Contents
- What is ketorolac?
- Ketorolac IUPAC name, description, molecular formula, weight, structure, drug class
- Other names for ketorolac
- Ketorolac identification and use
- What are the indications of using ketorolac?
- What is the mechanism of action of ketorolac?
- What are the pharmacokinetics of ketorolac?
- What are the possible side effects of ketorolac?
- Use of ketorolac in pregnancy and breastfeeding
- Ketorolac for intranasal use?
- Subcutaneous ketorolac
- What is the normal recommended dosage of ketorolac?
- The use of ketorolac and platelet aggregation inhibition
- What are dosing considerations necessary for using ketorolac?
- Ketorolac and fluid retention
- What are the precautions of using ketorolac?
- Can I take ketorolac with beta-blockers?
- Can I take ketorolac with busulfan?
- Can I take ketorolac in combination with other NSAIDs?
- Contraindications for using ketorolac?
- Can I take ketorolac with alcohol?
What is ketorolac?
Ketorolac is one of the most commonly used and a non-narcotic analgesic which belongs to a class of drugs known as non-steroidal anti-inflammatory drugs.
It is a prescription drug which is present in two dosage forms that are in the parenteral and oral dosage form.
It is mainly used in the management of short-term treatment of pain. Ketorolac along with analgesic property posses anti-inflammatory and antipyretic property.
Ketorolac IUPAC name, description, molecular formula, weight, structure, drug class
Ketorolac belongs to a group of pyrrolo-pyrrole NSAIDs. It has shown a structural and pharmacological resemblance to indomethacin but distinct from other pyrrole acetic acid derivatives, this drug is a cyclic propionic derivative.
This is commercially available as ketorolac tromethamine. This salt of ketorolac is a racemic mixture which contains (-)S and (+)R enantiomers.
S-form of ketorolac is responsible for the analgesic activity. This basically exists in three different crystal forms. It has been observed that all these crystal forms have shown equal solubility in water.
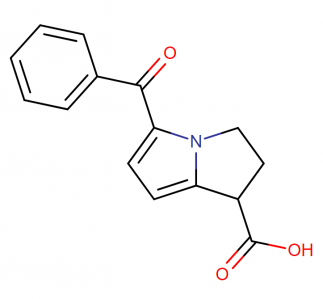
IUPAC name: 5-benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid
Molecular formula: C15H13NO3
Molecular weight: 255.26 g/mol
Molecular structure:
Drug class: This compound belongs to the class of organic compounds known as aryl-phenylketones. These are aromatic compounds containing a ketone substituted by one aryl group, and a phenyl group.
Other names for ketorolac
Toradol; Tora-dol; Dolac; Lixidol, Tarasyn; Toratex; Toradyl; Acular; Aculare; Droal; Tonum; 5-Benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid; 5-Benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid; Ketorolac
Ketorolac identification and use
The tablets of ketorolac are round in shape and white in color with a red print on them. These tablets are manufactured containing the active ingredient ketorolac and excipients which include lactose, microcrystalline cellulose, and magnesium stearate.
Ketorolac tromethamine is soluble in water and methanol; slightly soluble in ethanol and tetrahydrofuran; insoluble in acetonitrile, acetone, hexane, ethyl acetate, and toluene.
The parenteral form of ketorolac is manufactured as 1% and 3% sterile solution which is intended to be administered via IM route only. Along with the active, additional substances added are water for injection, ethanol, sodium hydroxide and hydrochloric acid.
What are the indications of using ketorolac?
According to FDA, ketorolac is indicated for short-term (not exceeding 5 days in adults) treatment of moderate to severe pain in which analgesia is required up to opioid level.
FDA approved indications are:
- Cystoid Macular Edema
- Inflammatory Reaction
- Migraine, Acute
- Ocular Itching
- Ocular Pain
- Pain, Acute
- Pseudophakic Cystoid Macular Edema
- Acute Pericarditis
- Chronic aphakic cystoid macular edema
- Intraoperative miosis
- Moderate Acute pain
- Postoperative ocular pain
- Severe Acute pain
What is the mechanism of action of ketorolac?
This drug exerts its action by competitively blocking cyclooxygenase isoenzymes including both COX-1 and COX-2 by preventing the binding with arachidonate.
This produces the analgesic, anti-inflammatory and antipyretic action of ketorolac. The COX isoenzymes act as a catalyst in the conversion of arachidonic acid to prostaglandins G2.
It has been observed that COX-1 is constitutively present in almost all the tissues whereas COX-2 presence is only limited till reproductive organs, brain, kidneys, bones and certain neoplasms (such as prostate and colon cancer).
COX-1 is responsible for the synthesis of prostaglandins in response to certain different stimulations and helps in maintaining various normal functions in the body. COX-2 is generated as a result of certain inflammatory mediators.
Ketorolac basically shows its anti-inflammatory action by reducing the synthesis of PG. this reduction is provided by the inhibition of both COX enzymes but majorly COX-2. COX-1, on the other hand, is present at some sites of inflammation for example joints.
The analgesic activity of ketorolac is only produced where inflammation has increased the sensitivity of pain receptors. Prostaglandins, particularly PGE and PGF have shown to increase the sensitivity of these receptors.
In short, ketorolac indirectly acts as an analgesic agent but have yet not demonstrated a direct effect on the pain threshold.
Moreover, ketorolac has also shown to reduce the body temperature to a normal set point. This anti-pyretic effect is produced by repressing the prostaglandin E2 synthesis in the hypothalamus or near the hypothalamus.
What are the pharmacokinetics of ketorolac?
As ketorolac is manufactured in different dosage forms, each route has displayed a different set of pharmacokinetic parameters. It is administered via oral, parenteral, nasal or ophthalmic routes.
Ketorolac which is administered via oral and parenteral have shown a similar pharmacokinetic profile and that which is administered intramuscularly and intranasally have shown a similar pharmacokinetic profile. About 99% of this drug is bind to plasma albumin.
It shows transportation across the placental barrier and has been detected in breast milk in small quantities.
It undergoes hepatic metabolism in the reaction of hydroxylation and is converted into the metabolite p-hydroxyketorolac.
This is a weak metabolite and has a potency even less than 1% that of the parent drug. Moreover, conjugation with glucuronic acid also takes place. Approximately 91% of ketorolac is eliminated from the body and only a small portion is excreted in feces.
When ketorolac is administered through the oral route, it has demonstrated a rapid and complete bioavailability of 100%. Maximum plasma concentrations can be obtained within an hour.
After the ingestion of the drug, analgesia occurs within 30 – 60 minutes and duration of action is seen within 6 – 8 hours of ingestion.
Mean elimination half-life of ketorolac is reported to be 5.3 hours and a range of 2.5 to 9 hours. Elderly patients have shown a longer half-life after ketorolac was administered orally.
When administered parenterally, a complete bioavailability is seen and analgesia is observed within 30 minutes. Peak analgesic effect is seen in 1 to 2 hours. Following IV injection, duration of action for ketorolac is between 4 – 6 hours.
The analgesic effect of ketorolac is dose-dependent i.e. when the dose increases, duration of analgesia increases.
When ketorolac is administered intramuscularly, the same pharmacokinetic profile is seen except in individuals who are 65 years or older and have a prolonged half-life of approximately 7 hours.
When ketorolac is administered via the nasal pathway, disbursement of the drug occurs majorly in the nasal cavity and pharynx (about 80%), in stomach and esophagus (about 15% – 20%) and a very small amount in lungs.
Other parameters which included Tmax, Cmax, half-life, and AUC were same as that of the intramuscular route.
What are the possible side effects of ketorolac?
Ketorolac is potent NSAID which although is used in the prophylaxis and treatment of certain medical conditions but at the same time is responsible to adversely affect the patient in a number of ways.
The intensity of these side effects directly depends on the dose administered and vary from mild degree to a severe life-threatening situation.
Those unwanted side effects which are of mild intensity and easily reversible on the discontinuation of the medicine are as below:
- Abdominal pain, nausea, dyspepsia, flatulence, diarrhea, and vomiting
- A headache, ocular pain, ocular irritation, xeropthalmia and lacrimation
- Rhinalgia, rhinitis, throat irritation and cough
- Injection site reaction, purpura, rash, and diaphoresis
- Tremor, paresthesia, hyperkinesis, and dizziness
- Epistaxis, tinnitus, pallor, and syncope
Those side effects which are of moderate intensity are as below:
- Corneal edema, ocular inflammation, conjunctival hyperemia and iritis
- Jaundice, cholestasis, hepatitis and raised liver enzymes
- Anemia, bleeding, leucopenia, thrombocytopenia and platelet dysfunction.
- Dysphagia, gastritis, esophagitis, and hematuria
- Euphoria, depression, psychosis, blurred vision, and hallucinations
- Hyponatremia, hypotension, and edema
- Chest pain, sinus tachycardia, palpitations and hyphemia
Some of the life-threatening side effects which may occur on using ketorolac are listed as below. On the occurrence of any of these effects, the patient should be immediately presented to the emergency:
- GI perforation, GI bleeding and peptic ulcer
- Pancreatitis and hepatic failure
- Permanent corneal erosion
- Stroke, myocardial infarction, angioedema, pulmonary edema and heart failure
- Hyperkalemia, oliguria, nephrotic syndrome and renal failure
- Vasculitis and thromboembolism
- Aseptic meningitis
- Anaphylactoid reactions and anaphylactic shock
- Steven Jhonson syndrome and toxic epidermal necrolysis
Use of ketorolac in pregnancy and breastfeeding
Pregnancy:
Ketorolac is not the drug of choice to be administered to pregnant females especially during late pregnancy.
This drug is classified as FDA pregnancy risk category C. this means that ketorolac should only be prescribed when the possible advantages are more as compared to the risks for the fetus and mother both.
The effects of NSAIDs have been studied on that fetal cardiovascular and have caused premature closure of fetal ductus arteriosus.
The nasal and systemic administration of ketorolac in late pregnancy at about 30 weeks is marked as FDA pregnancy risk category D.
When ketorolac is used through ophthalmic route, it shows a little absorption and thus is marked as FDA pregnancy risk category C. on a general; all dosage forms of ketorolac have not yet demonstrated safety and efficacy profiles so it should only be used with caution.
According to a study conducted and the result that was obtained from that study stated that excessive use of ketorolac in the first trimester of pregnancy increases the chances of miscarriages.
Another study showed babies with chronic maternal use of ketorolac were born with pulmonary hypertension, ductus arteriosis closure, respiratory distress syndrome, and asphyxia.
Use of the drug in late pregnancy had been associated with fatal complications in the neonates such as intracranial hemorrhage, necrotizing enterocolitis.
Ketorolac can be beneficially used to delay the preterm delivery of the baby but due to more dominating adverse effects were seen on the fetal blood circulation or on the uterine contractions, it is better to avoid this medicine.
Breastfeeding:
Prostaglandin inhibiting drugs have shown a number of adverse when used by nursing mothers, so caution is needed before the administration of the drug.
As ketorolac has been detected in breast milk in variable amounts so it is recommended to closely examine the breastfed infant for the occurrence of any unwanted effect.
Even though according to American Academy of Pediatrics, this medicine has been marked compatible with the breast milk but in case the breastfed infant experiences any side effect which is linked with the maternal use of the drug, it should be immediately reported by the healthcare providers to the Food and Drug Administration (FDA).
Ketorolac for intranasal use?
Intransal (IN) ketorolac is approved for short-term management of moderate to severe pain. The
IN formulation is generally well tolerated.
The standard adult dose is one 15.75-mg spray in each nostril for a total dose of 31.5 mg. Maintenance doses are given every 6 to 8 hours with a maximum daily dose of 126 mg.
In those over the age of 65, those less than 50 kg, or those with underlying renal impairment, the dose should be halved to one 15.75-mg spray every 6 to 8 hours to a maximum daily dose of 63 mg.
Adverse events occur in less than 2% of patients, with the most common being local nasal symptoms, including rhinalgia and rhinitis.
Subcutaneous ketorolac
The subcutaneous route is currently not approved by the US Food and Drug Administration
(FDA).
Optimal doses are unknown; the pharmacokinetics and pharmacodynamics are not completely defined, and the safety and efficacy are yet to be established.
However, subcutaneous administration of ketorolac maybe useful in patients with no IV access or low muscle mass.
It may also have some benefit in pain control due to its lower rate of absorption. Dosing is usually by slow continuous infusion of 30 to 120 mg over 24 hours.
What is the normal recommended dosage of ketorolac?
Oral administration of ketorolac should only be advised as a continuation of parenteral therapy. However, it is necessary to not to use ketorolac by any route in any condition for more than 5 continuous days.
Via oral route:
For adults who are less than 65 years of age, the normally recommended dosage guidelines state that the starting dose should be 20 mg which is then followed by 10 mg given 4 times a day with a gap of at least 4 – 5 hours. It should be kept in mind to not to exceed the intake of ketorolac greater than 40 mg in a single day.
In adults who are older than 65 years of age and weigh less than 50 kg, (or even are renally impaired) the dose of ketorolac administered should not be greater than 40 mg per day.
The medicine can be given in divided doses of 10 mg with at least 4 – 5 hours in between 2 consecutive doses.
Children who are from 2 years to 16 years old and have a weight of less than 50 kg, the appropriate dose recommended is 1 mg/kg as a single dose. Multiple dosing has yet not been evaluated for safe use in the children.
Via intramuscular and intravenous route:
For adults who are younger than 65 years, the normal recommended single dose is 60 mg/ day. When the patient has to be given multiple dosing it is advised not to exceed 120 mg/day. Divided dosing can be done as 30 mg given every 6 hours.
In adults who are older than 65 years of age and weigh less than 50 kg, (or even are renally impaired) the dose of ketorolac administered should not be greater than 60 mg per day. The medicine can be given in divided doses of 15 mg with at least 6 hours in between 2 consecutive doses.
In children who are in between 2 – 16 years old, the normal recommended single dosing is 0.4 to 1 mg/kg. It should not exceed 30 mg as a single dose. When given as divided doses, 0.5 mg/kg can be given every 6 hourly.
Via intranasal route:
In adults who are below 65 years of age, it is recommended to spray once in each nostril. Sprys should not exceed more than times per day with a gap of about 6- 8 hours.
In adults who are older than 65 years of age and weigh less than 50 kg, (or even are renally impaired) it is recommended to spray once in only one nostril. Sprays should not exceed more than times per day with a gap of about 6- 8 hours.
The use of ketorolac and platelet aggregation inhibition
The use of ketorolac is considered by the manufacturer to be contraindicated in patients with suspected or confirmed cerebrovascular bleeding, hemorrhagic diathesis, incomplete hemostasis, or a high risk of bleeding.
Ketorolac is a potent nonsteroidal anti-inflammatory drug (NSAID). NSAIDs reversibly inhibit platelet adhesion and aggregation and may prolong bleeding time in healthy individuals.
With the exception of aspirin, the platelet effects seen with most NSAIDs at usual recommended dosages are generally slight and of relatively short duration but may be more pronounced in patients with underlying hemostatic abnormalities.
Thrombocytopenia has also been reported rarely during NSAID use. Therapy with NSAIDs, including ketorolac, should be administered cautiously in patients with significant active bleeding.
What are dosing considerations necessary for using ketorolac?
The patient population who are experiencing any kind of hepatic illness should use ketorolac with caution. No modification in dosage guidelines has yet been present to be given in hepatic impairment.
In patients who are suffering from a serious renal impairment or those who are at risk of renal failure as a result of volume, depletion should be given modified doses of ketorolac.
Before starting the therapy with ketorolac in such patients, it is advised to correct hypovolemia first. Lowest effective doses of ketorolac should be used in such patients and they should be closely monitored for the occurrence of any adverse effects.
Ketorolac and fluid retention
Fluid retention and edema have been reported in association with the use of nonsteroidal anti-inflammatory drugs (NSAIDs).
Therapy with NSAIDs should be administered cautiously in patients with preexisting fluid retention, hypertension, or a history of heart failure.
Blood pressure and cardiovascular status should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
What are the precautions of using ketorolac?
Certain precautions must be considered while starting the therapy with ketorolac:
- Limited duration of use
The administration of ketorolac in any dosage form should not exceed greater than 5 days. Ketorolac’s systemic administration is indicated in a short-term treatment of moderate to severe pain in which analgesia is necessary at an opioid level.
Oral tablets are only prescribed as a continuation therapy after IM or IV administration of ketorolac. Use of this medicine is limited as the prolonged use is associated with the development of serious adverse effects.
- In geriatric
Elderly patients who are above 65 years of age are at a higher risk of experiencing the adverse effects of using ketorolac. Caution is required when prescribing this medicine to the elderly patients.
Most commonly occurring side effects which occur include GI bleeding, GI perforation, ulceration, fluid retention and decreased renal perfusion.
According to the data published by the Beers Criteria, all those patient who either had a previous medical history of ulcer or not, both are equally at an risk for suffering from GI bleeding and ulceration or even from acute kidney disease.
Ketorolac should only be prescribed when other analgesic or anti-inflammatory agents are not effective or have failed or can clinically interact either with the disease or any co-administered drug.
Ketorolac has also been reported to worsen up the condition of the patient if he is suffering from hypertension, renal disease or heart failure.
- Hepatic diseases
When ketorolac is given to patients systematically to patients having a hepatic impairment, great care is required. During the course of therapy, severe hepatic reactions are reported to occur.
Life-threatening complications may occur when patients with hepatic impairment are given ketorolac.
Regular monitoring of hepatic enzymes is necessary before, during and after the treatment and if the enzymes are increased from the normal range the drug should be immediately discontinued.
In case of hypoalbuminemia, the number of active drug increases in the serum thus increasing the chances of ketorolac associated toxicity.
In addition to this, when the prostaglandin synthesis is inhibited, the renal blood flow is reduced.
Due to the combined function of prostaglandins, patients with hypoalbuminemia must be examined throughout the treatment period. Such complications do not occur on using ketorolac through ophthalmic route.
Can I take ketorolac with beta-blockers?
When ketorolac is administered in combination with any of the beta blockers specifically cardioselective beta blocker for the management of hypertension, a moderate degree of drug interaction takes place.
The patient is closely monitored for any possible signs and symptoms of renal insufficiency or changes in blood pressure.
Dose adjustments for anti-hypertensive agents are necessary when this combination is to be given to the patient.
NSAIDs cause a decrease in the prostaglandin synthesis which leads decreased renal blood flow, raised blood pressure which is mostly accompanied by edema and weight gain.
Those patients who are also using diuretic agents are more prone to suffer from stroke and coronary heart disease when ketorolac is administered.
Can I take ketorolac with busulfan?
Drug interaction of a major intensity occurs when busulfan is concurrently administered with ketorolac.
This is due to the fact that ketorolac causes the inhibition of platelet aggregation by blocking the COX-1 enzyme present on the platelets. This reduces the amount of platelet thromboxane A2 ultimately prolonging the bleeding time.
Although this inhibition of platelet aggregation is reversible on the discontinuation of the drug when given in combination with busulfan, the bleeding effect is more enhanced.
Busulfan has thrombocytopenic effects which worsen up the condition when both the drugs are given together to the patient.
Can I take ketorolac in combination with other NSAIDs?
Two or more NSAIDs should never be given concomitantly. This will produce a severe degree of drug interaction which will ultimately adversely harm the user.
This combination will produce additive effects and produce gastrointestinal effects such as GI bleeding, GI perforation, and ulceration. This is more prominent when ketorolac is used in combination with COX-2 selective inhibitors.
Can you take Ketorolac and Tylenol together?
Contraindications for using ketorolac?
Use of ketorolac is contraindicated in the following conditions;
- Bleeding, surgery:
Ketorolac has the potential to cause bleeding and worsen up hematological complications. Using ketorolac systemically for the purpose of analgesia before any surgical procedure, and the ophthalmic solution before an ocular surgery is contraindicated.
When ketorolac is administered before operations, a hematoma is reported to occur.
Moreover in those patients who are either genetically or are suffering from any bleedings disorders should not use ketorolac as it is likely to prolong the bleeding time and further worsen up the bleeding problems.
Ketorolac has also been reported to cause increased bleeding during the ocular procedure from ocular tissues.
- Gastric diseases
In gastritis or peptic ulcer diseases, when ketorolac is administered systematically especially via the oral route, perforation and bleeding have been documented.
Ketorolac is thus contraindicated in all those who either are concurrently having or had a past medical history of ulcer.
Due to the inhibitory effect of ketorolac on prostaglandins, it has proven to be fatal in older patients. All those patients who are already being given any kind of therapy should be closely monitored for any adverse effects.
Can I take ketorolac with alcohol?
Ask your doctor before using ketorolac together with ethanol. Do not drink alcohol while taking ketorolac. Alcohol can increase your risk of stomach bleeding caused by ketorolac. Call your doctor at once if you have symptoms of bleeding in your stomach or intestines.
This includes black, bloody, or tarry stools, or coughing up blood or vomit that looks like coffee grounds.
It is important to tell your doctor about all other medications you use, including vitamins and herbs. Do not stop using any medications without first talking to your doctor.
Can you take Ketorolac and Hydrocodone together ?
“Can you take Ketorolac and Oxycodone together?
“Iophen NR -Cold medication and cough medicine“