Contents
- What is Sumatriptan?
- Sumatriptan molecular structure, formula, drug class, weight
- Why is sumatriptan prescribed?
- Sumatriptan brand names
- What are sumatriptan FDA approved indications?
- How does Sumatriptan work in the body?
- How must Sumatriptan be taken?
- Sumatriptan dosage for different indications
- What happens if a dose of sumatriptan is missed?
- Sumatriptan overdose
- Sumatriptan side effects
- For how long Sumatriptan stay in the body?
- Can Sumatriptan be used in pregnancy?
- Can Sumatriptan be used in nursing mothers?
- Can Sumatriptan be used in pediatric?
- Can Sumatriptan be used in old patients?
- Can Sumatriptan be used in patients with hepatic impairment?
- Can Sumatriptan cause Ischemia, Prinzmetal’s Angina and Myocardial Infarction?
- Can Sumatriptan cause arrhythmias?
- Can Sumatriptan cause angina?
- Can Sumatriptan cause cerebrovascular events?
- Can Sumatriptan cause vasospasm reactions?
- Can Sumatriptan cause serotonin syndrome?
- Can Sumatriptan cause increase in blood pressure?
- Can Sumatriptan cause anaphylactic reactions?
- Can Sumatriptan cause seizures?
- Can Sumatriptan can be taken with Duloxetine?
- Can Sumatriptan can be taken with Tramadol?
- Can Sumatriptan can be taken with Trazodone?
- Can Sumatriptan can be taken with Ondansetron?
- Can Sumatriptan can be taken with Sertraline?
What is Sumatriptan?
Sumatriptan is a generic name for a prescription drug that is used for the treatment of migraine headaches. It is most effective when it is taken as soon as the headache pain begins – not before, even if there are warning signs like aura (sensory or visual disturbances). It’s for migraine with or without aura. It works by narrowing blood vessels in the brain stem and reducing factors that can trigger nausea, light sensitivity, and other migraine symptoms. FDA first approved sumatriptan in 1992.
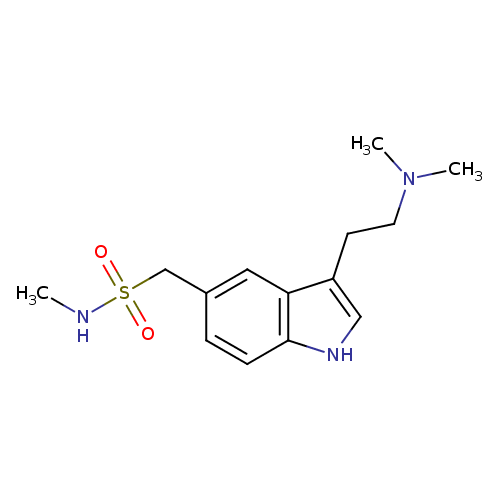
Sumatriptan molecular structure, formula, drug class, weight
Sumatriptan molecular structure
Molecular weight– 295.401 g/mol
Chemical or Molecular Formula– C14H21N3O2S
Sumatriptan pharmacological class– serotonin 5-HT1 receptor agonist
This compound belongs to the class of chemical entities known as tryptamines and derivatives. These are compounds containing the tryptamine backbone, which is structurally characterized by an indole ring substituted at the 3-position by an ethanamine.
Why is sumatriptan prescribed?
It is used to treat the migraine headache. It is especially effective in treating migraine aura symptoms such as wavy lines, flashing lights, and dark spots. It is also used for treating cluster headaches.
Sumatriptan brand names
Most common brand names for sumatriptan on the market are: Alsuma ,Imitrex, Sumavel Dose Pro, Imitrex STAT dose System.
What are sumatriptan FDA approved indications?
It is used for the treatment of the migraine headache and cluster headache.
How does Sumatriptan work in the body?
It binds to 5-HT1B/1D receptors. It exerts its therapeutic effects in treating migraine headache by the agonist effects at 5-HT1B/1D receptors. These receptors are present on sensory nerves of the trigeminal system and intracranial blood vessels. It results in the constriction of cranial vessel and inhibition of the release of the pro-inflammatory neuropeptide.
It has no significant affinity or pharmacological activity at 5-HT2, 5-HT3 receptor subtypes or at alpha1-, alpha2-, or beta-adrenergic; dopamine1; dopamine2; muscarinic; or benzodiazepine receptors.
How must Sumatriptan be taken?
You must use Sumatriptan as per directions given by your doctor. You must check the label for exact instructions of dosing.
- There comes an extra patient leaflet with Sumatriptan. You can talk to your pharmacist if you have any queries.
- Your doctor will teach you to how to use Sumatriptan. You must understand that how the Sumatriptan must be used. Kindly follow the procedures to take the dose when you use this drug. Contact your doctor if you have any queries.
- If you are prescribed for tablets, then take a dose of 50 mg or 100 mg tablet with a glass of water. You can take it as the headache starts. If the symptoms of your migraine improve and then returns back, you can take a second dose. But remember the dose of the gap must be at least two hoursafter the administration of the initial dose.
- If you are using nasal spray then use it as soon as your headache develops. If your migraine get improves but then return back, use the spray again. There must be the gap of at least two hoursafter the first dose.
- Not to use Sumatriptan if it is cloudy, contains particles, or discolored. If the device is damaged or cracked, don’t use it. Also, do not use it in the case tip of the device is broken or tilted.
- You must use sumatriptan only on a dry and clean area around the skin of your stomach and thigh. Never use Sumatriptan on arms and other areas of your body. Never use it on moles, scars and within two inches of your navel. Use sumatriptan on bare skin.
- Sumatriptan relieves migraine pain during an attack. It is better to inject sumatriptan dose as soon as you experience the symptoms of the migraine attack.
- If the symptoms of your migraine go away and return back, you can use a second dose as per the direction of your doctor. You must wait for at least an hour between the two doses. If you require the second dose of take it in a different area of skin.
- If the symptoms do not go away after the first injection, don’t use the second dose for the same attack. Consult your doctor.
- Not to use more than two doses in a day or take the dose of injection of more than four headaches within one month time. Check with your doctor and take the consultation from him.
- Keep this product away from the reach of children. Consult your doctor how to dispose of Sumatriptan after using it.
Sumatriptan dosage for different indications
Adult Dose for the Migraine
Oral– Initial dose: 100 mg, 50 mg or 25 mg orally once in a day.
Maximum dose: 200 mg in a day.
If the response to the first dosage occurs then the second dose can be taken at least 2 hours after the first dose if required. Higher Oral doses of 100 and 50 mg may provide a greater effect than 25 mg.
Subcutaneous– The Initial dose is 1 – 6 mg subcutaneously only once
Maximum dose: 12 mg in a day
If the response to the first dosage occurs, then the second dose can be taken at least 1 hour after the first dose if required.
Intranasal– Nasal spray: Initial dose: 20 mg, 10 mg, 5 mg into a nostril once
Maximum dose: 40 mg in a day
You must take 5 and 20 mg dosage as a single spray in one nostril. The10 mg dosage can be taken as 5 mg into each nostril. If the response to the first dosage occurs, then the second dose may be taken at least two hours after the first dose if required.
Nasal capsule/powder: Initial dose: 11 mg can be taken into each nostril once
Maximum dose: 2 doses (44 mg/4 nosepieces) in a day.
If the symptoms are not resolved in 2 hours or return then the second dose may be taken at least 2 hours after first dose.
Special Precautions:
-The treatment must be started at signs of the migraine headache, or other associated symptoms like vomiting, nausea, and photophobia.
-The higher doses have a greater effect, but carry a greater risk of adverse reactions.
-The efficiency of the treatment of more than four headaches in a day has not been established.
It is used for the treatment of the migraine with or without aura.
Adult dose for the Cluster Headache
Subcutaneous injection- Initial dose: 6 mg subcutaneously, once in a day.
Maximum dose: 12 mg in a day.
If symptoms recur, the dose can be repeated at least an hour after the first dose.
Special Precaution:
-The treatment must be started at the sign of the cluster headache or associated symptoms like vomiting, nausea and photophobia.
-The efficiency of the treatment of more than four headaches in a day has not been established.
Liver Dose Adjustments
In Mild to moderate liver dysfunction
Oral tablets: The Maximum single dose must not be exceeded beyond 50 mg.
Subcutaneous injection: No adjustment required
Severe liver dysfunction: it is contraindicated
Dose Adjustments
For Elderly
It must start at the lower dose due to greater frequency of renal, hepatic and cardiac impairment.
For patients with subcutaneous injections
If the migraine returns after the first dose of subcutaneous injection then another oral dose can be given to 100 mg in a day. It must be given least two hour time interval between two doses.
What happens if a dose of sumatriptan is missed?
In case you have missed a dose of sumatriptan and still have a headache then follow doctor’s instructions. Consult your doctor if you don’t know what to do in the case of missing a dose. Do not take an overdose of sumatriptan more often than it is prescribed. Do not take two doses at once.
Sumatriptan overdose
In clinical trials, headache suffers who got single doses of 140 to 300 mg did not have adverse reactions. Volunteers who took up to 400 mg did not suffer adverse reactions. Even so, doctors advise not taking more than 200 mg of sumatriptan in a 24-hour period and only using it as directed by your doctor.
If you think you have taken too much sumatriptan, call your doctor right away or go to the closest emergency room.
Sumatriptan side effects
Various side effects are as follows:
- The feeling of tightness and pain in your neck, jaw and throat.
- Chest pain or heavy feeling. The pain spreads to the shoulder or arm.
- Nausea, ill feeling and sweating.
- Weakness or sudden numbness especially on a side of your body
- A severe headache, confusion, speech, problems with vision, and balance.
- Severe pain in stomach and bloody diarrhea.
- Convulsions
- Blue-color appearance in toes or fingers.
- Numbness or tingling
- A mild headache but not a migraine
- Feeling cold or hot
- Heavy feeling in your body
- Drowsiness, spinning sensation and dizziness.
- Vomiting or nausea
- You can have unusual taste in mouth after administration of the nasal spray.
- Redness or warmth
You must tell your doctor if you are facing any of the following medical conditions.
- If you are pregnant, nursing mother or planning to conceive.
- If you are taking any medicine, dietary or herbal supplement
- If you are allergic to foods, and medicines.
- History of kidney or liver problems, heart problems or seizures.
- If you have chest pain, shortness of breath, high blood pressure, high cholesterol, stroke, heart attack, diabetes, or smoking
- Post -menopausal Woman
- If you are a man of more than 40 years of age.
- If you are overweight
For how long Sumatriptan stay in the body?
- It gest absorbed by 15%. The Volume of distribution is 7 L/kg. The Protein binding is14%-21%.
- Metabolism- It gets metabolized by Liver. Sumatriptan gets metabolized by monoamine oxidase (MAO), predominantly it is an isoenzyme. The Half- life- 2.5 hours.
- Route of elimination- 3% is excreted in the urine as unchanged drug. 42% of the dose is excreted as the major metabolite. The Clearance- 1200 mL/min
Can Sumatriptan be used in pregnancy?
Teratogenic Effects- There are not well-controlled trials and sufficient data in pregnant women. In the toxicity studies in pregnant rabbits and rat was associated with fetal abnormalities, embryo lethality, and pup mortality. When it is given intravenously, it was embryo lethal in pregnant rabbits. Sumatriptan tablets must not be taken in pregnancy.
The oral administration to pregnant rats during organogenesis resulted in an increased fetal blood vessel that includes umbilical and cervico thoracic abnormalities. The dose for embryo fetal toxicity in rats was 60 mg/kg/day. The oral administration in the pregnant rabbits also resulted in fetal cervico thoracic vascular, embryo lethality and skeletal abnormalities. The Intravenous administration to pregnant rabbits resulted in the increased incidence of embryo lethality.
Can Sumatriptan be used in nursing mothers?
It gets excreted in human milk. Avoid breastfeeding till twelve hours of the administering of Sumatriptan. The infant exposure can be minimized by avoiding breastfeeding for 12 hours after treatment with Sumatriptan tablets.
Can Sumatriptan be used in pediatric?
The safety and effectiveness in pediatric patients have not been established. These tablets are not advised to patients having less than 18 years of age. The analysis for nasal spray (5 to 20 mg) was carried on 1,248 adolescent having the migraine of age 12 to 17 years. The trials were not able to establish the efficacy of Sumatriptan nasal spray.
Five controlled clinical trials were performed to evaluate the efficacy of oral Sumatriptan 25 to 100 mg in patients of age 12 to 17 years. These trials were not able to establish the efficacy of oral Sumatriptan. The adverse reactions were observed in these clinical trials.
The various documents in post marketing ha stated various adverse reactions in the pediatric population. It was seen after the administration of Sumatriptan through intranasal, subcutaneous, and oral route. The loss of vision, stroke, myocardial infarction and even death was also reported.
Can Sumatriptan be used in old patients?
The Clinical trials of Sumatriptan tablets were not having sufficient numbers of patients of age 65 and older. The administration of Sumatriptan for an elderly patient must be cautious. The dose must be started at the low dose.
The patient must be under observation in the case of decreased renal, hepatic, or cardiac function. It is advised for the evaluation of heart in the old patient. Also, the patients who are at the risk of smoking, hypertension, diabetes, and the family history of CAD before the administration of Sumatriptan.
Can Sumatriptan be used in patients with hepatic impairment?
The maximum dose in patients with hepatic impairment must not exceed beyond 50 mg. This medicine is contraindicated in patients suffering from severe hepatic impairment.
Can Sumatriptan cause Ischemia, Prinzmetal’s Angina and Myocardial Infarction?
The administration of Sumatriptan tablets is contraindicated in patients suffering from vasospastic CAD or ischemic. There also have been reports of serious cardiac reactions that include myocardial infarction. It can occur within few hours after the administration of Sumatriptan tablets. Some of the adverse reaction were seen in patients without CAD. This drug can cause Prinzmetal’s angina. It occurs in the patients without a history of CAD.
The doctor must ask for the evaluation of cardiovascular risks like increased age, hypertension, diabetes, smoking, family history of CAD, and obesity before the administration of Sumatriptan tablets. In case of evidence of coronary artery vasospasm or CAD or, Sumatriptan tablets are contraindicated. The patients who are having multiple cardiovascular risk factors are advised for an ECG after the first dose of Sumatriptan tablets.
Can Sumatriptan cause arrhythmias?
Many life-threatening disturbances like ventricular tachycardia and fibrillation can lead to death. These have been reported after a few hours of the administration of Sumatriptan. You must discontinue Sumatriptan if these symptoms occur. These tablets are contraindicated in patients suffering from arrhythmias or Wolff-Parkinson-White syndrome.
Can Sumatriptan cause angina?
Heaviness in the precordium, sensations of tightness, pressure, pain in throat, neck, and jaw occur after taking Sumatriptan tablets. You must go for an ECG if the patients are at high cardiac risk. The administration of Sumatriptan is contraindicated in patients suffering from Prinzmetal’s variant angina and CAD.
Can Sumatriptan cause cerebrovascular events?
The patients who were treated with Sumatriptan can suffer from the cerebral hemorrhage, subarachnoid hemorrhage, and stroke. The patients with the migraine are at the high risk of the cerebrovascular events like stroke, TIA, and hemorrhage. You must discontinue the drug if cerebrovascular event occurs. Sumatriptan is contraindicated in the patients who have a history of stroke or TIA.
Can Sumatriptan cause vasospasm reactions?
Sumatriptan may cause non-coronary vasospastic reactions like gastrointestinal vascular ischemia, peripheral vascular ischemia, splenic infarction, and Raynaud’s syndrome. The patients who have the signs of non-coronary vasospasm reaction after the use of Sumatriptan that rules out the vasospastic reaction. The report of permanent blindness or partial vision loss is reported after the use of Sumatriptan.
Can Sumatriptan cause serotonin syndrome?
The serotonin syndrome can occur while taking Sumatriptan with SSRIs, serotonin norepinephrine reuptake inhibitors, MAO inhibitors and tricyclic antidepressants. The symptoms are changes in mental status like hallucinations, agitation, and coma. It can also cause autonomic instability like tachycardia and hyperthermia.
You can also face neuromuscular problems like coordination and hyperreflexia. The gastrointestinal problems like nausea, vomiting and diarrhea. The onset of symptoms occurs within minutes of receiving Sumatriptan. You must discontinue this medicine in the case of Serotonin syndrome.
Can Sumatriptan cause increase in blood pressure?
There can be an increase in blood pressure. It can also cause hypertension with acute impairment of organ systems in the patients who are given this drug. You must monitor blood pressure in patients who are treated with Sumatriptan. These tablets are contraindicated in the patients suffering from uncontrolled hypertension.
Can Sumatriptan cause anaphylactic reactions?
Anaphylactic reactions can occur in patients that are receiving Sumatriptan. These reactions can be fatal or life threatening. Anaphylactic reactions occur in individuals with the history of sensitivity to allergens. Sumatriptan is contraindicated in patients that have a history of hypersensitivity reaction to this drug.
Can Sumatriptan cause seizures?
The seizures are reported after the administration of Sumatriptan. It can occur in patients with the history of seizures or predispose to seizures. Sumatriptan must be used with caution in patients with the history of epilepsy or conditions associated with it.
Can Sumatriptan can be taken with Duloxetine?
The co-administration of Sumatriptan and Duloxetine can increase the risk of serotonin syndrome. It is a rare yet serious fatal condition that can be trigger from hyper stimulation of 5-HT1A and 2A receptors. The symptoms of the serotonin syndrome are changes in mental status like irritability, confusion, altered consciousness, coma and hallucination.
It also results in autonomic dysfunction like hyperthermia, tachycardia, shivering, and blood pressure. Neuromuscular issues like tremor, rigidity, and ataxia. Gastro-intestinal issues like nausea, abdominal cramping, vomiting, and diarrhea. It can be managed by avoiding the use of multiple serotonergic agents together. The patient must be monitored for symptoms of serotonin syndrome.
Particularly the caution must be taken while increasing the dose of these agents. If the serotonin syndrome gets developed or is suspected all serotonergic agents must be discontinued immediately.
Can Sumatriptan can be taken with Tramadol?
The co-administration of both these agents. It increases the risk of serotonin syndrome. The doctor must have a close monitoring of the symptoms of Serotonin syndrome. It is a rare but can be serious and fatal. It can be managed by avoiding administration of two drug together.
The patient must be closely under monitor when the doses are changed. You must take these both two doctors under the doctor’s guidance. If the serotonin syndrome is expected or gets develop, you must discontinue these both two drugs immediately. The serious cases must be managed under toxicologist.
Can Sumatriptan can be taken with Trazodone?
Using these two drugs together increases the risk of serotonin syndrome. The symptoms are a hallucination, fluctuations in blood pressure, confusion, seizure, increased heart rate, excessive sweating, shivering, blurred vision, stiffness, tremor, and incoordination.
The other symptoms are diarrhea, nausea, stomach cramp, and vomiting. The serious cases may result in death or coma. You must seek the advice of the doctor if you experience these symptoms.
Can Sumatriptan can be taken with Ondansetron?
The simultaneous administration of both these drugs results in the risk of increases the risk of serotonin syndrome. You must take both these drugs under the advice of the doctor. Your doctor will guide you how to take these medicines. He knows about the risk factors associated with the simultaneous administration of these drugs and how to tackle them.
Can Sumatriptan can be taken with Sertraline?
This combination must be taken under the care of the doctor. This combination increases the risk of serotonin syndrome. This condition can be fatal. So, you must take these medicines under the guidance of the doctor. Your doctor will closely monitor the symptoms of serotonin syndrome.
“What is aripiprazole (Abilify) 5 mg used for? What does aripiprazole do to the brain?“