Contents
- What is risedronate?
- Risedronate Chemistry
- Risedronate identification
- What is the mechanism of action of risedronate?
- What are the indications of risedronate?
- What are the contraindications of risedronate?
- Is risedronate the ‘best’ bisphosphonate?
- Risedronate pharmacokinetics
- How long risedronate stays in your system?
- Risedronate and upper GI adverse reactions
- Risedronate and metabolism disorders
- Risedronate and jaw osteonecrosis
- Risedronate and musculoskeletal pain
- Rsidernoante and atypical subtrochanteric and diaphyseal femoral fractures
- Renal impairment and risedronate use
- Glucocorticoid-Induced osteoporosis and risedronate
- Risedronate interactions with laboratory tests
- Risedronate side effects
- What if I overdose risedronate?
- What should you know before taking risedronate?
- What should you do if you have missed your dose?
- How to store and dispense the risedronate?
- What is the dosage and administration of risedronate?
- Can I take risedronate if I am pregnant?
- Can I take risedronate if I am breastfeeding?
- How safe is risedronate for children with osteogenesis imperfecta?
- Can I take risedronate if I have liver impairment?
- Can I take risedronate if I have kidney dysfunction?
- Do you need to decrease the dose of risedronate in elderly?
- Does risedronate cause infertility?
- Can risedronate cause cancer?
- What are some drugs that can interact with risedronate?
- Can I take risedronate with Aspirin?
What is risedronate?
Risedronate is a salt of a risedronic acid and belongs to the group of drugs called bisphosphonates. These drugs alter the cycle of bone formation and bone resorption in such a way that more bone is being formed that is removed.
Therefore the net effect is bone strengthening and increase in bone mass. This will reduce the incidence of pathological fractures.
Risedronate is given to individuals suffering from bone metabolism disorders such as osteoporosis, Paget’s disease of bone, etc.
It is produced and marketed by several companies such as Takeda in Japan, Warner Chilcott and Sanofi Aventis in the US and is marketed under the trade names Actonel, Atelvia and Benet.
Since calcium is an important supplement needed for bone formation, it is also available in a preparation that includes a calcium carbonate supplement. This particular preparation is marketed as ‘Actonel with Calcium’.
It is available in different strengths. These strengths are: 5 mg, 30 mg, 35 mg, 75 mg and 150 mg.
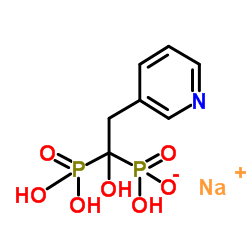
Risedronate Chemistry
IUPAC name: (1-hydroxy-1-phosphono-2-pyridin-3-ylethyl)phosphonic acid
Molecular formula: C7H11NO7P2
Molecular weight: 283.113 g/mol
Chemical structure:
Drug class: It is an organic compound belonging to the class of bisphosphonates. These compounds contain phosphonate groups linked together through a carbon atoms.
Risedronate identification
Risedronate sodium appears as white to off-white colored, fine crystalline powder which is odorless and soluble in water and in other aqueous solutions, but insoluble in common organic solvents.
What is the mechanism of action of risedronate?
Risedronate is a bisphosphonate which are structural analogues of inorganic pyrophosphate, a substance which is present in almost all body fluids and prevents soft tissue calcification. Thus it plays a regulatory role in bone mineralization process.
Normally bone deposition and bone resorption both are taking place and both are closely regulated by the body. This process is known as remodeling. However if due to any pathological process bone resorption is increased, then risedronate is given.
Risedronate has a strong antiresorptive action. It inhibits osteoclasts (cells responsible for bone resorption). The inhibition of osteoclasts is a result of the inhibition of the enzyme farnesyl diphosphate synthase (FPPS).
This enzyme is a pivotal element of mevalonate cascade that leads to isoprenoid lipids production.
These lipids are needed for post-translational modification of small GTP-binding proteins essential to osteoclast function. Therefore inhibition of this enzyme will lead to decreased osteoclastic function.
Risedronate also acts by attracting hydroxyapatite crystals (calcium crystals) at bone mineral matrix level. This will increase the amount of calcium deposition thereby increasing bone mineral density.
What are the indications of risedronate?
The indications of risedronate include:
- Treatment of osteoporosis
- Prevention of osteoporosis in high risk groups such as post-menopausal women (estrogen promotes bone formation, once menopause occurs there is no estrogen in the system) or those taking steroids (steroids cause bone breakdown)
- Paget’s disease of bone
Off-label uses of risedronate include:
- Metastatic disease of the bone
- Treatment of high calcium levels in the blood
What are the contraindications of risedronate?
Risedronate is contraindicated in the following conditions:
- Delayed esophageal emptying. Examples of such diseases include esophageal stricture, achalasia cardia etc.
- If the individual cannot stand or sit upright for at least 30 minutes
- Low calcium levels in blood. (Hypocalcemia)
- Allergy to risedronate or any other component of the pill
Is risedronate the ‘best’ bisphosphonate?
Current clinical guidelines put both risedronate and alendronate as the treatment of choice for osteoporosis as both reduce the risk of vertebral as well as non-vertebral fractures by over 50%.
However studies comparing these two drugs have not been carried out.
Risedronate pharmacokinetics
Absorption: After oral intake, it is absorbed in the upper GI tract. The peak serum concentration is achieved one hour after its administration and the absorption is same for a range of doses.
Steady state is achieved after 57 days of daily dosing. Mean absolute oral bioavailability is just 0.63%.
There is significant effect of food on its absorption. The summary of effect of food is given below and is compared with the absorption in fasting state (no food or drink for 10 hours prior to or 4 hours after dosing):
When risedronate is administered 0.5 hours before breakfast, its absorption is reduced by 55%. Similarly, when risedronate is administered 2 hours after dinner its absorption is reduced by 55%.
When risedronate is administered 1 hour before breakfast, its absorption is reduced by 30%
Distribution: the volume of distribution for risedronate is 13.8 L/kg in humans. At any given time, about 24% of the drug is bound to plasma proteins.
Preclinical studies showed that about 50-60% if the drug ends up in bone and the remaining was excreted in urine.
Distribution of risedronate to soft tissues is very low and ranges from 0.001% to 0.01% and this too after multiple doses have been taken.
Metabolism: There is no evidence of systemic metabolism of the drug despite multiple scientific studies been carried out to find it.
Excretion: Approximately 50% of the drug is excreted in the urine within 24 hours after administration and 85% of the drug is excreted in 28 days.
Mean renal clearance is 105 mL/min. The renal clearance in not dependent on concentration and there is a linear relationship between renal clearance and creatinine clearance.
The unabsorbed drug is eliminated unchanged in feces. The terminal exponential half-life of risedronate is 561 hours.
How long risedronate stays in your system?
The half-life of risedronate is approximately 23 days. Normally 5.5 half-lives are needed to completely clear the drug from the body. So 126 days would be needed to completely remove the drug from the body.
However much more research needs to be done to find this out as alendronate has been found to stay in the body for as much as 10 years. So there is potential for risedronate to stay in the body for years as well.
It is because of this reason that some clinicians will stop risedronate after 3 to 5 years of use as you will still be able to gain the benefits of the drug long after its cessation.
Risedronate and upper GI adverse reactions
All bisphosphonates are notorious for causing irritation of upper GI. This may result in the development of diseases such as esophageal ulcers, esophagitis, and esophageal erosions, which can cause bleeding and as well as esophageal strictures or perforation needing immediate hospitalization.
Similarly bisphosphonates can aggravate other medical conditions of the upper GI such as gastritis, duodenitis, ulcers or dysphagia to any cause.
Therefore patients should be advised to immediate seek medical attention if they start experiencing dysphagia, odynophagia, retrosternal pain or new or worsening heartburn.
Risedronate and metabolism disorders
Risedronate will take the calcium from the blood and deposit it in the bone therefore reducing the amount of calcium in the blood.
This might cause hypocalcemia. Therefore instruct the patients to take of their dietary intake of calcium and vitamin D or give supplements.
Risedronate and jaw osteonecrosis
Osteonecrosis of the jaw can occur while taking most bisphosphonates including risedronate. IT can after tooth extraction or local infection with delayed healing. In some cases it can occur spontaneously.
Risk factors for the development of osteonecrosis of the jaw include poor oral hygiene, invasive dental procedures such as dental implants, tooth extraction and boney surgery concomitant therapies (particularly notorious are steroids, chemotherapy, angiogenesis inhibitors), presence of cancer and co-morbid disorders such as a pre-existing dental disease, infection, anemia, coagulopathy, ill-fitting dentures.
Patients who develop osteonecrosis of the jaw need to consultant an oral surgeon right away and discontinuation of bisphosphonates can also be undertaken.
Risedronate and musculoskeletal pain
Bisphosphonates have been known to cause severe, intractable pain of the joints, bone or muscles which may even cause movement problems leading to incapacitation.
The onset of such type of pain varies from one day to several months after starting the drug.
It was seen that after stopping the drug, most patients were relieved of their pain however some did need additional pharmacological interventions. Restarting the drug or shifting to another drug of the same class might cause recurrence of these symptoms.
Rsidernoante and atypical subtrochanteric and diaphyseal femoral fractures
Fractures of femur with low energy trauma have been reported in individuals taking bisphosphonates. These fractures can occur anywhere in the femoral shaft and are short oblique or transverse without comminution.
Atypical fractures maybe unilateral or bilateral and present as dull, aching thigh pain, weeks to months before a complete fracture occurs. Undergoing steroid therapy will increase the patients risk to develop such fractures.
Ideally, any patient undergoing or having a history of undergoing bisphosphonate therapy and now experiencing pain in the thigh or groin region should be tested for femoral fracture.
If a diagnosis is made, then contralateral limb should also be checked for any symptoms and signs of fracture. Discontinuation of bisphosphonates should be considered after taking a risk/benefit assessment by the physician.
Renal impairment and risedronate use
Patients having a GFR of less than 30 ml/min should not be given bisphosphonates.
Glucocorticoid-Induced osteoporosis and risedronate
Status of sex hormones of both men and women should be checked (and appropriate replacement consider if the levels are low) before starting risedronate for the treatment and prevention of glucocorticoid-induced osteoporosis.
Risedronate interactions with laboratory tests
Bone imaging is vital in the field of radiology and maybe carried out by using specific bone imaging agents. Bisphosphonates interfere with them, although specific studies for risedronate have not been carried out.
Risedronate side effects
Risedronate can cause a number of side effects. Some of these mild side effects which usually do not require immediate medical attention and usually disappear as the body gets used to the drug
More common
- back pain
- cough
- hoarseness
- diarrhea
- fever with or without chills
- headache
- joint pain
- Pain or difficulty in urination
Less common
- Acidity and heartburn
- bladder pain
- bloody urine
- cloudy urine
- blurred vision
- change in visual acuity
- generalized or localized body pain
- constipation
- dizziness
- dry eyes
- soreness or dryness of the throat
- frequent urination
- anxiety
- leg cramps
- muscle stiffness
- nausea
- nervousness
- swelling and redness of joints
- ringing in the ears
- runny nose
- change in heart rate
- swelling of the feet extending up to calf
- tender swelling of the glands of the neck
- voice changes
- weakness
Rare
- Fainting
- Fear
- Skin Itch
- Anorexia
- Pale skin
- Bloating and flatulence
- Soreness of the tongue
- Sneezing
- Stomach fullness
- Tightness in the chest
- Troubled breathing
- Unusual bleeding or bruising
- Wheezing
The following side effects are due to a serious underlying condition. If they appear, consult your physician immediately
More common
- Abdominal pain
- skin rash
Less common
- belching
- bone pain
- stomach cramps
- difficulty in swallowing
Rare
- Red, sore eyes
What if I overdose risedronate?
The following are the symptoms of overdose:
- Confusion
- convulsions
- difficulty in breathing (dyspnea)
- irregular heartbeats
- Muscle cramps. Cramps can occur in either the small muscles of hands, feet or face or in the large muscles of limbs and trunks.
- numbnessor tingling around the mouth or in hands or feet
- Tremor
No specific guideline by a competent authority is available. However, these general steps can be taken for its management:
- Since risedronate causes hypocalcemia, signs and symptoms of hypocalcemia will be seen so calcium replacement will be needed.
- Milk or antacids containing calcium, aluminum or magnesium are given. These divalent cations bind with risedronate and reduce its absorption.
- In cases where a significant overdose is expected, gastric lavage may be done.
What should you know before taking risedronate?
- You should follow the dose prescribed by your doctor.
- Take risedronate with atleast one full glass of plain water while in sitting up or standing position to facilitate the delivery of the drug to stomach as soon as possible to avoid esophageal irritation. Take atleast 30 minutes before breakfast.
- Do not chew or suck on the tablet because of the risk of oropharyngeal irritation.
- Consult a physician if side effects appear.
- Take calcium and vitamin D supplements
- Life style modifications: cessation of smoking and alcohol, do regular excercises.
What should you do if you have missed your dose?
If you’re taking risedronate 35 mg once a week and if you have missed your dose, then take one tablet immediately if you’ve not had your breakfast and have breakfast 30 minutes later.
If you have had your breakfast, then take one tablet the next morning and then return to taking one tablet once-a-week, as originally scheduled on their chosen day. Never take two 35 mg tablets on the same day.
If you are taking risedronate 150 mg once a month, then check if your next month’s scheduled dose is more than 7 days away. If yes taken one tablet immediately if you’ve not had your breakfast and have breakfast 30 minutes later.
If you have had your breakfast, then take one tablet the next morning. Never take two 150 mg tablets within 7 days of each other.
How to store and dispense the risedronate?
Risedronate is stored at 20° to 25°C (68° to 77°F). Ideally it should be dispensed in a tight, light-resistant container with a child-resistant closure. It is not an over the counter drug and pharmacist should only dispense it after seeing the prescription.
What is the dosage and administration of risedronate?
Risedronate is available as an oral formulation only. The dose of risedronate depends upon the disease being treated. The recommended doses are:
- Treatment of postmenopausal osteoporosis: Daily one tablet 5 mg OR weekly one 35 mg tablet OR monthly 150 mg tablet.
- Prevention of postmenopausal osteoporosis: Daily one tablet 5 mg OR weekly one 35 mg tablet OR monthly 150 mg tablet.
- Treatment of osteoporosis in men: Weekly one tablet 35 mg
- Treatment and prevention of glucocorticoid osteoporosis: Daily one tablet 5 mg
- Treatment of Paget’s disease: Daily 30 mg for 2 months. 2 months after cessation of drug, the response to treatment is checked and if alkaline phosphatase fails to normalize re-treatment can be considered.
Can I take risedronate if I am pregnant?
Risedronate has been grouped as pregnancy category C drug. This means that there have been no adequate studies on the effect of risedronate on pregnancy in humans.
However, a theoretical risk to the human fetus remains because it was observed in animals that there was an increased likelihood of abortion and prematurity.
Therefore you should only start taking this drug if the benefits far outweigh the risks.
Can I take risedronate if I am breastfeeding?
There has been no study to identify the presence of risedronate in human breast milk, however, the drugs presence was identified in the milk of dogs indicating a small amount of lacteal transfer. Therefore this drug should only be taken if it is absolutely necessary for the mother.
How safe is risedronate for children with osteogenesis imperfecta?
The safety of risedronate has been studied in children. The two studies concluded that there was no decreased risk of fractures when risedronate was given to pediatric population with osteogenesis imperfecta. There was also an increased likelihood of developing the following side effects
- Vomiting
- Pain in the extremities
- Headache
- Back pain
- Upper abdominal pain
- Bone pain
Can I take risedronate if I have liver impairment?
Risedronate is not metabolized by the liver. So even if you have liver dysfunction, dose adjustment is not required and you can continue to use the drug as long as the underlying problem is not treated.
Can I take risedronate if I have kidney dysfunction?
The main form of excretion of the drug is through the kidneys. However, since the amount of drug that is excreted in the urine per hour is very small, mild to moderate kidney dysfunction would have no effect on the excretion of the drug.
However if there is sever kidney dysfunction, characterized by creatinine clearance less than 30 ml/min, then dose adjustment is necessary.
Do you need to decrease the dose of risedronate in elderly?
A significant proportion of the patients with bone metabolism are above 65 years of age. At this stage due to aging, the human physiology is altered.
Therefore a number of studies have been carried out to check for the efficacy and safety of risedronate in elder population.
In this regard a research carried out on elderly male patients of osteoporosis showed that there might be increased sensitivity of the drug in such population. Therefore there might be a need to decrease the dose of risedronate in some patients.
Does risedronate cause infertility?
Rats and dogs were exposed to doses 5 times the recommended dose for humans. It was found that at this high dose, females suffered from ovulation problems while males underwent testicular atrophy.
There is no such study on humans, however from this information it can be seen that risedronate will only cause infertility if it is taken at a very high dose for quite some time.
Can risedronate cause cancer?
High dose risedronate (more than 5 times the dose in humans) was given to mice to check for teratogenicity. The study was conducted over 104 weeks and even then there were no significant findings of drug induced tumor in mice.
Similarly, there was no evidence of chromosomal damage in mice exposed to such high dose of risedronate.
What are some drugs that can interact with risedronate?
The following drugs interact with risedronate
- Antacids/calcium supplements: Any medication containing divalent ions will bind with risedronate and decrease its absorption
- Hormone replacement therapy: Concomitant use of estrogen along with risedronate might prove to be beneficial in increasing bone mineral density.
- Nonsteroidal anti-inflammatory drugs: Combined use of aspirin and other NSAIDS will increase the likelihood of developing upper GI discomfort particularly GERD and ulcers.
- Taking proton pump inhibitors or H2 blockers does not reduce the risk of developing of ulcers. Apart from this, there is an increased likelihood of developing asthma if you are taking aspirin and risedronate together.
Can I take risedronate with Aspirin?
In theory, there is a concern and increased risk and severity of GI toxicity during concomitant use of oral bisphosphonates and NSAIDs due to additive or synergistic effects on the GI mucosa.
Because NSAIDs lower the rate of stomach and duodenum ulcer healing, it is also possible that NSAIDs may postpone healing and promote mucosal injury caused by oral bisphosphonates.
“Does Lansoprazole cause weight gain? What are the side effects of taking Lansoprazole?“