Contents
- What is Mobic?
- Mobic molecular formula, weight, molecular structure and drug class
- What is the pharmacology of Mobic?
- What are the indications of Mobic?
- What are the dosage forms and strength of Mobic?
- What are the contraindications of the Mobic?
- What are some side effects of the Mobic?
- How long Mobic stays in the system?
- Can Mobic cause cardiovascular thrombotic events?
- Can Mobic cause heart failure or edema?
- Can Mobic cause gastrointestinal bleeding ulceration, and perforation?
- What are the risk factors for gastrointestinal bleeding or ulceration in patients using Mobic?
- Can Mobic cause hepatotoxicity?
- Can Mobic cause hypertension?
- Can Mobic cause renal toxicity or hyperkalemia?
- Can Mobic be taken with Alcohol?
- Can Mobic cause platelet aggregation inhibition?
- Can Mobic be taken by anemic patient?
- Can Mobic cause skin reactions?
- Can Mobic be used in pregnancy?
- Can Mobic be taken with duloxetine?
- Can Mobic be taken with lisinopril?
- Can Mobic be taken with sertraline (Zoloft)?
- Can Mobic be taken with prednisone?
What is Mobic?
Mobic is an anti-inflammatory drug that contains meloxicam. It is a COX (cyclooxygenase) inhibitor. Meloxicam is an oxicam derivative and a non-steroidal anti-inflammatory drug (NSAID) with anti-inflammatory, antipyretic and analgesic activities.
Meloxicam is a long-acting nonsteroidal anti-inflammatory drug (NSAID) available by prescription only and used in therapy of chronic arthritis.
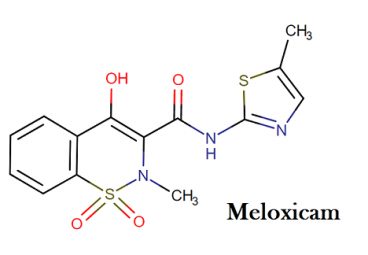
Mobic molecular formula, weight, molecular structure and drug class
Molecular formula of meloxicam: C14H13N3O4S2
Molecular weight of meloxicam: 351.395 g/mol
Drug class of meloxicam: This compound belongs to the class of organic compounds known as benzothiazines. These are organic compounds containing a benzene fused to a thiazine ring (a six-membered ring with four carbon atoms, one nitrogen atom and one sulfur atom).
Molecular structure of meloxicam:
What is the pharmacology of Mobic?
Mobic contains meloxicam which is an oxicam derivative and nonsteroidal anti-inflammatory drug that possess analgesic and antipyretic properties. The inflammation of joints is caused by the substance called prostaglandins.
The synthesis of prostaglandins is inhibited by the meloxicam by inhibiting the cyclooxygenase 1 and 2 responsible for the synthesis of prostaglandins. The inflammation is reduced by the inhibiting the synthesis of the prostaglandins.
What are the indications of Mobic?
Mobic is indicated for the treatment of:
- Osteoarthritis
- Rheumatoid arthritis
- Juvenile rheumatoid arthritis
What are the dosage forms and strength of Mobic?
Mobic (meloxicam) Oral Suspension: yellowish green tinged viscous suspension containing 7.5 mg meloxicam per 5 ml.
What are the contraindications of the Mobic?
Mobic is contraindicated in the following patients:
The patient who have hypersensitivity to meloxicam such as anaphylactic reactions or serious skin reactions. The patients who have a history of urticaria, asthma, or other allergic-type reactions after taking the medications like aspirin or any other NSAIDs.
In patients who have a history of anaphylactic reactions after taking NSAIDs, sometimes fatal, anaphylactic reactions can occur. Mobic is also contraindicated in the setting of coronary artery bypass graft.
What are some side effects of the Mobic?
You should seek for emergency medical help if you have signs of an allergic reaction to Mobic. The symptoms and signs of allergic reactions are sneezing, runny or stuffy nose; wheezing or trouble breathing; hives; swelling of your face, lips, tongue, or throat.
If you have signs of heart attack or stroke, such as chest pain which spreads to your jaw or shoulder, sudden numbness or weakness on one side of the body, slurred speech, feeling short of breath.
You should ask for medical help if you see side effects like:
- Signs of skin rash (even if it is mild)
- Shortness of breath
- Swelling or rapid weight gain
- Signs of stomach bleeding – bloody or tarry stools, coughing up blood or vomit that looks like coffee grounds
- Liver problems – nausea, upper stomach pain, itching, tired feeling, flu-like symptoms, loss of appetite, dark urine, clay-colored stools, jaundice (yellowing of the skin or eyes)
- Kidney problems – little or no urinating, painful or difficult urination, swelling in your feet or ankles, feeling tired or short of breath
- Low red blood cells (anemia) – pale skin, feeling light-headed or short of breath, rapid heart rate, trouble concentrating
- Severe skin reaction – fever, sore throat, swelling of your face or tongue, burning in your eyes, skin pain followed by a red or purple skin rash that spreads (especially in the face or upper body) and causes blistering and peeling.
Common Mobic side effects may include:
- Upset stomach
- Nausea
- Vomiting
- Heartburn
- Diarrhea
- Constipation and Gas
- Dizziness
- Cold and flu symptoms
How long Mobic stays in the system?
The half-life of meloxicam is 15 to 20 hours so it will take about 80 hours to be out of body.
Can Mobic cause cardiovascular thrombotic events?
There is an increased risk of serious cardiovascular thrombotic events including myocardial infarction, and stroke has been reported as increased with three-year longer duration and usage of COX-2 selective and nonselective NSAIDs.
- The cardiovascular thrombotic events increased over baseline with the use of Mobic or other NSAIDs is reported as similar in patients with or without existing cardiovascular disease or even in those who have the risk of cardiovascular events.
- However, patients with known cardiovascular disease or risk factors had a higher absolute incidence of excess serious cardiovascular thrombotic events, due to their increased baseline rate.
- Sometimes in the first week of treatment, the increased risk of serious cardiovascular thrombotic events began as per some observational studies. In higher doses, the increase in cardiovascular thrombotic risk has been observed most consistently.
- One should use the lowest effective dose of Mobic or other NSAIDs, for the minimum duration of time possible in order to minimize the potential risk for adverse cardiovascular events.
- Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous cardiovascular symptoms.
- The patient should keep an eye on the new or recurring symptoms of cardiovascular events and should immediately inform the doctor so that proper and on time treatment can be done.
- The risk of cardiovascular events is same for all nonselective anti-inflammatory inhibitors is not yet clear as per available data.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious cardiovascular thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as meloxicam, increases the risk of serious gastrointestinal (GI) events.
The increased risk of thrombotic events such as myocardial infarction and stroke has been reported with the use of NSAIDs such as Mobic. These events can prove fatal. If the NSAIDs are taken for a longer duration the risk of thrombotic events can get increased. Patients who have a history of the cardiovascular or cerebrovascular disease should use medications such as NSAIDs with extreme caution.
Patients should be treated with the lowest effective dosage for the shortest duration necessary. The patients who require cardio protection should administer appropriate antiplatelet therapy. If patients experience symptoms such as chest pain, shortness of breath, weakness, and slurring of speech (the symptoms of cardiovascular thrombotic events), they should immediately seek medical attention and should cease the NSAIDs therapy.
Can Mobic cause heart failure or edema?
There are two-fold increases in heart failure and hospitalizations for the same reasons have been reported in patients treated with NSAIDs (nonsteroidal anti-inflammatory drugs) as per controlled trials as compared to the patients treated with placebo.
- NSAIDs also increase the risk of myocardial infarction (MI), hospitalizations for heart failure and death.
- Fluid retention and edema have also been reported with the prolonged use of Mobic and other NSAIDs.
- The cardiovascular effects of the medications such as (diuretics, angiotensin-converting enzyme inhibitors, or angiotensin receptors blockers) have been altered by the concomitant use of Mobic (meloxicam).
- In patients with severe heart failure, the Mobic should not be used until and unless the benefit outweighs the risks.
- The patients for signs of heart failure or worsening cardiovascular events should be monitored if Mobic is used in such patients.
- In some topical formulations of NSAIDs, the fluid retention and edema have also been observed. Therefore precaution is necessary if using Mobic in patients with fluid retention or heart failure.
Can Mobic cause gastrointestinal bleeding ulceration, and perforation?
Yes, many adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine can be caused by NSAIDs such as Mobic. One can suffer from these adverse events any time that can occur with or without prior symptoms if they are using NSAIDs like Mobic.
Sometimes these adverse events can be symptomatic but it occurs in one in five patients. In patients (approx. 1%) who used NSAIDs for 3 to 6 months, upper gastrointestinal ulcers, gross bleeding, or perforation can be seen. Sometimes in 2 to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
What are the risk factors for gastrointestinal bleeding or ulceration in patients using Mobic?
- There are 10 fold higher risks of developing gastrointestinal bleed in patient who have a history of peptic ulcer and using NSAIDs like Mobic as compared to the patients without these risk factors.
The risk of severe and fatal gastrointestinal events in patients using NSAIDs is high in case of:
- Longer duration of NSAIDs therapy (including Mobic)
- Concomitant use of oral corticosteroids or aspirin
- Age and poor health status
- Selective serotonin reuptake inhibitors (SSRIs)
- Smoking and use of alcohol
- Patients with advanced liver disease or coagulopathy
Fatal gastrointestinal reports have been reported in elderly or debilitated patients. You can use various strategies to minimize the risk of gastrointestinal bleeding in patients treated with NSAIDs.
- You can use the lowest effective dosage for the shortest possible duration.
- You should avoid administration of more than one NSAID at a time.
- You should also avoid using NSAIDs in patients who are higher risk unless benefits outweigh the risks
- One should consider alternative therapies for patients who have active gastrointestinal bleeding because giving NSAIDs to such patients can exacerbate the condition and one can suffer from serious or fatal gastrointestinal event.
- You should always remain alert for signs and symptoms of gastrointestinal ulceration and bleeding during NSAID therapy.
- If a serious gastrointestinal adverse event is suspected, promptly initiate evaluation and treatment. You should consider discontinuing Mobic until an adverse event is ruled out.
In the setting of concomitant use of low-dose aspirin for cardiac prophylaxis, monitor patients more closely for evidence of GI bleeding.
Can Mobic cause hepatotoxicity?
In approximately 1% patients who were treated with NSAIDs, the elevations of ALT (alanine aminotransferase) and AST (Aspartate aminotransferase) is reported. These are the enzymes which are used to check the liver damage or liver disease.
And elevations in these enzymes can occur in up to 15% of patients using NSAIDs like Mobic. There are very less or rare or even fatal cases of severe hepatic injury such as liver necrosis and hepatic failure have been reported in patients using NSAIDs.
One should inform the patients about symptoms of nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness and flu-like symptoms; these are the symptoms of hepatotoxicity. Discontinue Mobic immediately if clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations like eosinophilia or rash occur. One should perform a clinical evaluation of the patient.
Mobic should not be given to the patients with liver disease as it can cause hepatotoxicity. Because:
- There are many abnormalities such as elevations in serum transaminases and alkaline phosphatase can be enhanced or remain unchanged with continuing therapy with Mobic or other NSAIDs. These studies have been reported in approximately 15% of the patients treated with nonsteroidal anti-inflammatory drugs.
- 3 times increase in a liver enzyme, from the upper limit of normal, has also been reported with the use of NSAIDs.
- Other notable rare cases of severe toxicity reported by NSAIDs (such as Mobic) use are liver necrosis, hepatic failure, jaundice and fatal fulminant hepatitis.
One should use nonsteroidal anti-inflammatory drugs with extreme caution in patients with the preexisting liver disease. If prolonged therapy of NSAIDs has to continue, the periodic monitoring of liver function is advised because the medications like NSAIDs are highly protein bound and extensively metabolized by the liver. Such activities may be altered or changed in patients with hepatic impairment. In few cases, the dose reduction may also be needed.
Can Mobic cause hypertension?
New onset of hypertension or worsening of existing hypertension can occur due to Mobic or other NSAIDs. The increased incidence of cardiovascular events was reported in patients using NSAIDs.
The patients may see the impaired response from the therapies such as ACE (angiotensin-converting enzyme) inhibitors, loop diuretics, or thiazide diuretics when taking with NSAIDs. One should monitor the blood pressure during the initiation of NSAIDs and throughout the course of the therapy.
The use of Mobic and other nonsteroidal anti-inflammatory drugs (NSAIDs) has also been associated with fluid retention and edema. In patients with existing fluid retention conditions, hypertension, or a history of heart failure, the use of NSAIDs should be done with caution. The other parameter that needs close monitoring is blood pressure and cardiovascular status of the patient during the initiation or throughout the course of therapy.
Can Mobic cause renal toxicity or hyperkalemia?
Renal papillary necrosis, renal insufficiency, acute renal failure and another renal injury can result from the prolonged use of Mobic or other nonsteroidal anti-inflammatory drugs (NSAIDs). In patients where the renal prostaglandins have a compensatory role in the maintenance of renal perfusion, NSAIDs seems to cause renal toxicity.
A dose-dependent reduction in renal prostaglandins formation has been reported with the use of NSAIDs. Also, renal blood flow also reported getting decreased, which can precipitate overt renal decompensation due to prolonged NSAIDs usage.
The patients with impaired renal function, dehydration, hypovolemia, heart failure, liver dysfunction, those taking diuretics and ACE (angiotensin-converting enzyme) inhibitors, and the elderly are at greater risk.
The progression of renal dysfunction in patients with the already existing renal disease can be hastened due to renal effects of Mobic. One should monitor the patient for worsening condition of renal function because some metabolites of Mobic are excreted by the kidneys.
Regarding the use of Mobic in patients with the advanced renal disease, there is no information is available from controlled clinical studies. Avoid the use of Mobic in patients with advanced renal disease unless the benefits are expected to outweigh the risk of worsening renal function. If Mobic is used in patients with the advanced renal disease, monitor patients for signs of worsening renal function.
The prolonged use of NSAIDs is associated with increased in serum potassium concentration including hyperkalemia even in patients with or without impaired renal function. These effects have been attributed to the hyporeninemic-hypoaldosteronism state in patients with normal renal function.
Can Mobic be taken with Alcohol?
One should generally avoid the concurrent administration of Mobic and alcohol. It may cause gastrointestinal blood loss if aspirin or nonsteroidal anti-inflammatory drugs are taken with alcohol.
This can occur due to a loss of the integrity of gastrointestinal lining and local effect of prostaglandin synthesis or maybe by the combined effects of both mechanisms. Patients should be counseled on this potential interaction and advised to refrain from alcohol consumption while taking aspirin or NSAIDs.
Can Mobic cause platelet aggregation inhibition?
The platelet adhesion and aggregation can be inhibited and the bleeding time can also be prolonged in patients with the use of nonsteroidal anti-inflammatory drugs. With the exception of aspirin, the platelet effects seen with most NSAIDs at usual recommended dosages are generally slight and of relatively short duration but may be more pronounced in patients with underlying hemostatic abnormalities. During the use of the nonsteroidal anti-inflammatory drugs, thrombocytopenia has been reported very rarely.
In patients with symptoms like significant active bleeding or a hemorrhagic diathesis, including hemostatic or coagulation defects associated with hemophilia, vitamin K deficiency, hypoprothrombinemia, thrombocytopenia, thrombocytopathy, or severe hepatic impairment, NSAIDs should be used with extra caution. NSAIDs such as Mobic do not appear to affect platelet function or bleeding time at recommended doses as it selectively inhibits cyclooxygenase – 2, and if the risk of bleeding is a concern, it can be preferable.
Can Mobic be taken by anemic patient?
In patients treated with Mobic and other nonsteroidal anti-inflammatory drugs (NSAIDs), there is a dose-dependent reduction in serum hemoglobin and hematocrit has been observed. However, anemia has been reported occasionally. Induced fluid retention or gastrointestinal blood loss or erythropoiesis (incompletely described effect) could be the possible mechanisms behind it.
In patients with pre-existing anemia, when large doses of NSAIDs (such as Mobic) are given, the decrease in hemoglobin concentration can be increased 1g/dL which otherwise remain slight with average doses of NSAIDs.
Therefore the Mobic should be used with caution in patients with anemia or in patients who are predisposed to anemia. Clinical monitoring of hematopoietic function may be appropriate, particularly during chronic therapy.
- Due to occult or gross blood loss, fluid retention or an incompletely described effect of erythropoiesis, anemia can occur in patients treated with anemia.
- Any signs or symptoms of anemia, hemoglobin or hematocrit should be monitored in patients treated with Mobic or other NSAIDs. It can even increase the risk of bleeding events.
- Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding.
Can Mobic cause skin reactions?
The use of NSAIDs has been associated with severe and fatal dermatologic reactions such as Stevens-Johnsons syndrome, toxic epidermal necrolysis, and another exfoliative dermatitis. These events may occur without warning. The patient should seek immediate medical help in case of the first sign of skin reaction, rash even if it is mild, fever, itching, blisters, itching, or any other sign of hypersensitivity, and should stop the Mobic immediately.
In patients with or without known hypersensitivity to meloxicam, and in patients with aspirin-sensitive asthma, the Mobic can cause severe anaphylactic reactions.
Can Mobic be used in pregnancy?
Mobic comes under the pregnancy category C, the drugs which even after owing to the pharmacological effects, showed to be caused or may be suspected of causing harmful effects on the human fetus or neonates without causing many malformations.
These effects may be reversible. The adverse effects have been shown on the fetus as per animal studies but there are no controlled and adequate studies in human. However, the potential benefits of the Mobic may warrant the use of this drug in pregnant women despite its severe risks.
Mobic which contains meloxicam also can cause premature closure of fetal ductus ateriosus. One should completely avoid the use of Mobic in a pregnant woman in her third trimester (starting at 30 weeks of gestation).
Mobic should also not be used in a nursing mother. Because this medication is known to excrete in the human milk. However, the severe or dangerous effects of the Mobic in the nursing infant are unknown. Other agents in order to treat the conditions like arthritis, or rheumatoid arthritis, should be preferred in order to minimize the potential risk of Mobic to a nursing infant or the mother.
Can Mobic be taken with duloxetine?
Duloxetine is an SRI (serotonin reuptake inhibitor). The risk of bleeding in patients treated with ulcerogenic agents can be increased with serotonin reuptake inhibitors (SRIs) and agents that affect hemostasis such as anticoagulants, platelet inhibitors, thrombin inhibitors, thrombolytic agents, or agents that commonly cause thrombocytopenia.
Other drugs that can interact with it are clomipramine which is also a strong SRI (serotonin reuptake inhibitor). In hemostasis, serotonin release may play an important role. SRIs can induce bleeding by altering the platelet function in patients treated with psychotropic agents (that can interfere with serotonin reuptake).
The concurrent use of NSAIDs (Nonsteroidal anti-inflammatory agents such as Mobic) and SRIs (serotonin reuptake inhibitors such as duloxetine) have shown the association between upper gastrointestinal bleeding.
There may be a pharmacodynamic interaction between SSRIs and oral anticoagulants (for example Warfarin) and studies showed that this combination can cause increased bleeding diathesis without apparent changes in the disposition of either drug or changes in the time of prothrombin.
One should practice caution if SRIs and NSAIDs are used in combination. The close observation by the laboratory is recommended for hematologic complications.
Any signs of bleeding, pain, swelling, headache, dizziness, weakness, prolonged bleeding from cuts, increased menstrual flow, vaginal bleeding, nosebleeds, red or brown urine, or red or black stools like symptoms should be reported immediately and should be reported to the physician without any delay in order to prevent any severe complication.
Can Mobic be taken with lisinopril?
Lisinopril is an ACE (angiotensin-converting enzyme) inhibitor and is used as an antihypertensive agent. When given along with NSAIDs (Nonsteroidal anti-inflammatory agents such as meloxicam), the antihypertensive effects of ACE inhibitors can get attenuated. The NSAIDs acts by inhibiting the renal prostaglandins which also results in unopposed pressor activity thus producing hypertension.
Along with it, NSAIDs can also cause fluid retention that also aids in increasing the blood pressure. The pharmacokinetics of ACE inhibitors also get altered by the concurrent administration of NSAIDs, like The systemic exposure of enalapril (and its metabolite – enalaprilat) has been reduced by the oxaprozin.
One should completely avoid the concurrent administration of NSAIDs and ACE inhibitors as they can deteriorate the renal function, especially in the patients where volume already depleted (such as in case of diuretics therapy) or where the patient has a compromised renal function. The patient can suffer from acute renal failure; however, the effects are reversible.
The renal toxicities, such as elevations in serum creatinine, tubular necrosis, glomerulitis, renal papillary necrosis, acute interstitial nephritis, nephrotic syndrome and renal failure can also cause by the chronic use of NSAIDs alone.
These nonsteroidal anti-inflammatory agents can also precipitate the overt renal decompensation via a dose-related prostaglandin synthesis in patients with prerenal conditions (where renal function dependent on the prostaglandin functions).
And when these NSAIDs are given with the Ace inhibitors, they can further worsen the renal function by reducing the glomerular filtration by blocking the effects of angiotensin-II mediated vasoconstriction.
The regular blood pressure monitoring is mandatory for the patients receiving ACE inhibitors and NSAIDs concurrently. Blood pressure monitoring should be followed by initiation, discontinuation, or change of dosage of NSAIDs. If the co-administration of these medicines is done for a long time, the renal function should also be evaluated. The low doses or short-term administration of NSAIDs are not expected to cause any interaction.
Can Mobic be taken with sertraline (Zoloft)?
Agents that affect the hemostasis (like anticoagulants, platelet inhibitors, thrombin inhibitors, thrombolytic agents, or agents responsible for thrombocytopenia) and ulcerogenic agents should be avoided in patients treated with serotonin reuptake inhibitors as they can potentiate the risk of bleeding in such patients.
Other agents such as tricyclic antidepressants (clomipramine) can also interact in the same way. Serotonin release by platelets plays an important role in hemostasis, thus SRIs may alter platelet function and induce bleeding.
The psychotropic agents can interfere with serotonin reuptake and can cause bleeding episodes in patients as per published case reports. Events such as ecchymosis (discoloration of the skin underneath due to bleeding, mainly due to bruises), hematoma, epistaxis and petechiae (red or purple spotting on the skin due to bleeding) to life-threatening hemorrhages can be caused by psychotropic agents as a result of interference in serotonin reuptake.
One should use the concurrent administration of clomipramine and NSAIDs or even with the drugs that can cause hemostasis. Close clinical and laboratory observation for hematologic complications is recommended.
Patients should be advised to promptly report any signs of bleeding to their physician, including pain, swelling, headache, dizziness, weakness, prolonged bleeding from cuts, increased menstrual flow, vaginal bleeding, nosebleeds, bleeding of gums from brushing, unusual bleeding or bruising, red or brown urine, or red or black stools.
Can Mobic be taken with prednisone?
The concurrent administration of Prednisone (a corticosteroid) and NSAIDs (nonsteroidal anti-inflammatory drugs) can enhance the serious gastrointestinal events such as inflammation, bleeding, ulceration, and perforation. The risk of peptic ulcer disease or gastrointestinal hemorrhage has also been reported in elderly patients after concurrent administration of corticosteroids and NSAIDs.
The risk is seen as around 14.6 times higher as compared to zero in patients who did not use any medication (neither corticosteroid nor NSAIDs). The risk is higher in the case when corticosteroids are taken orally.
The additive effects of both medications, when taken concurrently, can result in ulcerogenic effects or additive effects in gastrointestinal mucosa. The erosions caused by the NSAIDs may get delayed in healing due to primary effects of corticosteroids (when administered in combination).
In patients with a history of peptic ulcer disease or gastrointestinal bleeding in elderly patients or debilitated patients, one should practice extreme caution, if corticosteroids and NSAIDs are taken altogether.
These medications on combination should be administrated with food and if a patient seeks the symptoms of gastrointestinal bleeding or gastrointestinal ulceration (like abdominal pain, dizziness, lightheadedness, the appearance of black or tarry stools) should immediately inform the doctor or seek medical attention immediately. Antacids and H2 antagonists should be used as prophylactic antiulcer therapy in such cases.
“Can I take hydrocodone with meloxicam?”
What does saphris do to the brain?“