Contents
- What is Azithromycin
- Azithromycin generic and brand name
- What is azithromycin used for
- Pharmacophore structure: Information about the chemical structure of the drug
- Chemical information of the drug
- Azithromycin available strength
- How does azithromycin work (mode of action)
- What are the recommended doses of Azithromycin
- When should I discontinue, withhold or modify the dose of Azithromycin
- What are the pharmacokinetic properties of the drug
- Which pregnancy category (A; B; C; D; X) has been assigned to Azithromycin
- How to use azithromycin
- How to store the drug
- How to dispose the medicine
- Does Azithromycin has approval from government / FDA /or any other related agencies
- Other uses of the drug
- What special dietary precautions should I follow
- What special precautions should I follow/ What should I avoid while using Azithromycin
- Azithromycin side effects
- What should I do in case of overdose
- What should I do in case of missed a dose
- Azithromycin drug interactions
- Does Azithromycin have any interaction with diseases
- Where can I get more information
- Clinical research and current scenario of the drug
- References from chemical, biological and toxicological databases
What is Azithromycin
Azithromycin is an antibiotic that is used for the treatment of variety of bacterial infections such as skin, ear, respiratory infections and sexually transmitted diseases (STDs).
Azithromycin generic and brand name
- The drug is available under generic name Azithromycin and marketed under the various brand names such as Zithromax, Azithrocin and
- Azithromycin was originally developed by Croatian pharmaceutical company Pliva in 1980.
- Initially Pfizer was accountable for the manufacturing and marketing of Azithromycin under the licence of Pliva.
- Azithromycin is also manufactured by Pliva and marketed by Teva pharmaceuticals.
- Furthermore, Azithromycin is marketed as an ophthalmic solution under the brand name AzaSite by Inspire Pharmaceuticals (a subsidiary of Merck & Co.).
What is the source of the drug (natural or synthetic)
- Azithromycin is a semi-synthetic, relatively broad spectrum, macrolide
What is azithromycin used for
- Azithromycin plays a key role in the treatment of complication related to the bacterial infections such bronchitis, pneumonia, skin infections and infections of ear and throat.
- It is also recommended to treat the sexually transmitted diseases and Urinary tract infections.
- It is also used to prevent or treat the disseminated Mycobacterium avium complex (MAC) infection.
- Azithromycin (in form of ophthalmic solution) is used to treat bacterial conjunctivitis (a bacterial eye infection).
- However, this drug does not provide any protection against flu, cold and other viral infections.
- Azithromycin is also prescribed for the treatment of acute bacterial sinusitis caused by influenza, M. catarrhalis or S. pneumonia.
- Sometimes it is also used to treat the infection of pylori and Legionnaires’ disease (inflammatory lung infection caused by Legionella).
- It is also prescribed for the treatment of Whooping cough.
Pharmacophore structure: Information about the chemical structure of the drug
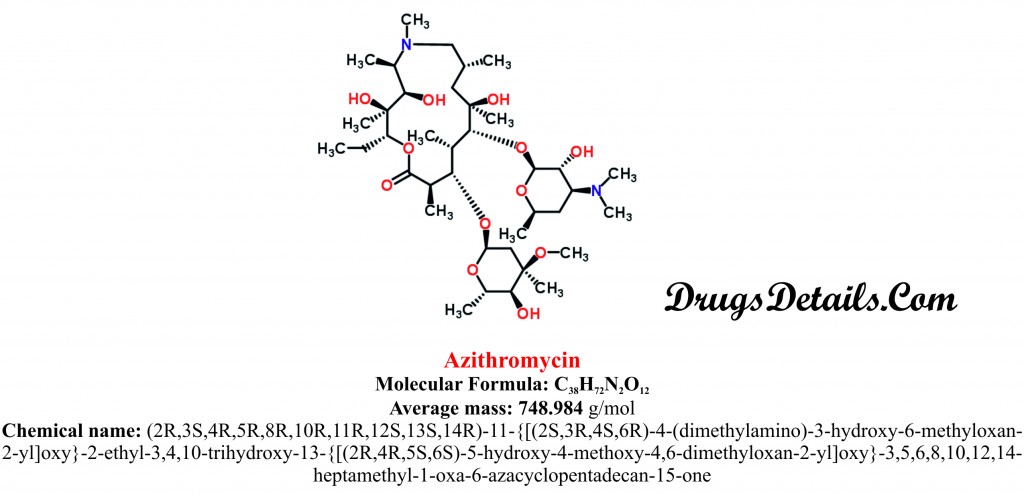
Azithromycin chemically belongs to the class of organic compounds which are known as Macrolides and analogues characterized by lactone ring of at least twelve members. The detailed chemical classification of Azithromycin is described below:
| Kingdom | Organic compounds |
| Super Class | Phenylpropanoids and polyketides |
| Class | Macrolides and analogues |
| Sub Class | Not available |
| Direct Parent | Macrolides and analogues |
Chemical information of the drug
- Azithromycin is a derivative of erythromycin and available as a dihydrate salt.
- It is a semi-synthetic aliphatic heteromonocyclic antimicrobial agent with a molecular formula C38H72N2O12.
- The molecular weight of the compound is 984 g/mol.
- Azithromycin is a dihydrate salt of (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-{[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy}-2-ethyl-3,4,10-trihydroxy-13-{[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy}-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-azacyclopentadecan-15-one.
- Azithromycin is white crystalline powder and has a water solubility of 514 mg/mL.
- Azithromycin is freely soluble in methanol, methylene chloride, ethanol, and chloroform.
- The melting point of Azithromycin is 114°C.
Azithromycin available strength
- Azithromycin is available in tablet and oral suspension form for oral administration as well as in the form of ophthalmic solution.
- Azithromycin tablets are available in two different dosages of 250 mg and 500 mg.
- Azithromycin tablets of 250 and 500 mg are pink in color, capsule shaped, engraved, and film-coated.
- 250 mg tablets are engraved with “Pfizer” on one side and “306” on another side.
- 500 mg tablets are engraved with “Pfizer” on one side and “ZTM500” on another side.
- Azithromycin tablets contains azithromycin dihydrate as active ingredient and pregelatinized starch, lactose, dibasic calcium phosphate anhydrous, hypromellose, sodium croscarmellose, magnesium stearate, sodium lauryl sulfate, titanium dioxide, triacetin and D&C Red No. 30 Aluminum Lake as inactive ingredients.
- Azithromycin oral suspensions are available in two different dosages of 100 mg/5ml and 200 mg/5ml.
- Azithromycin oral suspension contains azithromycin dihydrate as active ingredient and sucrose, sodium phosphate, tribasic, anhydrous; hydroxypropyl cellulose, xanthan gum, spray dried artificial cherry, vanilla or banana flavours and FD&C Red No. 40 as inactive ingredients.
- Azithromycin ophthalmic solution (AzaSite®) is available as 1% sterile solution which is an off-white viscous liquid.
- It contains citric acid, sodium citrate, poloxamer 407, polycarbophil, mannitol, sodium chloride, edentate disodium and water as inactive ingredients. Furthermore, it also contains 0.003% benzalkonium chloride as preservative agent and sodium hydroxide to adjust pH (6.3).
How does azithromycin work (mode of action)
- Azithromycin is effective against gram positive bacteria such as Staphylococcus aureus, S. agalactiae, S. Pneumonia and pyogenes, gram-negative bacteria such as Haemophilus ducreyi, H. influenza, Moraxella catarrhalis and Neisseria gonorrhoeae and some other bacteria like Chlamydophila pneumonia, Chlamydia trachomatis and Mycoplasma pneumonia.
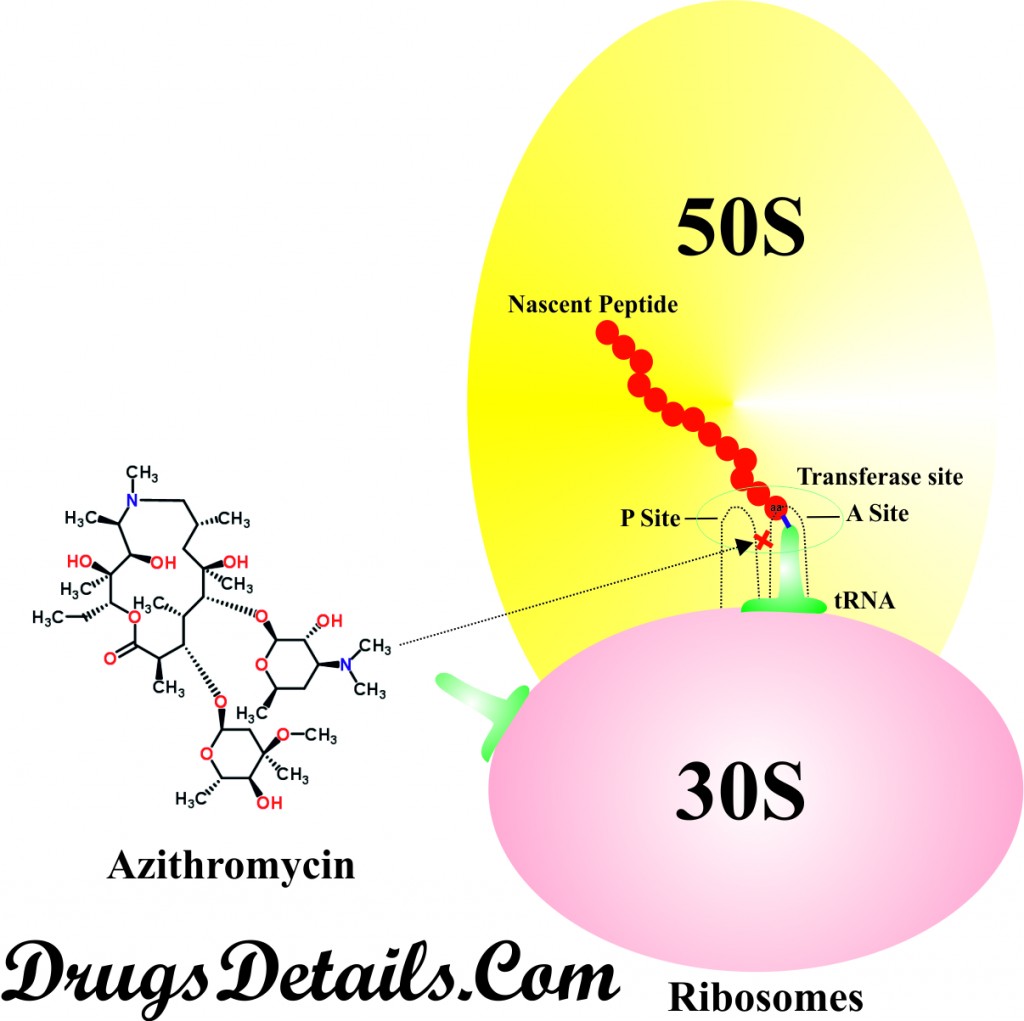
- Azithromycin is a macrolides antibiotic (such as Erythromycin and Carbomycin) which acts by inhibiting the bacterial protein synthesis, quorum-sensing and formation of bacterial biofilm.
- Macrolides primarily act through enhancing the dissociation of peptidyl-tRNA from ribosome, thus inhibiting transpeptidation and translocation step of protein synthesis (an intermediate steps of protein synthesis).
- It has been observed that Azithromycin binds with the 50S subunit of ribosome and interferes with polypeptide synthesis. Therefore, it is predicted that stimulated dissociation of peptidyl-tRNA from ribosome is the major mechanism of action of Azithromycin.
- Azithromycin competes for [14C] erythromycin ribosome binding site, suggesting that it binds to the same receptor as erythromycin. However, it does not affect the synthesis of nucleic acid.
- The Azithromycin accumulates at very higher concentration in cells, predominantly in phagocytes and therefore delivered effectively in high concentrations to sites of infection.
What are the recommended doses of Azithromycin
The dose of Azithromycin varies depending upon the age and diseased state of the patient.
Adult dose
- Acute pelvic inflammatory disease
- Initial dose: 500 mg once intravenously for 1 or 2 days.
- Maintenance day: 250 mg once in a day orally for 7 days.
- Babesiosis
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day in combination with Atovaquone orally for 7-10 days.
- Bacterial endocarditis: 500 mg as a single dose orally 1 hour previous to process.
- Bacterial sinusitis: 500 mg once in a day orally for 3 days.
- Bronchitis
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- Cervicitis: 1000 mg as a single dose orally.
- Chlamydia infection: 1000 mg as a single dose orally.
- Chronic obstructive pulmonary disease: 500 mg once orally for 3 days.
- Community acquired pneumonia
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- Erythema chronicum migrans: 500 mg once in a day orally for 7-10 days.
- Genital ulcer disease (Chancroid): 1000 mg as a single dose orally.
- Granuloma inguinale: 1000 mg orally once weekly for 3 weeks or more.
- Mycobacterium avium complex infection
- Prophylaxis: 1200 mg once in a week orally.
- Treatment: 600 mg once in a day orally with Ethambutol.
- Nongonococcal urethritis: 1000 mg as a single dose orally.
- Otitis media
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- Pertussis
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- Skin infections
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- STD prophylaxis: 1000 mg as a single dose orally.
- Tonsillitis/Pharyngitis
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
- Toxoplasmosis: 900-1200 mg once a day orally for 6 weeks or more.
- Typhoid fever: 1000 mg once in a day orally for 5 days.
- Uncomplicated Gonococcal infection: 2000 mg as a single dose for Gonococcal urethritis and cervicitis orally.
- Upper respiratory tract infection
- Initial dose: 500 mg once orally for a day.
- Maintenance day: 250 mg once in a day orally for 4 days.
Paediatric dose
- Acute bacterial sinusitis
- ≥ 6 months: 10 mg/kg daily for 3 days orally.
- ≥ 16 months: 500 mg daily for 3 days orally.
- Bacterial endocarditis prophylaxis: 15 mg/kg as a single dose orally 1 hour previous to process.
- Chlamydia trachomatis infection
- ≥ 16 months: single dose of 1000 mg orally.
- Chronic obstructive pulmonary disease
- ≥ 16 months: 500 mg orally for initial day and then 250 mg once daily for 4 days orally or 500 mg daily for 3 days orally.
- Community acquired pneumonia
- ≥ 6 months: 10 mg/kg orally for initial day and then 5 mg/kg once daily for 4 days orally.
- ≥ 16 months: 500 mg orally for initial day and then 250 mg once daily for 4 days orally.
- Cystic fibrosis
- ≥ 16 months
- 25 – <40 kg: 250 mg thrice in a week at regular interval (one dose in 48 hours).
- ≥ 40 kg: 500 mg thrice in a week at regular interval (one dose in 48 hours).
- Erythema chronicum migrans: 10 mg/kg daily for 7-10 days orally.
- Gonococcal infection
- ≥ 16 months: single dose of 2000 mg orally.
- Mycobacterium aviam complex infection
- Prophylaxis:
- ≥ 16 months: 1200 mg once in a week orally.
- Treatment
- ≥ 16 months: 600 mg once in a day orally with Ethambutol.
- Nongonococcal infection
- ≥ 16 months: single dose of 1000 mg orally.
- Otitis media
- ≥ 6 months: 30 mg/kg as a single oral dose or 10 mg/mg once a day orally for 3 days.
- Pelvic inflammatory disease
- ≥ 16 months: 500 mg intravenously daily for 1-2 days and then 250 mg in a day for 7 days orally.
- Pertussis
- <5 months: 10 mg/kg daily for 5 days orally.
- 1-5 months: 10 mg/kg daily for 5 days orally.
- ≥ 6 months: 10 mg/kg orally for initial day (not more than 500 mg) and then 5 mg/kg once daily (not more than 250 mg) for 4 days orally.
- Skin infection
- ≥ 16 months: 500 mg orally for initial day and then 250 mg once daily for 4 days orally.
- Tonsillitis/Pharyngitis
- ≥ 2 months: 12 mg/kg daily for 5 days orally.
- ≥ 16 months: 500 mg orally for initial day and then 250 mg once daily for 4 days orally.
- Typhoid fever
- ≥ 3 months: 20 mg/kg daily for 5-7 days orally.
- Upper respiratory tract infection
- ≥ 6 months: 10 mg/kg orally for initial day and then 5 mg/kg once daily for 4 days orally.
- Renal dose adjustments: Caution should be exercised in patients with GFR <10 ml/min.
- Hepatic dose adjustments: Caution should be exercised in patients with liver dysfunction.
When should I discontinue, withhold or modify the dose of Azithromycin
- The usual dosing of the drug may vary depending upon the efficiency and side effects of the drug in a particular individual.
- Do not use the medicine if you are hypersensitive or allergic to Azithromycin or any other antibiotic such as Clarithromycin, Dirithromycin and Erythromycin.
- No dosage adjustment is required in case of renal impairment and patient with hepatic impairment.
- Use of ergot derivatives with Azithromycin should be contraindicated because ergot derivatives may be precipitated by Azithromycin.
- Caution should be exercised with the use of Azithromycin oral solution in diabetic patients because it contains sucrose.
- Azithromycin should not be used along with antacids that contain aluminium or magnesium hydroxide because antacids bind to the Azithromycin and prevent the absorption in intestine. Consult to your pharmacist for the use of antacids with Azithromycin.
- Azithromycin is not recommended for those patients who have Niemann-Pick or Tay-Sachs disease.
What are the pharmacokinetic properties of the drug
- Pharmacokinetic studies suggest that after oral administration Azithromycin is rapidly absorbed and has a bio-availability of approximately 37%.
- Following absorption approximately 51% (at 02mcg/ml concentration) to 7% (at 2mcg/ml concentration) of the drug is bound to serum proteins.
- It has been observed that following a 500 mg dose of maximum (or peak) plasma concentration (35 to 0.45mg/L) is achieved in 120 minutes.
- The drug is metabolized in liver.
- The average median half-life of Azithromycin is 68
- Azithromycin is primarily eliminated in feces as uncharged drug via biliary excretion and approximately 6% of dose is excreted in urine as an uncharged drug.
- The average steady state volume of distribution of the drug is 1 L/kg.
Which pregnancy category (A; B; C; D; X) has been assigned to Azithromycin
- The Azithromycin is classified by US FDA pregnancy category: B
- Due to lack of adequate and well-controlled studies the use of Azithromycin in pregnant women is contraindicated and recommended only when benefit justifies the risk.
- Laboratory animal studies have shown no adverse effect on the fetus.
- Studies support the excretion of the Azithromycin into human milk. Even though, breast-feeding may be permissible with caution.
- Despite these facts caution should be exercised when taking Azithromycin.
How to use azithromycin
- Azithromycin oral suspension and tablets are generally for oral administration with or without food.
- Azithromycin tablets should not be chewed, split or crushed. Whole tablet/capsules should be swallowed.
- Azithromycin oral liquid should be shaken well before every use.
- Azithromycin oral liquid should be measured with measuring spoon that comes along with medication.
- Normally one drop of Azithromycin ophthalmic solution is used in a day at any time.
- It is also recommended to take drug at almost the same time every day.
- Follow the instructions carefully as directed on prescription leaflet and take Azithromycin exactly as directed.
- Do not change the dose of the drug as prescribed by your doctor since the dosage is based on patient medical condition, treatment responses and usage with other drugs.
How to store the drug
- Azithromycin is stored at 25°C (77°F) and excursion permitted to 15-30°C (59-86°F).
- Store the medicine away from light and moisture.
- Azithromycin oral solution may be stored in refrigerator but should not be frozen.
- Ophthalmic solution should be stored at 2 to 8°C (36°F to 46°F).
- The drug should be kept away from children and pets.
- Medicine should not be stored in the bathroom.
How to dispose the medicine
- Throw away unused and opened, outdated or no longer used container.
- Also dispose the old medicine after the expiration date.
- Consult your pharmacist or local waste disposal company for proper disposal.
- Azithromycin was approved fromS. Food and Drug Administration (FDA) in November 1991 to treat variety of bacterial infections such asskin, ear, respiratory infections and sexually transmitted diseases.
- Azithromycin oral suspension was first approved in September 1994 by U.S. FDA.
- Azithromycin ophthalmic solution was approved in April 2007 by U.S. FDA.
Other uses of the drug
- Azithromycin is also used for the travelers’ diarrhoea and other gastrointestinal infections and babesiosis (tick-borne malaria like illness caused by species of the intra-erythrocytic protozoanBabesi).
- Azithromycin may also be used for other uses not listed here. It is advisable to ask your doctor or pharmacist for more information.
What special dietary precautions should I follow
- Take diet as prescribed by your doctor otherwise follow usual diet.
What special precautions should I follow/ What should I avoid while using Azithromycin
- Before taking Azithromycin, tell your doctor about your medical history if you have liver or kidney problem, blood infection and myasthenia gravis.
- Consult with your doctor if you have cystic fibrosis, irregular heartbeats, heart failure, infection of HIV and prolonged Qt intervals.
- Do not stop taking Azithromycin until you do not finish the prescription. If you do not follow the prescription or stop taking Azithromycin too early, it may lead to the development of antibiotic resistance in bacteria.
- Consult with your doctor and pharmacist if you are taking any prescription and non-prescription medications, nutritional supplements, vitamins or herbal products.
- Avoid touching the Azithromycin ophthalmic solution bottle tip with your finger, eye or other objects.
- Also avoid the use of contact lenses during the treatment of bacterial conjunctivitis.
- Consult your doctor in case of any query.
Azithromycin side effects
In addition to the associated benefits, Azithromycin also is accompanied with the side effects some of which are more common, others less common whereas some that fade away with time while you take the drug. It is always recommended to consult a doctor if you encounter any of the side effects.
Some of the commonly occurring side effects which require medical attention are outlined as:
- Diarrhoea
- Headache
- Nausea
- Pain in stomach
- Vomiting
Sometime, it may cause serious side effects. On appearance of these symptoms stop taking the medicine and immediately talk to your doctor:
- Blisters or peeling of the skin
- Dark-colored urine
- Dizziness
- Extreme tiredness
- Fainting
- Flu-like symptoms
- Hives
- Hoarseness
- Irregular or fast heart beat
- Itching
- Lack of energy
- Loss of appetite
- Mouth sores
- Rash
- Severe diarrhoea with or without fever and stomach cramps
- Swelling of the face, throat, tongue, lips, eyes, hands, feet, ankles, or lower legs
- Unusual bleeding or bruising
- Unusual muscle weakness
- Wheezing or difficulty breathing or swallowing
Azithromycin ophthalmic solution may cause some side effects in eye. These include:
- Blurred vision
- Changes to the surface of your eye
- Discharge coming from your eye
- Eye burning, stinging and irritation when the drop hits your eye
- Eye dryness
- Irritation on your eyelid and the skin around your eye
- Pain and itching in eye
- Stuffy nose and sinus infection
- Swelling around your eye or of your face
Besides these, Azithromycin may also be associated with some other side effects. These include:
- Cardiovascular effects: hypotension, palpitations, arrhythmias and prolongation of the QT intervels.
- Dermatologic effects: rashes, urticaria, pruritus and photosensitivity.
- Gastrointestinal effects: nausea, abdominal pain, vomiting, diarrhoea, pseudomembranous colitis, mucositis, oral moniliasis, flatulence, constipation and gastritis.
- Genitourinary effects: vaginitis and moniliasis.
- Hematologic effect: leukopenia, neutropenia, decreased lymphocyte, increased eosinophils and thrombocytopenia.
- Hepatic effect: elevated bilirubin, hepatic and cholestatic jaundice, hepatic necrosis and failure.
- Hypersensitivity effect: anaphylaxis, angioedema and Stevens-Johnson syndrome.
- Nervous system effects: dizziness, headache, somnolence, vertigo, hearing disturbance, paresthesia, convulsion, hyperactivity, nervousness and syncope.
- Renal effect: interstitial nephritis, acute renal failure and elevation in serum creatinine and blood urea nitrogen.
What should I do in case of overdose
- If you overdose the drug contact with your doctor or pharmacist for symptomatic and supportive measures.
- Overdose of Azithromycin may cause gastrointestinal effects such as abdominal pain, vomiting, diarrhoea and constipation.
What should I do in case of missed a dose
- Take a missed dose as soon as possible.
- To make up the missed dose, do not take extra medicine.
- Avoid taking the missed dose if it is about time for your next dose.
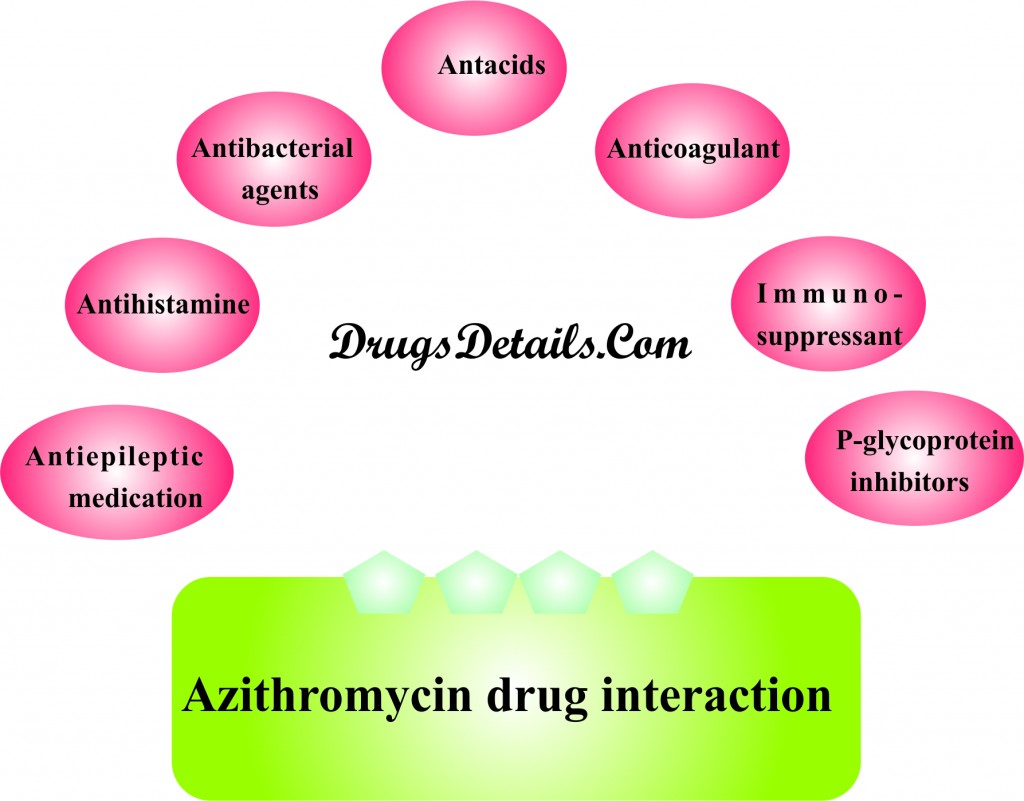
Azithromycin drug interactions
A number of drugs are known to interact with Azithromycin that can suppress or elicit the effective of the drug and poses the risk for serious side effects. It is always advisable to discuss about all the prescription, non-prescription drugs, vitamins and herbal supplements you are taking before you go on Azithromycin medication therapy. Also inform your doctor if you are undergoing any type of allergy skin testing or allergy treatments. Azithromycin may interact with one of the following drugs. Care should be taken when you are taking these medications together. Lists of few medications that interact with Azithromycin include:
- Drugs for the treatment of irregular heartbeats such as sotalol (Betapace, Sorine), quinidine, procainamide (Procanbid), dofetilide (Tikosyn) and amiodarone (Cordarone, Pacerone).
- Antacids which contain aluminium or magnesium such as Maalox, Mylanta and Tums.
- Anticoagulant medicines such as digoxin (Lanoxin), dihydroergotamine (D.H.E 45 and Migranal), ergotamine (Ergomar) and warfarin (Coumadin and Jantoven).
- Immunosuppressant drugs such as cyclosporins (Neoral and Sandimmune), midazolam (Dormicum, Hypnovel, Versed).
- Antiepileptic medications such as phenytoin (Dilantin,Dilantin) and carbamazepine (Carbatrol, Equetro, Epitol, Tegretol, Teril).
- Bronchodilator agents such as theophyllines (such as Norphyl, Phyllocontin, Theochorn).
- Drugs for treating pulmonary hypertension such as sildenafil.
- HMG-CoA reductase inhibitors such as atorvastatin, lovastatin, pravastatin, simvastatin, rosuvastatin, fluvastatin, pitavastatins.
- Drugs for the treatment of allergy such as antihistamine (Terfenadine).
- P-glycoprotein inhibitors such as colchicines, ritonavir, amiodarone, captopril, clarithromycin, reserpine, tariquidar, verapamil.
- Dihydrofolate reductase inhibitors such as trimethoprim (Proloprim, Monotrim, triprim).
- Drugs for HIV/AIDS including such as zidovudine (Retrovir), nelfinavir (Viracept), indinavir (Crixivan), efavirenz (Sustiva, Efavir, Stocrin), didanosine (Videx).
- Antibacterial agents such as rifabutin (Mycobutin), fluconazole (Monicure, Canesten, Alfumet or Dofil), sulfamethoxazole (Gantanol).
This list of drugs interacting with Azithromycin is not complete and other drugs may also interact with it. Always consult your doctor about the medications you use.
Does Azithromycin have any interaction with diseases
Before you begin to take Azithromycin, it is necessary to discuss any medical condition or allergies you have or any other significant medical fact. Chemically, Azithromycin is macrolides antibiotic and may interact with one or more than one of the following disease conditions.
- Antibiotics induced colitis: Antibiotic therapy usually associated with significant reduction in gut microbial flora, that permits overgrowth of some of the harmful bacteria over normal gut flora.Clostridium difficile is one of such bacterial species that flourish well under antibiotic therapy. Its toxin is supposed to be a primary cause of antibiotic-associated colitis. The most common antibiotics that cause colitis is Azithromycin, Lincomycin, Amoxicillin, Ampicillin (Aminopenicillins), Clindamycin, and Cephalosporins. Besides Clostridium, Pseudomembranous colitis has also been reported with broad spectrum antibacterial agents. Colitis is a medical condition caused by inflammation of large intestine and associated with growth of unusal harmful bacteria (microbial infection) and range in severity from mild to life-threatening. The disease is associated with severe, persistent diarrhea, abdominal cramps, and passage of blood and mucus. It has been observed that colitis is occasionally caused by taking antibiotics and thus named as antibiotic associated colitis. Thus, antibiotic therapy with Azithromycin and other broad-spectrum antibiotics agents should be managed cautiously in patients with a history of gastrointestinal diseases, particularly colitis. In such patients, antibiotic therapy should be discontinued immediately if significant diarrhea occurs and stool assay and large bowel endoscopy recommended to establish a definitive diagnosis.
- Liver Disease: Azithromycin and other broad spectrum, macrolide antibiotics are primarily eliminated from the body by the hepatobiliary system. Although clinical trials and studies have shown that Azithromycin is well-tolerated and less associated with adverse/negative side effects. However, it is recommended that Azithromycin therapy should be administered cautiously in patients with liver and/or biliary impairment.
- Myasthenia Gravis: Myasthenia gravis is a neuromuscular disease which is characterized by fluctuating muscle weakness and fatigue. It has been observed that administration of macrolide antibiotics such as Azithromycin can exacerbate symptoms of myasthenia gravis. Besides this, it has also been observed that Azithromycin treatment also initiate the onset of symptoms of myasthenic syndrome. The underlying cause of this is supposed to be due to presynaptic suppression of acetylcholine release from neurotransmitter. Therefore, Amoxiciliin therapy should be administered cautiously most important in patients which have a previous history of myasthenia gravis
Where can I get more information
- Your pharmacist or health care provider can provide more information about Azithromycin.
Clinical research and current scenario of the drug
- No studies have been performed either In vitro or In vivo for assessing the metabolism of Azithromycin.
- Studies indicate similar pharmacokinetic parameters of Azithromycin in elderly subjects (aged 65 to 85 years) and young adults.
- Long-term studies involving Azithromycin have not been performed in animals for evaluating carcinogenic potential.
- Safety and effectiveness of Azithromycin in the treatment of pediatric patients with otitis media, acute bacterial sinusitis and community-acquired pneumonia under 6 months of age have not been established.
- Clinical studies have not established the safety and effectiveness of Azithromycin in the treatment of pediatric patients with pharyngitis/tonsillitis below 2 years of age.
- Clinical trials indicated non-discontinuation of Azithromycin therapy due to any treatment-related laboratory abnormalities.
- Study indicates a 40% reduction in genital trachomatis infection after treatment with Azithromycin MDA (Mass Drug Administration) for the elimination of trachoma.
- Studies indicate the concomitant use of Ceftriaxone/Azithromycin for the treatment of pharyngeal gonorrhea in HIV-negative man.
- Cefazolin and Azithromycin are significantly more effective in the prevention of postcesarean endometritis.
References from chemical, biological and toxicological databases
- DrugBank: Erythromycin. www.drugbank.ca/drugs/DB00199
- Azithromycin | C38H72N2O12 | ChemSpider. http://www.chemspider.com/Chemical-Structure.10482163.html
- Azithromycin – National Library of Medicine – PubMed Health. http://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0000192/
- Azithromycin: MedlinePlus Drug Information. https://www.nlm.nih.gov/medlineplus/druginfo/meds/a697037.html
- Azithromycin – Wikipedia. https://en.wikipedia.org/wiki/Azithromycin
- A review of its antimicrobial activity, pharmacokinetic properties and clinical efficacy. http://www.ncbi.nlm.nih.gov/pubmed/1280567
- Effect of amoxicillin use on oral microbiota in young children. http://www.ncbi.nlm.nih.gov/pubmed/15273096
- Treatment of streptococcal pharyngitis with once-daily compared with twice-daily amoxicillin: a noninferiority trial. http://www.ncbi.nlm.nih.gov/pubmed/16940830
- Common harms from amoxicillin: a systematic review and meta-analysis of randomized placebo-controlled trials for any indication. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4284189/
- Amoxicillin for acute lower respiratory tract infection in primary care: subgroup analysis of potential high-risk groups. http://www.ncbi.nlm.nih.gov/pubmed/24567620
- In vivo activities of amoxicillin and amoxicillin-clavulanate against Streptococcus pneumoniae: application to breakpoint determinations. http://www.ncbi.nlm.nih.gov/pubmed/9736566
- Amoxicillin/clavulanic acid: a review of its use in the management of paediatric patients with acute otitis media. http://www.ncbi.nlm.nih.gov/pubmed/12534334
- Association of amoxicillin use during early childhood with developmental tooth enamel defects. http://www.ncbi.nlm.nih.gov/pubmed/16203939
- Amoxicillin-potassium clavulanate, a beta-lactamase-resistant antibiotic combination. http://www.ncbi.nlm.nih.gov/pubmed/6391783
- Comparative effectiveness of amoxicillin and amoxicillin-clavulanate potassium in acute paranasal sinus infections in children: a double-blind, placebo-controlled trial. http://www.ncbi.nlm.nih.gov/pubmed/3520469
- A case of persistent and possibly treatment resistant pharyngeal gonorrhea. http://www.ncbi.nlm.nih.gov/pubmed/26967304
- Mass drug administration of azithromycin for trachoma reduces the prevalence of genital Chlamydia trachomatis infection in the Solomon Islands.http://www.ncbi.nlm.nih.gov/pubmed/26888658
- A comparison of 3 antibiotic regimens for prevention of postcesarean endometritis: an historical cohort study. http://www.ncbi.nlm.nih.gov/pubmed/26901276
- Common harms from amoxicillin: a systematic review and meta-analysis of randomized placebo-controlled trials for any indication. http://www.ncbi.nlm.nih.gov/pubmed/25404399
- Augmentin (amoxicillin/clavulanate) in the treatment of community-acquired respiratory tract infection: a review of the continuing development of an innovative antimicrobial agent. http://www.ncbi.nlm.nih.gov/pubmed/14726431
- Amoxicillin or myringotomy or both for acute otitis media: results of a randomized clinical trial. http://www.ncbi.nlm.nih.gov/pubmed/2011422
