Contents
- What is nebivolol?
- Nebivolol molecular structure, weight, formula, IUPAC name, desription and drug class
- Nebivolol appearence and identification
- What is the mechanism of action of nebivolol?
- What are the indications of nebivolol?
- What are the contraindications of nebivolol?
- Nebivolol and abrupt cessation of therapy
- Nebivolol and bronchospastic diseases
- Anesthesia and major surgery and nebivolol
- Nebivolol and diabetes
- Nebivolol and thyroid storm
- Nebivolol and peripheral vascular disease
- Nebivolol and pheochromocytoma
- Pharmacokinetics of nebivolol
- How is nebivolol stored and dispensed?
- Side effects of nebivolol
- What should I do if I overdose on nebivolol?
- Nebivolol dosage
- Price and prescription of Nebivolol
- Can I take nebivolol if I am pregnant?
- Can I take nebivolol if I am breastfeeding?
- Can I take nebivolol if I have liver impairment?
- Can I take nebivolol if I have kidney dysfunction?
- Does nebivolol cause infertility?
- Can nebivolol cause cancer?
- Should I stop using nebivolol now that I have heart failure?
- How safe is nebivolol for people aged above 65 years of age?
- What are some drugs that can interact with nebivolol?
- How long does nebivolol stay in your system?
- Can I take alcohol with nebivolol?
- May nebivolol provoke high cholesterol levels
- Can I take nebivolol and verapamil together?
- Can I take nebivolol and aminophylline together?
- Can I take nebivolol and digoxine together?
What is nebivolol?
Nebivolol belongs to the group of drugs called beta blockers. It acts only on the heart and vessels and hence is called cardiovascular specific. Nebivolol is used to treat hypertension and left heart failure.
Nebivolol is available in four different strengths: 2.5 mg, 5 mg, 10 mg and 30 mg. It is not available as generic nebivolol till now and will be available as generic medicine after the year 2021 after which its price is expected to fall. Currently it is marketed under different brand names such as Bystolic, Nodon, Nubeta etc. In most countries, it is available as prescription only drug.
Nebivolol is also available as a mixture product. The name of that drug is Byvalson and contains nebivolol 5 mg and valsartan 80 mg in each of its tablets.
Nebivolol molecular structure, weight, formula, IUPAC name, desription and drug class
Nebivolol is a highly cardioselective vasodilatory beta1 receptor blocker used in treatment of hypertension. In most countries, this medication is available only by prescription.
IUPAC name: 1-(6-fluoro-3,4-dihydro-2H-chromen-2-yl)-2-[[2-(6-fluoro-3,4-dihydro-2H-chromen-2-yl)-2-hydroxyethyl]amino]ethanol
Molecular formula: C22H25F2NO4
Molecular weight: 405.442 g/mol
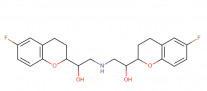
Molecular structure:
Drug Class: Nebivolol is an organic compound belonging to the class of 1-benzopyrans. These compounds contain a bicyclic compound made up of a benzene ring which is fused to a pyran, so that the oxygen atom is at the 1-position.
Nebivolol appearence and identification
Nebivolol Identification: Nebivolol is a white to off white powder that is soluble in dimethylsulfoxide, methanol and N,N-dimethylformamide. It is sparingly soluble in propylene glycol, polyethylene glycol and ethanol and only slightly soluble in methylbenzene, hexane and dichloromethane.
What is the mechanism of action of nebivolol?
The mechanism of action of nebivolol has not been clearly understood till now. However the following factors contribute towards the antihypertensive action of nebivolol
- It is a selective β1-receptor antagonist. These receptors are present on the sinoatrial node as well as on the muscular wall of the heart. Normally epinephrine and norepinephrine bind with these receptors to increase the heart rate and contractility of myocardium which will pump more blood into the vessels and increase blood pressure.
- Nebivolol being an antagonist will block these two action from taking place and contribute towards decreasing blood pressure.
- Nebivolol is also found to suppress the release of renin, an enzyme which converts angiotensinogen to angiotensin I which is then converted to angiotensin II. Angiotensin II is a vasoconstrictor.
- It also stimulates the release of aldosterone which is responsible for salt and water retention. Blocking renin will block this entire pathway and result in a decreased blood pressure.
- Nebivolol is also responsible for vasodilation as it has a nitric oxide like action on the beta-3 receptors present directly on the wall of the vessels. This will reduce the total peripheral resistance and bring down the blood pressure.
What are the indications of nebivolol?
The only FDA approved indication of nebivolol is treatment of essential hypertension. However it can be given to other individuals suffering from diabetic nephropathy or left sided heart failure.
What are the contraindications of nebivolol?
Nebivolol is absolutely contraindicated in the following patients
- Severe bradycardia
- Heart block greater than 1st degree
- Patients suffering from cardiogenic shock
- Decompensated heart failure
- Sick sinus syndrome (not a contraindication if a pacemaker is inserted)
- Patients with moderate to severe hepatic disease
- Patients with known hypersensitivity to nebivolol or any other component of the pill
Nebivolol and abrupt cessation of therapy
Abrupt cessation of nebivolol in patients with coronary artery disease will cause exacerbation of the condition any may present as angina, myocardial infarction or ventricular arrhythmias.
Patients should be informed about these side effects. If discontinuation is needed, it should be gradual and planned. Dose should be tapered over 1 to 2 weeks and patients should be advised to minimize physical activity. In case ischemic pain or arrhythmias develop, restart nebivolol immediately.
Nebivolol and bronchospastic diseases
Beta receptors present on the airways are responsible for bronchodilation. Giving a beta blocker will therefore cause bronchoconstriction. If an individual is suffering from a bronchospastic disorder e.g. asthma, his condition is likely to be worsened with the use of beta blockers.
Anesthesia and major surgery and nebivolol
There are certain anesthetics that depress myocardial function. Giving nebivolol (which also reduces contractility of the heart) raises the possibility of significant reduction in cardiac output. However, when beta blockers are given there is usually overexpression of the adrenergic receptors on the heart.
So when therapy is withdrawn and epinephrine released due to the stress of major surgery, the heart rate and contractility both will increase by many folds. This will increase myocardium oxygen demand and exacerbate underlying ischemia of the heart.
Evaluating both the pros and cons of stopping therapy, it has been advised to continue giving nebivolol during the perioperative period but such pateints should be monitored closely
Nebivolol and diabetes
One of the signs of hypoglycemia is shivering. This is because during hypoglycemia, skeletal muscle glycogen is broken down to glucose (this step is mediated by beta receptors).
The muscle uses this glucose and contracts and the patient appears to be shivering. So when a beta blocker is given, the conversion of glycogen to glucose is inhibited.
As a result, shivering due to hypoglycemia does not take place. Another sign of hypoglycemia is tachycardia. Since nebivolol is responsible for keeping a check on the heart rate, this sign too can be masked with the use of nebivolol. Beta blockers may also potentiate insulin-induced hypoglycemia.
All these things mean that mild hypoglycemia will go undiagnosed and will only be diagnosed when it is quite severe. Such severe hypoglycemia can cause brain damage.
Nebivolol and thyroid storm
Patients with thyrotoxicosis are usually prescribed beta blockers to treat tachycardia. They are also found to inhibit the peripheral conversion of T4 to T3.
Sudden withdrawal of beta blockers in such patients will expose the heart to unopposed action of thyroid hormones and can cause arrhythmias such as atrial fibrillation. Sudden withdrawal can also precipitate a thyroid storm.
Nebivolol and peripheral vascular disease
Generally beta blockers are known to worsen the condition of patients with peripheral vascular disease. Although nebivolol is one of the 4 beta blockers which can cause vasodilation and can be expected to improve the condition, however since no research has been done on it, the general rule for beta blockers should be applied to observe caution in patients with PVD.
Nebivolol and pheochromocytoma
Patients suspected of having pheochromocytoma should be given alpha blockers before prescribing beta blockers. Beta blockers alone will cause unopposed vasoconstriction of the vessels which will further increase blood pressure.
Pharmacokinetics of nebivolol
Absorption: Nebivolol is given through the oral route. Its bioavailability has not been studied. The absorption of nebivolol is not affected by food. Peak plasma concentration of nebivolol is reached between 1.5 to 4 hours and depends upon how quicly an individual can metabolize the drug.
Distribution: Nebivolol is highly bound to plasma proteins. At any given time, the amount of drug bound to these proteins is 98%.
Metabolism: Nebivolol undergoes a number of metabolic pathways. The most common pathway is glucoronidation. Other pathways the parent drug might undergo are hydroxylation, N-dealkylation and oxidation. All these reactions occur in the liver and are carried out by the P450 enzymes of the liver (enzyme subgroup involved is the CYP2D6 system).
The main metabolic pathway yields an active metabolite called d-nebivolol. The half-life of the drug varies from individual to individual as the ability to metabolize the drug varies from person to person.
Those who have a very active CYP2D6 system of enzymes are called extensive metabolizers (EMs) and those who a slow CYP2D6 system of enzymes are called poor metabolizers (PMs). The half-life in extensive metabolizers is 12 hours while the half-life in poor metabolizers is 19 hours.
Excretion: Urinary and fecal route are the two most common routes for the excretion of nebivolol. In extensive metabolizers 38% of the drug is excreted via the urinary route while 44% of the drug is excreted in the feces.
In poor metabolizers 67% of the drug is excreted via the urinary route while 13% of the drug is excreted in the feces. All of the drug is excreted as metabolic products and the amount of parent drug which is excreted is very little.
How is nebivolol stored and dispensed?
Nebivolol is stored at temperatures of 20 °C to 25 °C. The environment should be moisture free and it should be stored in an air tight container whose lid is child-proof.
The rules for dispensing the medicine vary for each country. In most countries it is a prescription only drug.
Side effects of nebivolol
The following side effects of nebivolol are considered to be mild in nature and usually, do not require any immediate medical attention:
- Mild headache
- Diarrhea
- flatulence or bloating
- rash
- agitation
- trouble sleeping
- mild anxiety
The following side effects of nebivolol are considered to be severe and need immediate medical attention:
- Fatigue
- Swelling of the subcutaneous tissues of the face, arms, hands, lower legs, or feet (angioedema)
- Pain in the chest or epigastrium
- Feeling of squeezing the chest
- Difficulty in breathing
- Fast breathing
- lightheadedness, dizziness, or fainting
- weight gain in a short period of time
- Unexplained weight loss
- slow heartbeat
- irregular heartbeat
- tingling sensations in the hinds or feet
- unusual tiredness or weakness
- new onset wheezing
What should I do if I overdose on nebivolol?
The following are the signs and symptoms of an overdose on nebivolol:
- Absent body movement
- Decreased body movements
- anxiety
- blue lips, fingernails, or skin (signs of cyanosis)
- change in visual acuity
- chills
- cold sweats
- coma
- confusion
- cold and pale skin
- continuous cough
- decreased urine output
- depression
- dilated neck veins
- signs of postural hypotension: dizziness, faintness, or lightheadedness when getting up suddenly from a lying or sitting position
- dry mouth
- extreme fatigue
- fast heartbeat
- headache
- increased hunger
- increased sweating
- increased thirst
- irregular, fast or slow, or shallow breathing
- loss of appetite (anorexia)
- mood changes
- muscle pain or cramps
- nauseaor vomiting
- nervousness
- nightmares
- noisy breathing: stridor or wheeze
- numbnessin the hands, feet, or lips
- seizures
- shakiness
- slurred speech
- troubled breathing
If you have taken or suspect of taking more than the prescribed dose of nebivolol and are now experiencing any of the following signs and symptoms, go to your nearest ER immediately.
Management in the ER involves stomach wash as well as symptomatic treatment. The supportive measure should continue till the time the patient is clinically stable.
The half-life of normal doses of nebivolol is between 12 to 19 hours but in cases of overdose it is expected to be much higher. Therefore it might take 3 to 4 days for the patient to achieve clinical stability.
Common symptoms of overdose along with their specific treatment is given below:
- Bradycardia: Give IV atropine immediately. If the clinical response is unsatisfactory, give isoproterenol or any other drug which has a positive chronotropic affect. Observe the patient while giving these drugs. In some cases, transvenous or transthoracic pacemaker installation maybe needed.
- Hypotension: Immediately administer 500 to 1000 ml of fluids. If the response is insufficient add vasopressors. Intravenous glucagon may also be given.
- Second or third degree heart block: Initiate isoproterenol infusion and monitor the patient. In some cases, transvenous or transthoracic pacemaker installation maybe needed.
- Congestive cardiac failure: First line of therapy includes giving digoxin and diuretics. Other drugs which are shown to reduce mortality in congestive heart failure are beta blockers, hydralazine, spironolactone and installation of a pacemaker.
- Bronchospasm: Short acting beta agonists such as albuterol are given as the first line of therapy. Other drugs used in case of no response are inhaled corticosteroids, anticholinergics (e.g. ipratropium), systemic steroids, magnesium, and finally epinephrine. If all fails endotracheal intubation is done. Aminophylline is no longer used in cases of acute bronchospasm.
- Hypoglycemia: Initiate IV glucose or 5% dextrose. Monitor blood sugar level every 20-30 mins. Repeated doses of IV glucose or 5% dextrose or glucagon infusion maybe needed to treat hypoglycemia.
Nebivolol dosage
The initial recommended dose of nebivolol for the treatment of hypertension is 5 mg orally once a day. If needed, the dose can be increased to a maximum of 40 mg once a day. For increasing the dose, it is recommended to titrate the dose after every 2 weeks. Titrating dose earlier than 2 weeks is not considered to be beneficial.
The minimum dose needed for maximum response is called the optimum dose and is variable for every individual.
Nebivolol can be used alone, or in combination with other antihypertensive drugs.
Nebivolol should be taken once a day at the same time each day with or without food. If you have missed a dose, you can take it as soon as you remember, however, if the next dose is close, skip the missed dose. Never take double the dose.
General consideration
- Transient worsening of bradycardia, heart failure or hypotension may take place during the titration period. If these are severe, therapy should be discontinued.
- During the titration period, following monitoring is needed:
- Monitoring of heart rate and blood pressure
- Be on the lookout for signs and symptoms of heart failure (e.g., shortness of breath which worsens by lying down, edema, and weight gain), angina and myocardial infarction.
Price and prescription of Nebivolol
Nebivolol is used to treat hypertension and when it is compared with other antihypertensives, it is a relatively expensive drug. If you’re covered by the government funded medicare or have private insurance, nebivolol is most likely to be covered by it.
However if you don’t have any medical cover, you should look out for various coupons and discounts by pharmacies as it is an expensive drug.
An example of such a pharmacy is the online drug retailer GoodRx where the cheapest price a 30 tablet pack is $138.44. This price is 15% less than the average retail price of Bystolic which is $163.49.
Can I take nebivolol if I am pregnant?
There is no scientific research in humans to answer this question. However research was carried out on animals, the results of which are given below:
When very high dose nebivolol was administered to rats during organogenesis period, reduced fetal body weight was observed. Apart from this, there were small reversible delays in sternal and thoracic ossification.
When very high dose nebivolol was administered to pups during the perinatal period, prolonged gestation, dystocia, increases in late fetal deaths and stillbirths and decreased birth weight were observed.
Due to the above data collected in animals, nebivolol has been classified as a pregnancy category C drug i.e. not enough research has been done to determine if these drugs are safe during pregnancy hence should only be given when benefits far outweigh the risks.
Can I take nebivolol if I am breastfeeding?
There is no scientific research to assess the safety of breastfeeding while breastfeeding in humans. However it has been seen in rats that both nebivolol as well as its metabolic products cross the placental barrier and their presence is also found in the breast milk of rats.
Once that milk is ingested by the infant, he is now exposed to nebivolol himself which can cause bradycardia or heart block in the infant. It is due to this potential side effect that you should either breastfeed your child or take nebivolol, but don’t do both things at the same time.
Can I take nebivolol if I have liver impairment?
Almost all of the nebivolol that is absorbed undergoes metabolization in the liver. Therefore hepatic dysfunction is going to be associated with slow metabolization and increased half-life of the drug. The peak plasma concentration of nebivolol is also seen to be raised by 3 folds in such a population.
Patients having moderate hepatic impairment can initiate nebivolol at 2.5 mg once a day and titrated slowly if needed up to a level that is well tolerated. However there has been no study on the dosing and pharmacokinetics of nebivolol in patients with severe hepatic impairment and as a result this drug is contraindicated in this population.
Can I take nebivolol if I have kidney dysfunction?
Patients with mild renal dysfunction (ClCr 50 to 80 mL/min) showed no change in the clearance of nebivolol. Patients with moderate renal dysfunction (ClCr 30 to 50 mL/min) showed only minor reduction in clearance of nebivolol, however, patients with severe renal dysfunction (ClCr <30 mL/min) showed a 53% decrease in the clearance of nebivolol.
Another problem with nebivolol is that it shows 98% plasma protein binding. Therefore hemodialysis will not be useful.
From the above data the following recommendations have been drawn:
- Patients with mild or moderate kidney dysfunction do not need any dose adjustments and should continue their normal dose. Those who are starting this medicine can start with the usual 5 mg dose once a day.
- Patients with severe kidney dysfunction are recommended to start with an initial dose of 2.5 mg and if needed, it can be titrated up slowly.
Does nebivolol cause infertility?
When nebivolol was given to mice at a dose which were 0.3 to 5 times the maximum recommended human dose for 2 years, it was noticed that there was a significant increase in the incidence of testicular Leydig cell hyperplasia and adenomas.
Co-administration of dihydrotestosterone reduced blood LH levels and decreased the incidence of Leydig cell hyperplasia. Effect of nebivolol on sperms was noted to be reversible.
In humans, there has only been one research and that too for only 4 weeks to determine the effects of nebivolol on adrenal function, luteinizing hormone, and testosterone levels.
The study concluded that nebivolol had no effect on adrenal function or fertility of males. However since the study was short term, it cannot completely rule out the risk of Leydig cell hyperplasia and adenomas which were seen in mice. Therefore caution must be exercised when taking nebivolol for years.
Can nebivolol cause cancer?
Nebivolol was not found to be harmful to DNA in a number of assays. However it did cause testicular adenomas (benign tumors) in mice when administered for 2 years. Therefore more research needs to be done to ascertain the relationship between nebivolol and cancer.
Should I stop using nebivolol now that I have heart failure?
1067 patients suffering from congestive heart failure taking nebivolol were compared with 1061 patients suffering from congestive heart failure but on placebo.
There was no worsening of heart failure while taking nebivolol. From the conclusion of this study, the recommendation made was that you can continue to use nebivolol even if you suffer from heart failure, but if it starts to worse, then discontinue nebivolol.
How safe is nebivolol for people aged above 65 years of age?
The incidence of adverse effects and efficacy of nebivolol was studied on 478 patients above the age of 65and compared with 2322 patients below the age of 65. There was no overall difference in the safety or efficacy between older and younger patients.
What are some drugs that can interact with nebivolol?
Nebivolol has a number of minor, moderate and major drug interactions. The major drug interactions are:
- CYP2D6 Inhibitors: Nebivolol is metabolized by CYP2D6 enzyme system of liver. Any drug which inhibits these enzymes will cause the metabolism of nebivolol to be delayed and its half-life increased. Examples of CYP2D6 inhibitors include quinidine, propafenone, fluoxetine, paroxetine, etc.
- Hypotensive Agents: Do not use nebivolol with other beta blockers. Patients receiving catecholamine-depleting drugs, such as reserpine or guanethidine should be closely monitored because using nebivolol in combination with these drugs may produce excessive reduction in sympathetic activity.
- Cardiac Glycosides: Cardiac glycosides such as digoxin slow atrioventricular conduction and decrease heart rate. This same effect is also brought on by beta blockers. Concomitant use of digoxin with beta blockers increases the risk of bradycardia and heart block.
- Calcium Channel Blockers: Non-dihydropyridine Calcium Channel Blockers such as verapamil and diltiazem will slow atrioventricular conduction and decrease heart rate. Concomitant use of these calcium channel blockers with beta blockers increases the risk of bradycardia and heart block.
- Aminophylline: Aminophylline and beta blockers work in opposition to each other. Combined use of these two drugs with produce only a partial response.
- Alpha-2 adrenergic agonist: Alpha-2 adrenergic agonist such as clonidine decrease the activity of the autonomic nervous system. This has the potential to decrease blood pressure and heart rate. Combined use of Alpha-2 adrenergic agonist can cause hypotension, bradycardia or heart block.
How long does nebivolol stay in your system?
In a normal healthy individual, nebivolol has a half-life of 12 hours in early metabolizers and it may increase up to 19 hours in poor metabolizers. It takes 5.5 half-lives to completely remove any drug from our body.
Therefore the time needed to completely remove nebivolol from an individual’s system after complete cessation of the drug is variable and ranges from 66 hours to 105 hours.
The time for complete removal of the drug is also influenced by kidney function, liver function as well as age of the patient. Therefore it is advisable to adjust the dose of nebivolol if you are aged, or have suboptimum renal or liver function.
Can I take alcohol with nebivolol?
Coadministration of alcohol with antihypertensives and other hypotensive agents, in particular vasodilators and alpha-blockers such as nebivolol, may result in additive effects on blood pressure and orthostasis.
Nebivolol and ethanol may have additive effects in lowering your blood pressure. You may experience headache, dizziness, lightheadedness, fainting, and/or changes in pulse or heart rate.
These side effects are most likely to be seen at the beginning of treatment, following a dose increase, or when treatment is restarted after an interruption. Let your doctor know if you develop these symptoms and they do not go away after a few days or they become troublesome.
Avoid driving or operating hazardous machinery until you know how the medications affect you, and use caution when getting up from a sitting or lying position. It is important to tell your doctor about all other medications you use, including vitamins and herbs.
Do not stop using any medications without first talking to your doctor.
May nebivolol provoke high cholesterol levels
Beta-adrenergic receptor blocking agents including nebivolol may alter serum lipid profiles. Increases in serum VLDL and LDL cholesterol and triglycerides, as well as decreases in HDL cholesterol, have been reported with some beta-blockers.
Patients with preexisting hyperlipidemia may require closer monitoring during beta-blocker therapy, and adjustments made accordingly in their lipid-lowering regimen.
Can I take nebivolol and verapamil together?
dditive reductions in heart rate, cardiac conduction, and cardiac contractility may occur when calcium channel blockers, especially verapamil and diltiazem, are used concomitantly with beta-blockers.
While this combination may be useful and effective in some situations, potentially serious cardiovascular adverse effects such as congestive heart failure, severe hypotension, and/or exacerbation of angina may occur.
Ventricular asystole, sinus arrest, and heart block have also been reported. The risk is increased with high dosages, IV administration, left ventricular dysfunction, or AV conduction abnormalities. Beta-blocker ophthalmic solutions may also interact, as they are systemically absorbed and can produce clinically significant systemic effects even at low or undetectable plasma levels.
Bradycardia (36 bpm) with wandering atrial pacemaker occurred in a patient receiving oral verapamil and timolol ophthalmic drops.
The proposed mechanisms include additive slowing in AV conduction, reduced cardiac contractility secondary to beta-blockade, and decreased peripheral vascular resistance secondary to calcium channel blockade. Verapamil and diltiazem may also decrease the clearance of some beta-blockers.
Can I take nebivolol and aminophylline together?
The pharmacologic effects of theophyllines and beta-blockers are opposite. Nonselective and high doses of cardioselective beta-blockers may cause severe or fatal bronchospasm by opposing theophylline-induced bronchodilation.
Ophthalmic beta-blockers undergo significant systemic absorption and may also interact.
In addition, propranolol and other beta-blockers may reduce the CYP450 hepatic metabolism of theophylline, and serum theophylline levels may be increased.
Can I take nebivolol and digoxine together?
Concomitant use of digitalis glycosides and beta-blockers may increase the risk of bradycardia. These agents slow atrioventricular conduction and decrease heart rate, hence they may have additive cardiac effects during coadministration.
Some beta-blockers such as carvedilol, esmolol, and talinolol have also been reported to increase the systemic bioavailability of digoxin.
The mechanism may involve enhanced absorption as well as reduced renal excretion of digoxin due to inhibition of intestinal and renal P-glycoprotein efflux transporter.