Contents
- What is ipratropium bromide?
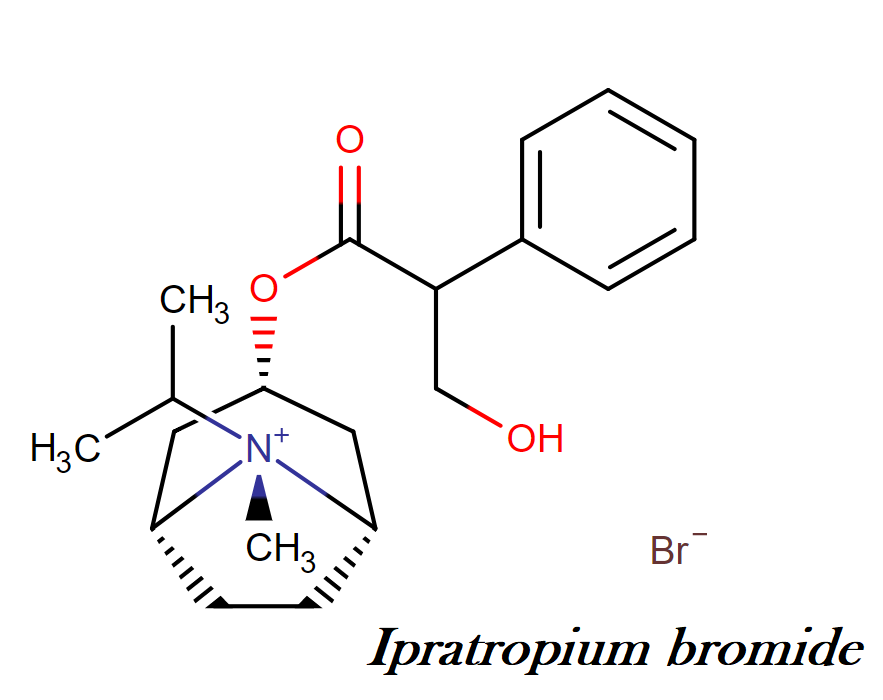
- Ipratropium bromide: chemical name, molecular formula, weight, structure, class
- Ipratropium bromide description
- What is the mechanism of action of ipratropium bromide?
- What are the indications of ipratropium bromide?
- Ipratropium bromide pharmacokinetics
- How long ipratropium bromide stays in your system?
- What are the contraindications of ipratropium bromide?
- What are the warnings associated with use of ipratropium bromide?
- Use of ipratropium bromide in pregnancy and lactation
- What are the adverse reactions caused by ipratropium bromide?
- What if I overdose ipratropium bromide?
- What are precautions needed with ipratropium bromide inhalation?
- Can I use ipratropium bromide inhalation if it is expired?
- What is the dosage and administration of ipratropium bromide?
- How to store the ipratropium nasal?
- What is the information for patients for using ipratropium bromide?
- How to use ipratropium bromide nasal?
- What are the side effects of ipratropium bromide?
- What are some drugs that can interact with ipratropium bromide?
- Can patients with increased heartbeat (tachycardia) take ipratropium bromide?
- Can patients with liver diseases take ipratropium bromide?
- Interactions between drugs
- Diphenhydramine and ipratropium bromide
- Ipratropium bromide and chlorpheniramine
- Promethazine and ipratropium bromide
- Ipratropium bromide and scopolamine
What is ipratropium bromide?
Ipratropium bromide is a synthetic derivative of alkaloid atropine with anticholinergic properties. It is similar to atropine and is a muscarinic antagonist. Ipratropium bromide solution is best for inhalation use in terms of safety and efficacy. It is mainly used for the treatment of acute bronchospasm due to chronic bronchitis and emphysema. Ipratropium bromide is not mainly known to cause any liver enzyme elevation or associated liver injury.
Ipratropium bromide, as an inhalant formulation is known to control a runny nose caused by seasonal allergies or the common cold comes under the brand name of Atrovent. It works by helping to expand the airways leading from the windpipe into the lungs by blocking receptors that normally cause these pipes to constrict or tighten.
Ipratropium bromide: chemical name, molecular formula, weight, structure, class
Chemical name: 3-((3-Hydroxy-2-phenylpropanoyl) oxy)-8-isopropyl-8-methyl-8-azabicyclo [3.2.1] octan-8-ium bromide
Molecular formula: C20H30BrNO3
Molecular weight: 412.368 g/mol
Chemical structure:
Drug class: This compound belongs to the class of organic compounds known as tropane alkaloids. These are organic compounds containing the nitrogenous bicyclic alkaloid parent N-Methyl-8-azabicyclo[3.2.1]octane.
Ipratropium bromide description
The active ingredient in Ipratropium bromide inhalation solution is Ipratropium bromide monohydrate. It is an anticholinergic bronchodilator chemically described as 8-Azoniabicyclo [3.2.1]-octane,-3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-(1-methylethyl)-, bromide, monohydrate ( endo, syn)-,(±)-; a synthetic quaternary ammonium compound, chemically related to atropine.
Ipratropium bromide is a white crystalline substance, freely soluble in water and lower alcohols. It is a quaternary ammonium compound and thus exists in an ionized state in aqueous solutions. It is relatively insoluble in non-polar media.
Ipratropium bromide inhalation solution is administered by oral inhalation with the aid of a nebulizer. Each mL contains Ipratropium bromide 0.02% (anhydrous basis) in a sterile, preservative-free, isotonic saline solution, pH-adjusted to 3.4 (3 to 4) with hydrochloric acid.
What is the mechanism of action of ipratropium bromide?
Ipratropium bromide is an anticholinergic (parasympatholytic) agent, who blocks the muscarinic receptors of acetylcholine, and, based on animal studies, appears to inhibit vagally mediated reflexes by antagonizing the action of acetylcholine which is a transmitter agent released from the vagus nerve.
Anticholinergics prevent the increases in intracellular concentration of cyclic guanosine monophosphate (cGMP), resulting from the interaction of acetylcholine with the muscarinic receptors of bronchial smooth muscle.
Ipratropium bromide by means of inhalation can cause the site specific and a local effect but not produce a systemic effect. Following nebulization of a 2 mg dose, a mean 7% of the dose was absorbed into the systemic circulation either from the surface of the lung or from the gastrointestinal tract.
The half-life of elimination is about 1.6 hours after intravenous administration. Ipratropium bromide is minimally (0 to 9% in vitro) bound to plasma albumin and a 1-acid glycoproteins. It is partially metabolized.
What are the indications of ipratropium bromide?
Ipratropium bromide inhalation solution is prescribed alone or in combination with bronchodilators (especially beta adrenergics) for the treatment of:
- Bronchospasm associated with COPD (chronic obstructive pulmonary disease)
- Chronic Bronchitis
- Emphysema
Ipratropium bromide pharmacokinetics
Ipratropium bromide cause bronchodilation within 15 minutes followed by oral aerosol inhalation and within 15 to 30 minutes after oral inhalation via nebulization. It is minimally absorbed into systemic circulation after oral inhalation. Bronchodilation caused by ipratropium bromide lasts for 3 to 4 hours after aerosol inhalation and for 4 to 5 hours following nebulization.
Ipratropium bromide is a drug that does not penetrate the central nervous system. Also there is no evidence reported about its excretion in human milk or its distribution to the placenta. 0 to 9% of the drug is plasma protein bind. It is metabolized into at least eight metabolites. Ipratropium bromide is a medication that is excreted unchanged in the feces.
How long ipratropium bromide stays in your system?
The half- life of ipratropium bromide is 1.6 hours, which means half of the drug is excreted in 1.6 hours after administration of the drug. Therefore it will completely remove from the system in 3 to 4 hours completely.
What are the contraindications of ipratropium bromide?
Ipratropium bromide is contraindicated in patients who have hypersensitivity to ipratropium bromide or to atropine and its derivatives.
What are the warnings associated with use of ipratropium bromide?
As a single agent, ipratropium bromide inhalation solution is not reported for the relief of bronchospasm in acute chronic obstructive pulmonary disease. As an initial therapy, drugs with faster onset of action are preferable.
In reversing the bronchospasm associated with acute COPD (chronic obstructive pulmonary disease) exacerbation, the combination of ipratropium bromide inhalation solution and beta agonists has not been shown to be more effective than either drug alone. After administration of ipratropium bromide, immediate hypersensitivity reaction can occur. In rare cases, urticaria, angioedema, bronchospasm, rashes and oropharyngeal edema.
Use of ipratropium bromide in pregnancy and lactation
Pregnancy:
Ipratropium bromide comes under pregnancy category B, The women of childbearing age who have taken this medication reported without an increase in malformation or other direct or indirect harmful effects on the fetus. Ipratropium bromide have failed to demonstrate the risk to the fetus, however there are no adequate studies reported for safety in pregnant women. Therefore a pregnant woman should always consult her doctor before taking this medication.
Lactation:
The excretion of the ipratropium bromide in breast milk is not known. Because after inhalation, the systemic absorption of ipratropium is very low, therefore there are very less chances that this medication reaches to the fetus in a significant extent, especially in case of inhalation. However, because many drugs are excreted in human milk, caution should be exercised when ipratropium bromide inhalation solution is administered to a nursing women or a lactating mother.
What are the adverse reactions caused by ipratropium bromide?
The adverse reactions that have been reported in a few patients (< 3%) treated with ipratropium bromide are:
- Tachycardia
- Palpitations
- Urinary retention
- Eyes pain
- Urinary tract infection
- Urticaria
- Worsening of narrow-angle glaucoma
There were few severe and most common adverse effects of ipratropium bromide that leads to discontinuation of ipratropium bromide are:
- Lower respiratory reactions such as:
- Bronchitis
- Dyspnea
- Bronchospasm
Some adverse events occur that caused due to total daily dose of ipratropium bromide (equals to 2000 mcg), which includes:
- Headache
- Mouth dryness
- Aggregation of COPD (chronic obstructive pulmonary disease)
- Allergic-type reactions such as skin rash
- Angioedema of tongue, lips, and face
- Laryngospasm
- Anaphylactic reaction
What if I overdose ipratropium bromide?
There are very less chance that you overdose with Ipratropium bromide because it is not wel absorbed after inhalation at even four-fold of the recommended dose or after oral administration at up to forty-fold recommended dose. The oral lethal dose of Ipratropium bromide ranged between 1001 and 2010 mg/kg which have been tested in mice, and 400 to 1300 mg/kg (tested in dogs).
What are precautions needed with ipratropium bromide inhalation?
- You should always talk to your doctor or pharmacist about your allergies to various drugs such as tiotropium or atropine or other belladona type drugs. You should talk to your pharmacist if other ingredients in this drug can cause allergies or severe reactions.
- You should also discuss your medical history with your doctor or let your doctor know if anybody in your family history had angle closure type glaucoma, difficulty in urinating (due to enlarged prostate).
- One should take this medication cautiously because it cause vision changes and blur your vision, also it can make you feel dizzy. You should not do anything that need alertness or avoid working on heavy machinery. You should also limit alcoholic beverages.
Can I use ipratropium bromide inhalation if it is expired?
One should not be used an expired ipratropium inhalation solution or avoid using the ipratropium bromide after the expiry date passed, which is printed on carton of this medication. Ipratropium bromide can cause serious adverse effects if used after expiration date.
What is the dosage and administration of ipratropium bromide?
1 unit dose vial that contains 500 mcg of Ipratropium bromide is the initial dose as an inhalation solution. It should be administered three to four times a day by oral nebulization with six to eight doses apart.
A unit dose vial contains 500 mcg of Ipratropium bromide anhydrous in 2.5 ml normal saline. If used within one hour, ipratropium bromide inhalation solution can be mixed in the nebulizer with albuterol or metaproterenol. The drug safety and stability of Ipratropium bromide inhalation have not been mentioned, when mixed with other drugs in a nebulizer.
How to store the ipratropium nasal?
Store between 15° C and 30° C (59° F and 86° F). Protect from light. You should store in pouch until time of use.
What is the information for patients for using ipratropium bromide?
One should let their patients know about the side effects that can occur after ipratropium bromide contact directly with their eyes, such side effects include temporary blurring of vision, precipitation or worsening of narrow-angle glaucoma or eyes pain.
Give the necessary instruction to the patients that they should use a nebulizer solution with a mouth piece rather than a face mask may be preferable, to reduce the likelihood of the nebulizer solution reaching the eyes. Patients should be advised that if used within one hour, ipratropium bromide inhalation solution can be mixed with albuterol or metaproterenol.
A patients should be aware about the efficacy and safety of ipratropium bromide with other drugs in a nebulizer, which has not been established. One should know that ipratropium bromide inhalation solution should be used consistently as prescribed throughout the course of therapy.
How to use ipratropium bromide nasal?
You should follow the instructions given in the leaflet that comes inside the box of Ipratropium bromide. So, before using, read the instructions carefully.
- Remove vial from the foil pouch, open the top of one unit-dose vial by twisting it and empty the pouch by squeezing the content into the nebulizer reservoir.
- Now connect the nebulizer reservoir to the mouthpiece or face mask.
- Connect the compressor to the nebulizer.
- Place the mouthpiece in your mouth, after ensuring your sitting that in comfortable position and in upright straight position (Figure 3). You can also put on the face mask and turn on the compressor. You should take proper care while using a mask that is already used. Make sure to avoid leakage around the mask as temporary blurring of vision, precipitation or worsening of narrow angle glaucoma, or eye pain may occur if the solution comes into direct contact with the eyes.
- Take the deep breaths calmly and deeply, until you see there is no more mist is formed in the chamber. It will take around 5 to 15 minutes. Breathe as calmly, deeply and evenly as possible until no more mist is formed in the nebulizer chamber (about 5 to 15 minutes). At this point, the treatment is finished.
- Clean the nebulizer completely to prevent any infection when again in use.
What are the side effects of ipratropium bromide?
- Some common side effects that occur with administration of Ipratropium nasal are bronchitis, COPD exacerbation, dyspnea, headache, throat irritation, cough, dry mouth, gastro-intestinal motility disorders (including constipation, diarrhea and vomiting), nausea, and dizziness.
- Respiratory side effects that commonly occur are bronchitis, upper respiratory tract infection, chest pain, cough, dyspnea, bronchospsasm, increased sputum, pharyngitis, rhinitis, sinusitis, throat infection. Some uncommon or rare respiratory side effects are paradoxical bronchospasm, laryngospasm, pharyngeal edema, dry throat.
- Patients have also suffered from gastrointestinal side effects such as gastrointestinal motility disorder, dyspnea, nausea, and mouth dryness. Some uncommon side effects include constipation, diarrhea, taste perversion, vomiting, stomatitis and rarely mouth edema can occur.
- Urinary tract infection and urinary retention are other common genitourinary side effects caused by Ipratropium bromide.
- Back pain and arthritis are side effects that are noticed as musculoskeletal side effects.
- Some side effects that occur at level of nervous system level are headache, dizziness, and rarely can it cause tremor or nervousness.
- Some uncommon and rare cardiovascular side effects are hypertension, palpitations, supraventricular tachycardia, arterial fibrillation, increased heart rate. Rarely can it cause hypotension and tachycardia.
- Rashes, pruritis and urticaria are some uncommon side effects cause by Ipratropium bromide.
- Hypersensitivity side effects caused by this drug which are rare but can occur are: anaphylactic reactions, angioedema of tongue, lips or face. Urticaria and laryngospasm are its other rare side effects.
- Ocular side effects include: blurred vision, mydriasis, increased intraocular pressure, glaucoma, eye pain, halo vision, conjuctivital hyperemia, and corneal edema.
Can ipratropium bromide be given to person with urinary retention and glaucoma?
Ipratropium is an anticholinergic agent, when given by intranasal route it cause less side effects due to less systemic absorption of quaternary ammonium compounds from gastrointestinal and nasal mucosa. Administration of ipratropium nasal can worsen the urinary retention and glaucoma.
Due to inadvertent contact of the eye with aerosolized or nebulized drug can increase intraocular pressure and precipitation or exacerbation of angle-closure glaucoma may also occur. One with urinary retention and obstruction or open angle glaucoma should use quaternary ammonium compounds therapy cautiously.
Measures should be taken whenever possible to minimize ocular exposure to these drugs, such as keeping eyes closed during oral inhalation or use of a mouthpiece rather than face mask during nebulization. If you see any kind of urinary difficulty or signs or symptoms of angle-closure glaucoma, contact immediately with your health care provider.
Difficulties such as eye pain or discomfort, blurred vision, visual halos, colored images in association with red eyes from conjunctival congestion or corneal edema should be considered immediately and need to get checked immediately.
What are some drugs that can interact with ipratropium bromide?
Ipratropium nasal spray doesn’t really have any major drug interactions, but the inhaled form is more likely to interact with other drugs. Ipratropium bromide is safe and effective bronchodilator when used along with beta adrenergic bronchodilators. Ipratropium bromide has also been used with other pulmonary medications including methylxanthines and corticosteroids, without adverse drug interactions.
Ask your doctor or pharmacist about the inhaled form of ipratropium bromide if you are taking:
- Any opiates, such as Alfenta (alfentanil); codeine; Actiq, Duragesic, Abstral, Fentora, Lazanda, Sublimaze, or Subsys (fentanyl); Oxycontin, Roxicodone, and Oxaydo (oxycodone); or Dilaudid (hydromorphone)
- Drugs for bladder control, like Detrol (tolteridine); Sanctura or Sanctura XR (trospium); or Detrol LA, Oxytrol, Oxytrol for Women, or Gelnique (oxybutynin)
- Transderm Scop (scopolamine)
- Amantadine
- Dolophine or Methadose (methadone)
- Iosat, ThyroSafe, or ThyroShield (potassium iodide)
- K-Tab or Klor-Con (potassium chloride)
- Urocit-K (potassium citrate)
Can patients with increased heartbeat (tachycardia) take ipratropium bromide?
The use of inhaled anticholinergic agents such as ipratropium and tiotropium, can cause supraventricular tachycardia and arterial fibrillation in patients with COPD (chronic obstructive pulmonary disease). When prescribing these agents to patients who are at risk, careful monitoring of cardiac function should be done.
Can patients with liver diseases take ipratropium bromide?
There are no sufficient evident are given, in terms of usage of Ipratropium in the patients with liver disease. However, caution should be practiced while giving Ipratropium bromide medications to the patients with liver diseases.
Interactions between drugs
Ipratropium bromide is effective and safe bronchodilator when used along with beta adrenergic bronchodilators. Ipratropium bromide has also been used with other pulmonary medications including corticosteroids and methylxanthines, without adverse drug interactions.
Here are given some most common drugs that can interact with the ipratropium bromide, it can alter the absorption if such medications or sometimes also responsible for collective anticholinergic effects.
Diphenhydramine and ipratropium bromide
Other anticholinergic effects such as mydriasis, blurred vision heat intolerance, fever, dry mouth, tachycardia, urine retention, constipation, and glaucoma (new onset or excerbation) can occur when you administer ipratropium bromide along with diphenhyramine.
Anticholinergic agents such as antihistamines, antispasmodics, neuroleptics, phenothiazines, skeletal muscle relaxants, tricyclic antidepressants, antiarrhythmics (mainly disopyramide) can also cause severe side effects when administered concurrently with ipratropium nasal.
The interactions are unlikely at regularly recommended dosages, due to poor systemic absorption that occur when ipratropium bromide is administered by intranasal route.
Ipratropium bromide and chlorpheniramine
You should talk to your doctor before using chlorpheniramine together with ipratropium bromide. There are very less chances of side effects due to poorer systemic absorption of ipratropium bromide. It hardly crosses the bloodstream and reaches out to lungs. When it is used with other medications, the risk of side effects can be increased.
There are many side effects which include drowsiness, blurred vision, dry mouth, heat intolerance, flushing, decreased sweating, difficulty urinating, abdominal cramping, constipation, rapid heartbeat, confusion, memory problems, and glaucoma.
The administration of this medication should be used with extreme cautions, and you should always keep your eyes closed when inhaling ipratropium bromide, in order to prevent the drug from entering into the eyes.
Avoid activities requiring mental alertness such as driving or operating hazardous machinery until you know how the medications affect you. It is important to tell your doctor about all other medications you use, including vitamins and herbs. Do not stop using any medications without first talking to your doctor.
Promethazine and ipratropium bromide
When both ipratropium and promethazine are given together, it can produce potential and additive anticholinergic effects such as mydrasis, urinary retention, constipation and glaucoma which can be new or exacerbation of glaucoma.
Other agents that can produce anticholinergic effects are antihistamines, anstispasmodics, neuroleptics, phenothiiazines, skeletal muscle relaxants, tricyclic antidepressants, antiarrhythmics (mainly disopyramide). They can also cause severe side effects when administered concurrently with ipratropium nasal.
Ipratropium bromide and scopolamine
One should generally avoid the concurrent administration of ipratropium and scopolamine, as it can cause additive anticholinergic effects such as mydriasis, blurred vision, heat intolerance, fever, dry mouth, tachycardia, urinary retention, constipation, and glaucoma (onset or exacerbation).
However, the systemic effects are uncommon due to poor administration of quaternary ammonium compounds from gastrointestinal and nasal mucosa. The use of orally inhaled anticholinergic agents can worsen the urinary retention or angle-closure glaucoma.
When inhaled anticholinergic agents are used with each other or with other agents that possess anticholinergic properties. Although systemic effects are uncommon due to the poor absorption of quaternary ammonium compounds from gastrointestinal and nasal mucosa, worsening of urinary retention or angle-closure glaucoma has been reported with the use of orally inhaled anticholinergic agents.
Increased intraocular pressure and precipitation or exacerbation of angle-closure glaucoma may also occur due to inadvertent contact of the eye with aerosolized or nebulized drug.
“Why does Pedialyte help hangovers?”
“What is colloidal silver used to treat? How does colloidal silver work with the body?”