ What should I avoid while using Enoxaparin?
- Do not use the medicine if you are hypersensitive or allergic to any of the ingredients.
- Before taking Enoxaparin, tell your doctor about your medical history preferentially if you have any kind of kidney disease, infection in heart, a stroke, bleeding disorders, ulcers or low platelet count.
- Tell to your doctor if you are having any surgery.
- Consult with your doctor and pharmacist if you are taking any prescription and nonprescription medications, and herbal products.
- During pregnancy this medication is recommended only when it is essential and under doctor or pharmacist supervision.
- Since the information about excretion of drug in milk is not explored consult your doctor before breast feeding to your child.
- Consumption of alcohol should be avoided while taking Enoxaparin as it increases stomach bleeding.
- Do not share this medication with other persons having the similar kind of problems. Consult your doctor for more details.
- The drug should be used with high caution in older people.
- It is generally recommended to avoid activities that lead to increased risk of bleeding or injury. Extra precautions should be taken to prevent bleeding while brushing or shaving.
What are the possible side effects of this drug?
In addition to the associated benefits, Enoxaparin also is accompanied with the side effects some of which are more common, others less common whereas some that fade away with time while you take the drug. It is always recommended to consult a doctor if you encounter any of the side effects.
Some more common side effect of Enoxaparin which require medical attention is as follows:
- Black or red, tarry stools
- Bleeding gum
- Bleeding nose
- Blood in cough
- Dark brown or red urine
- Difficulty in breathing or swallowing
- Dizziness and headache
- Increased vaginal bleeding or menstrual flow
- Paralysis
- Shortness of breath
Some of the less commonly occurring side effects but requiring medical attention is outlined as:
- Blood collection under the skin
- Burning or pain during urination
- Confusion
- Continuing bleeding from surgical wound or nose or mouth
- Discomfort and tightness in chest
- Fever
- Irritation
- Pain in lower part of back
- Swelling in feet or hands
- Uncontrolled bleeding at the site of injection
Some side effect of Enoxaparin was rare but they also require medical attention are:
- Bladder function problems
- Bronchial secretions thickening
- Cough and sneezing
- Decreased urine output
- Dilated neck veins
- Dizziness
- Fainting
- Feeling of illness and discomfort
- Hives or skin rashes
- Irregular breathing
- Irregular or fast heartbeat
- Pain in chest
- Sensation of burning, tickling or prickling
- Sore throat
- Swelling of the fingers, feet, face, mouth, tongue or genitals
- Weakness in legs
- Weight gain
There are some adverse effects that disappear while consuming the drug with time. But, if any of them keep on persisting, consult your healthcare provider or pharmacist.
- Diarrhoea
- Redness, pain or irritation at the place of injection
Beside these, Enoxaparin may also be associated with some side effects which are developed after the long medication and may be rarely observed or may persist for long time. On appearing of these symptoms, immediately call your pharmacist or doctor. These include:
- Thrombocytopenia
- Eosinophilia
- Hemorrhagic anemia
- Osteoporosis
- Skin necrosis
- Neurological injuries including long term or permanent paralysis due to spinal hematoma
What should I do in case of overdose?
- Overdose of Enoxaparin may cause bruising and excessive bleeding.
- Equal dose of Protamine sulphate (1% solution) is intravenously injected to neutralize the Enoxaparin (100mg/mL Protamine sulphate is used to neutralize 100mg/mL Enoxaparin dose). Complete neutralization of anti-factor Xa activity can never be achieved (maximum 60%).
- If you overdose the drug contact with your doctor or pharmacist for symptomatic and supportive measures.
- Seek emergency medical attention or call the Poison Help line at 1-800-222-1222.
What should I do in case of missed a dose?
- In case of missed dosage, take it as soon as you remember and maintain a regular dosing schedule.
- Keep in mind to not use a double dose to make up a missed dose.
- Skip the missed dose if it is almost time for your next scheduled dose.
Does Enoxaparin have any interaction with other drugs?
It has been observed that Enoxaparin may interact with or increase or decrease the effect of following drugs. Coadministration of these drugs with Enoxaparin may enhance the risk of hemorrhage. Hence, when taken together they can result in harmful side effects. It is always advisable to consult your doctor in case you are taking:
- Medicines that bring about in reduction in the blood clot formation chances such as warfarin (Coumadin), cilostazol (Pletal), dabigatran (Pradaxa), ticagrelor (Brilinta), dipyridamole (Aggrenox, Persantine), rivaroxaban (Xarelto), heparin, abciximab (ReoPro), enoxaparin (Lovenox), apixaban (Eliquis), fondaparinux (Arixtra), clopidogrel (Plavix), prasugrel (Effient), anagrelide (Agrylin), and dalteparin (Fragmin).
- Platelet inhibitors including dipyridamole, or sulfinpyrazone.
- Aspirin and other salicylates such as Tricosal, Knee Relief, Kaopectate, Pamprin Cramp Formula, Caplet, Pepto-Bismol, Nuprin Backache, and Trilisate etc.
- Medicines that dissolve blood clots like tenecteplase (TNKase), reteplase (Retavase), and alteplase (Activase).
- Nonsteroidal anti-inflammatory medicines (NSAIDs) including, naproxen, piroxicam, oxaprozin, celecoxib, indomethacin, diclofenac , sulindac and ketorolac etc.
- Medicines for treatment of cancer such as tositumomab (Bexxar), ibritumomab (Zevalin), and dasatinib (Sprycel).
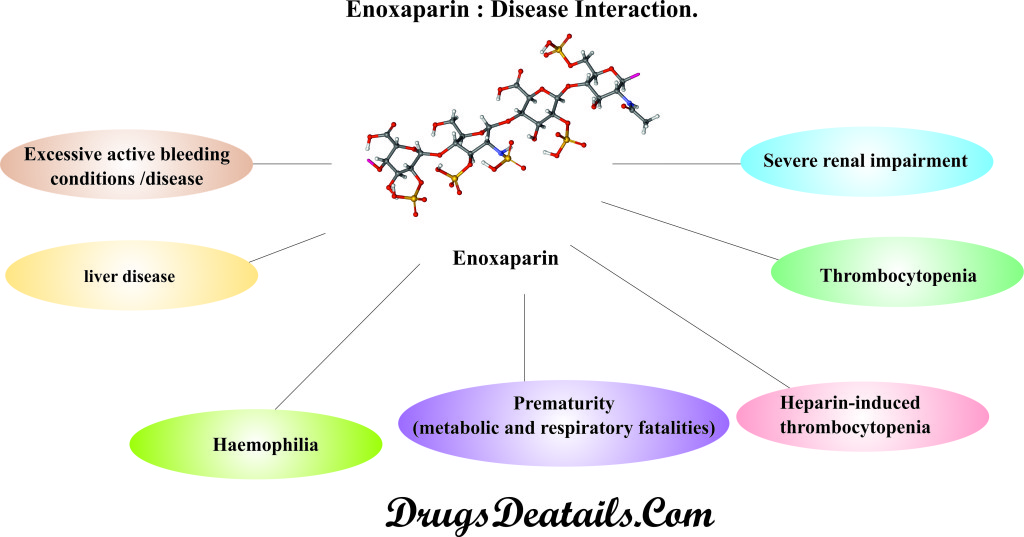
Does Enoxaparin have any interaction with diseases?
Prior to initiation of Enoxaparin therapy, it is necessary to discuss your medical condition or allergies you have or any other significant medical history with your doctor. It has been observed that following medical conditions (disease) may interact with Enoxaparin:
- Active Bleeding: Clinical studies suggested that use of Enoxaparin or heparin and its derivative is highly contraindicated in patients with excessive active bleeding conditions such coagulation defect, peptic ulcer, colonic ulceration, ulcerative colitis, diverticulitis, hypertension, myeloproliferative disorders, hemophilia, thrombocytopenia, liver disease, infectious endocarditis, and abnormal uterine bleeding. Under these situations use of Enoxaparin may cause serious consideration of a hemorrhagic event. It is usually recommended to perform blood coagulation test at appropriate intervals during full-dose heparin administration to prevent the incidence of hemorrhagic event.
- Renal Dysfunction: It has been observed that Enoxaparin is primarily eliminated by urinary excretion and hence, the exposure to Enoxaparin in patients with renal impairment is significantly higher than the normal individuals. In this view, risk of bleeding is significantly higher when Enoxaparin is used in patients with severe renal impairment.
- Thrombocytopenia: The use of Enoxaparin is highly contraindicated in patients with Thrombocytopenia (a disorder characterized by deficiency of platelets in the blood). It has been reported that incidence of thrombocytopenia can occur up to 30% of the total patients receiving heparin. It has been recommended to undergo regular platelet counts before and periodically during heparin administration and therapy should be discontinued if the count falls below 100,000/mm3.
- Heparin-induced thrombocytopenia (HIT)/ heparin-induced thrombocytopenia and thrombosis (HITT): Heparin-induced thrombocytopenia (HIT) is referred to anti-heparin antibody-mediated reaction which causes irreversible aggregation of platelets. In advance situation HIT lead to the development of venous and arterial thromboses, which is commonly known as heparin-induced thrombocytopenia and thrombosis (HITT). It has been observed that occurrence of HIT and HITT may be reported up to several weeks after the discontinuation of heparin (Enoxaparin) therapy. Therefore, it is advisable to monitor the patients for HIT and HITT, if symptom of thrombocytopenia or thrombosis appears following discontinuation of heparin.
- Prematurity: Some of the pharmaceutical preparation of Enoxaparin (low molecular weight heparin) such as multiple dose vials contains benzyl alcohol as preservative. It has been reported that presence of benzyl alcohol (in endotracheal tube lavage solutions and bacteriostatic saline intravascular flush) is associated with severe metabolic and respiratory fatalities in new born low birth weight premature infants. Therefore, it is recommended to avoid the products which contain benzyl alcohol as preservative in premature infants or all neonantes which contain benzyl alcohol as preservative.
Where can I get more information?
Your pharmacist or health care provider can provide more information about Enoxaparin.
Clinical research and current scenario of the drug.
- Both, in vitro and in vivo studies have demonstrated that Enoxaparin acts at multiple sites in the normal coagulation system to prevent blood clot formation. It has been noticed that small amounts of Enoxaparin in combination with antithrombin III (Enoxaparin cofactor) can significantly inhibit thrombosis by initiating a cascade reaction of inactivation of activated Factor X and inhibition of prothrombin to thrombin conversion.
- At present, Enoxaparin is widely used as prophylaxis treatment in medically ill patients, who are at increased risk for thromboembolism. In comparison to placebo, Enoxaparin therapy has shown significantly increased efficacy in reducing the incidence of deep vein thrombosis and pulmonary embolism
- Randomized, parallel-group, open-label clinical trials have revealed that the rate of deep venous thrombosis was significantly lower in Enoxaparin treated patients (30mg per twelve hours) as compared with the group that received heparin. . Furthermore, it has also been observed that Enoxaparin recipients experienced less adverse events than did heparin recipients.
- Comparative efficacy and safety studies with Apixaban and Enoxaparin for prophylaxis for venous thromboembolism have revealed the fact that shorter course with Enoxaparin was more superior than an extended course of thromboprophylaxis with Apixaban.
References from chemical, biological and toxicological databases.
- Drug Bank (DB01225) Information. http://www.drugbank.ca/drugs/DB01225
- Enoxaparin sodium. Wikipedia Information. https://en.wikipedia.org/wiki/Enoxaparin_sodium
- Enoxaparin – PubChem. https://pubchem.ncbi.nlm.nih.gov/substance/17397770
- MedlinePlus Drug Information. http://www.nlm.nih.gov/medlineplus/druginfo/meds/a601210.html
- ChemSpider Information. http://www.chemspider.com/Chemical-Structure.29786968.html
- Enoxaparin: in the prevention of venous thromboembolism in medical patients. http://www.ncbi.nlm.nih.gov/pubmed/14728009
- Enoxaparin: a review of its use in ST-segment elevation myocardial infarction. http://www.ncbi.nlm.nih.gov/pubmed/18370449
- TIMI 11B. Enoxaparin versus unfractionated heparin for unstable angina or non-Q-wave myocardial infarction: a double-blind, placebo-controlled, parallel-group, multicenter trial. Rationale, study design, and methods. Thrombolysis in Myocardial Infarction (TIMI) 11B Trial Investigators. http://www.ncbi.nlm.nih.gov/pubmed/9628449
- Apixaban versus enoxaparin for thromboprophylaxis in medically ill patients. http://www.ncbi.nlm.nih.gov/pubmed/22077144
- Use of enoxaparin, a low-molecular-weight heparin, and unfractionated heparin for the prevention of deep venous thrombosis after elective hip replacement. A clinical trial comparing efficacy and safety. Enoxaparin Clinical Trial Group. http://www.ncbi.nlm.nih.gov/pubmed/8288662
Read about,
“Mupirocin, drug class, uses, strength, side effects, mechanism of action”
“Rivastigmine, drug class, uses, strength, side effects, mechanism of action“
