Contents
- What is PLIVA 433 pill?
- Active ingredients of PLIVA 433 pill and identification
- PLIVA 433 pill chemistry
- PLIVA 433 pill uses
- PLIVA 433 pill legal status
- PLIVA 433 pharmacokinetics
- PLIVA 433 pill use during pregnancy and breastfeeding
- What are different brand names for trazodone?
- PLIVA 433 pill side effects
- How long PLIVA 433 pill stays in the system?
- PLIVA 433 pill precautions and warnings
What is PLIVA 433 pill?
PLIVA 433 is an imprint on a pill identified as white in a round shape. Tablets are biconvex beveled and bisect on one side and plain on other side. The bisected side of tablet is debossed with ‘8’ on the upper side of bisect and ’05’ on the lower side of bisect. The pill contains trazodone hydrochloride as an active ingredient in the dose of 50 mg. Trazodone belongs to the drug class phenylpiperazine antidepressants and is used in the treatment of insomnia, anxiety, fibromyalgia, sedation and major depressive disorder. It is supplied by Major Pharmaceuticals.
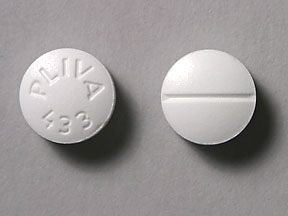
- Imprint: PLIVA 433
- Strength: 50 mg
- Color: White
- Size: 9.00 mm
- Shape: Round
- Availability: Prescription only
- Drug Class: Phenylpiperazine antidepressants
- Pregnancy Category: C – Risk cannot be ruled out
- CSA Schedule: Not a controlled drug
- Labeler / Supplier: Major Pharmaceuticals
- Inactive Ingredients: hypromellose, lactose monohydrate, magnesium stearate, polydextrose, polyethylene glycol, povidone, pregelatinized starch, silicon dioxide, and titanium dioxide
Active ingredients of PLIVA 433 pill and identification
Trazodone hydrochloride is an antidepressant drug that affects chemicals in the brain that may be unbalanced in people with depression. It is atypical antidepressive drug chemically unrelated to tricyclic, tetracyclic, or other known antidepressant agents. It is used to treat the major depressive disorder.
Trazodone hydrochloride appears as a white odorless crystalline powder, freely soluble in water.
PLIVA 433 pill chemistry
Trazodone hydrochloride:
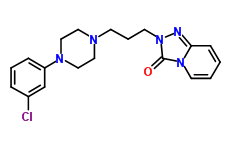
IUPAC name: 2-{3-[4-(3-chlorophenyl)piperazin-1-yl]propyl}-2H,3H-[1,2,4]triazolo[4,3-a]pyridin-3-one
Molecular formula: C19H22ClN5O
Molecular weight: 371.864 g/mol
Molecular structure:
Drug class: Trazodone hydrochloride belongs to the class of organic molecules known as phenylpiperazines which contain a phenylpiperazine moiety made of piperazine bound to a phenyl group.
PLIVA 433 pill uses
PLIVA 433 pills are indicated for the treatment of depression. The efficacy of the drug has been showed in both outpatient and inpatient settings and for depressed patients with and without prominent anxiety.
FDA approved indications are:
- Alcohol Dependence
- Feeling Anxious
- Major Depressive Disorder (MDD)
- Sleeplessness
PLIVA 433 pill legal status
Trazodone hydrochloride is not a controlled substance. It is available via prescription.
PLIVA 433 pharmacokinetics
Absorption. PLIVA 433 is well absorbed after oral administration with no selective localization in any tissue. When the medicine is taken shortly after ingestion of food, there may be an increase in the amount of drug which is absorbed, a decrease in maximum concentration and a lengthening in the time to maximum concentration. Maximum levels occur about one hour after ingestion if drug is taken on an empty stomach or two hours after dosing when taken with food.
Metabolism. In vitro studies showed that trazodone is metabolized to an active metabolite, m-chlorophenylpiperazine (mCPP) via cytochrome P450 3A4 (CYP3A4). Other metabolic pathways that may be involved in metabolism of trazodone have not been well characterized.
Elimination. In some patients, trazodone may accumulate in the plasma.
PLIVA 433 pill use during pregnancy and breastfeeding
PLIVA 433 pills may be used during pregnancy only if the benefit outweighs the risk to the fetus. Trazodone is classified in US FDA pregnancy category: C which means that if the drug is used until delivery, newborns should be monitored for the occurrence of withdrawal symptoms.
Limited data indicate that trazodone levels in milk are low and would not be expected to cause any side effects in breastfed infants, especially if the infants are older than 2 months or when doses of 100 mg or less are used at bedtime for sleep.
What are different brand names for trazodone?
Different brand names for trazodone are: Desyrel, Oleptro, Desyrel Dividose
PLIVA 433 pill side effects
Trazodone hydrochloride may cause following side effects:
Common side effects:
- Headache
- Muscle ache
- Nausea, vomiting, loss of appetite, or stomachache
- Constipation or diarrhea
- Loss of interest in sex (erectile dysfunction in men)
- Dizziness or loss of balance
- Dry mouth or dry eyes
- Numbness, burning, or tingling sensations
- Confusion
- Blurred vision
- Ringing in the ears
- Nervousness or confusion
- Rash
- Sweating
- Weakness or fatigue
Serious side effects:
- Worsening depression
- Suicidal thoughts
- A severe rash or hives
- Swelling of the face, lips, or tongue
- Chest pain
- Difficulty breathing
- A painful erection that will not go away (priapism)
- Panic attack
- Irregular heartbeat
- Fainting
- Unusual bruising or bleeding
- Seizure
How long PLIVA 433 pill stays in the system?
The elimination half-life of PLIVA 433 pills is considered to be in the range of 5 to 9 hours, although some sources estimate 7.3 hours. According to this data, after discontinuation, it will take around 7.3 hours to eliminate 50% of the drug from the system. To completely clear PLIVA 433 pills from the body, it’ll take about 1.67 days.
After ingestion, trazodone is broken down into different metabolites, some of which are thought to linger for a longer period than the parent compound. Trazodone is metabolized into a primary metabolite known as “m-chlorophenylpiperazine” (mCPP) which has an elimination half-life of 2.4 to 6.8 hours. However, there are also other metabolites of trazodone that have been reported to carry an elimination half-life of up to 13 hours.
Although trazodone will be fully excreted in 2 days, elimination of different metabolites could take 72 hours. According to all stated, it can be expected that trazodone will be fully eliminated in 4 days after cessation.
PLIVA 433 pill precautions and warnings
- The FDA requires a black-box warning on trazodone, noting that taking the drug could increase the chance of becoming suicidal. The suicidal risk is greatest at the beginning of treatment or when increasing or decreasing the dose of trazodone. The risk may be higher for people younger than 24.
- Depression may become worse before it gets better at the beginning of therapy with trazodone.
- Healthcare provider should monitor carefully for symptoms of aggression, panic attacks, irritability, extreme worry, acting without thinking, restlessness, abnormal excitement, or any thoughts of suicide when one start the drug.
- Friends and family members should know if one has these symptoms or worsening symptoms of depression.
- If trazodone is abrupt suddenly it may cause withdrawal symptoms such as anxiety, agitation, and difficulty sleeping.
- Children or teens should not take trazodone.
“Does Pliva 433 get you high?”
“What is a pill marked L484?“