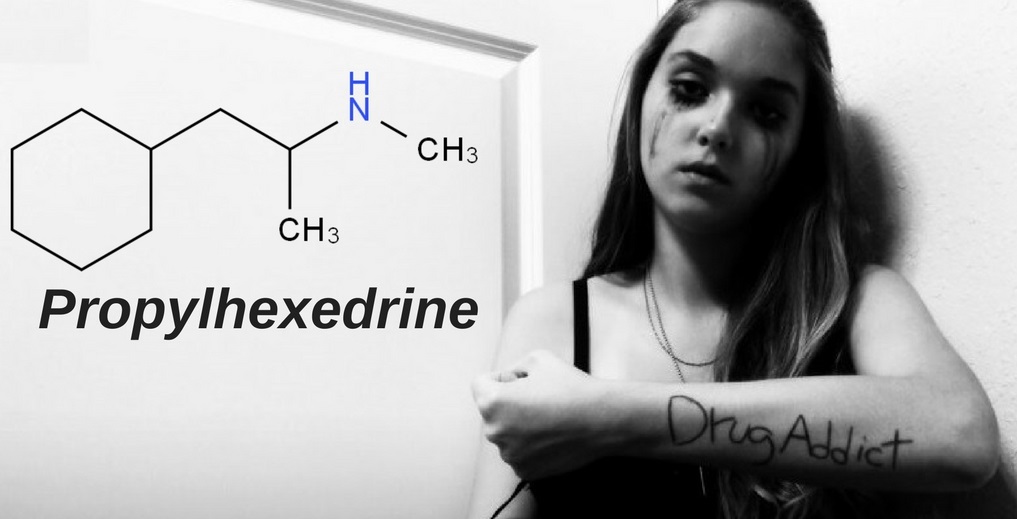Contents
- What is Propylhexedrine?
- Propylhexedrine IUPAC name, chemical structure, mass, formula
- Chemical characteristics and drug class of propylhexedrine
- Are amphetamines and propylhexedrine similar?
- Propylhexedrine mechanism of action
- Propylhexedrine history of use
- How is Propylhexedrine (Benzedrine and Benzedrex) abused?
- What are the street names for Propylhexedrine?
- Propylhexedrine abuse
- Propylhexedrine doses of abuse
- Propylhexedrine addiction and tolerance
- Propylhexedrine withdrawal
- Propylhexedrine pharmacokinetics. How long does propylhexedrine stay in the system?
- Propylhexedrine effects
- Propylhexedrine physical effects
- Propylhexedrine cognitive effects
- Stimulative effects of propylhexedrine
- Propylhexedrine after effects
- Propylhexedrine visual effects
- Propylhexedrine side effects and toxicity
- Can Propylhexedrine provoke mental disorders?
- How long Propylhexedrine effects lasts? How long Propylhexedrine stay in the system?
- Propylhexedrine detection
- Propylhexedrine legal status in the US
- Does Propylhexedrine show up on standard drug tests?
- Propylhexedrine intoxication treatment
- Propylhexedrine systemic intoxication and clinical case-reports
- Propylhexedrine interactions with other drugs
What is Propylhexedrine?
Propylhexedrine (on the market known as Benzedrex and Obesin) is a synthetic stimulant drug and structural analog of methamphetamine which is widely used as a nasal decongestant for relief of the symptoms of congestion due to colds, allergies and allergic rhinitis and sometimes used recreationally as an OTC “legal high”.
In the US, propylhexedrine is most commonly found in OTC Benzedrex inhalers. Benzedrex was first manufactured by Smith, Kline and French after the Benzedrine inhaler, which contained racemic amphetamine, became unavailable following the placement of amphetamines on the US Schedule II status (highest abuse potential, yet with accepted medicinal uses). Under the trade name Obesin, propylhexedrine has also seen use in Europe as an appetite suppressant.
Propylhexedrine IUPAC name, chemical structure, mass, formula
IUPAC name: N,α-dimethyl-cyclohexylethylamine
Other (trade) names: Benzedrex; Propylhexedrin; Dristan; Obesin; Hexahydrodesoxyephedrine
Molecular formula: C10H21N
Molecular weight: 155.285 g/mol
Molecular structure:
Chemical characteristics and drug class of propylhexedrine
Moreover, propylhexedrine is a chiral compound (the α-carbon is chiral), an active ingredient contained in Benzedrex inhalers is racemic (RS)-propylhexedrine as the free base. (S)-Propylhexedrine, also known as levopropylhexedrine, is believed to be the more biologically active isomer of the two. (S)-Propylhexedrine can be synthesized from methamphetamine.
This compound belongs to the class of organic compounds known as dialkylamines. These are organic compounds containing a dialkylamine group, characterized by two alkyl groups bonded to the amino nitrogen.
This slightly water-soluble compound is a clear, colorless liquid that vaporizes slowly at room temperature. The hydrochloride salt of propylhexedrine is a crystalline solid with a melting point of approximately 127 degree Celsius.
Are amphetamines and propylhexedrine similar?
Yes, there is a structural similarity between propylhexedrine and other common aromatic sympathomimetic compounds including methamphetamine and the neurotransmitter, epinephrine.
Propylhexedrine mechanism of action
Propylhexidrine causes the norepinephrine, dopamine, and serotonin (5HT) transporters to reverse their direction of flow. This inversion leads to a release of these transmitters from the vesicles to the cytoplasm and from the cytoplasm to the synapse. It also antagonizes the action of VMAT2, causing the release of more neurotransmitters.
Propylhexedrine history of use
The Smith, Kline, and French Company introduced Benzedrine inhalers during the early 1930s as racemic amphetamine. One Benzedrine inhaler contained the equivalent of 250 mg of racemic amphetamine. These inhalers remained available as OTC products until 1949 when Smith Kline & French voluntarily removed amphetamine inhalers from the market in response to reports of abuse and sudden death.
The Benzedrine inhaler was replaced by the Benzedrex inhaler (B.F. Ascher & Co., Lenexa, KS), which contained 250 mg of propylhexedrine (i.e., no amphetamine) in a combination with 12.5 mg methanol and various aromatics hydrocarbons. Some airlines (e.g., Pan American) distributed Benzedrex inhalers in the 1950s as decongestants to reduce discomfort resulting from changes in cabin pressures.
How is Propylhexedrine (Benzedrine and Benzedrex) abused?
The usual method of Benzedrine and Benzedrex abuse involved the chewing and swallowing of the amphetamine- containing cotton pledget, preferably with chewing gum to reduce the irritant properties of the base. Occasionally, the strips were soaked in beverages (i.e., coffee, alcohol) or swallowed whole.
One study of an incarcerated population in the 1940s revealed that 25% of the inmate population used the inhalant material for recreational purposes. By the mid – the 1970s, the medical literature contained reports of both oral and intravenous (IV) abuse of the propylhexedrine inhalers.
The abuse of propylhexedrine was sporadic throughout North America with cases reported from Dallas, Toronto, and San Francisco during the 1980s; however, there is little current data on propylhexedrine abuse because most national US databases on drug abuse do not include propylhexedrine.
Abuse of propylhexedrine nasal inhalers involves habitual inhalation, ingestion or chewing of the propylhexedrine -containing cotton plug, or injection of an acidic solution prepared from the dismantled inhaler. Nasal abuse of propylhexedrine is uncommon.
What are the street names for Propylhexedrine?
The street names for the propylhexedrine extract include Bathtub Crystal, Crystal, and Bathtub Crank.
Propylhexedrine abuse
Propylhexedrine is a potent α – adrenergic sympathomimetic agent, which is a “stimulant of last resort” for abusers when more desirable stimulants are unavailable.
However, the abuse potential of propylhexedrine is low compared with methamphetamine, phenmetrazine, or methylphenidate. Most drug users prefer cocaine and methamphetamine to propylhexedrine, and propylhexedrine abuse is typically brief as the users rapidly lose interest in the drug.
Propylhexedrine doses of abuse
Habitual use of these inhalers involves the daily use of the contents of 2 to 11 inhalers. Following the disassembly of the inhaler, the cotton pledget is soaked in muriatic (hydrochloric) acid to remove the propylhexedrine. Heating of this extract produces a brown residue that subsequently is redissolved in water and injected.
Depending on the purity of the process, the resultant solution contains a variety of impurities including polymeric material, fine fibers, methanol, and lavender oil. Some patients with previous amphetamine experience consider the euphoria induced by the IV injection of Benzedrex extracts, 15 mg of methamphetamine, and 75 mg of phenmetrazine similar. Benzedrex abuse is commonly associated with polydrug abuse and homosexual activities. Euphoria apparently does not result from inhalation.
Dose-effects:
- Threshold effects – 10 – 31.25 mg
- Light effects – 31.25 – 62.5 mg
- Common effects – 62.5 – 125 mg
- Strong effects – 125 – 187.5 mg
- Heavy effects – 187.5 mg +
Propylhexedrine addiction and tolerance
Propylhexedrine is a drug, so the chronic use can be extremely addictive with a high potential for abuse. This drug is also capable of causing psychological dependence among certain users. Once addiction is developed, withdrawal effects and cravings may occur if an abuser suddenly stops the usage.
Tolerance to the effects of propylhexedrine likely occurs as with amphetamine. It rapidly develops with prolonged and repeated use. In that manner, the abuser has to administer increasingly large doses to achieve the same effects.
Therefore, it takes up to 3 – 7 days for the tolerance to be reduced to half and 1 – 2 weeks to be back at baseline (in the absence of further consumption). Additionally, propylhexedrine presents cross-tolerance with all dopaminergic stimulants, meaning that after the consumption of propylhexedrine all stimulants will have a reduced effect.
Like with methamphetamine and amphetamine, the evidence on effective treatments for dependence and abuse is limited. Fluoxetine and imipramine appear to have some limited benefits in treating propylhexedrine’s abuse and addiction.
Propylhexedrine withdrawal
It is known that in highly dependent methamphetamine and amphetamine abusers, “when chronic heavy abusers abruptly stop with methamphetamine use, many report a time-limited withdrawal syndrome that usually occurs within 24 hours of their last dose”.Withdrawal symptoms in high-dose and chronic users are often, occurring in up to 87.6% of cases, and persist for 3-4 weeks with a marked “crash” phase occurring during the first week.
Withdrawal symptoms of propylhexedrine can include anxiety, drug craving, dysphoric mood, increased appetite, fatigue, increased movement or decreased movement, lack of motivation, sleeplessness or sleepiness, and vivid or lucid dreams. Withdrawal symptoms are associated with the degree of dependence.
Propylhexedrine pharmacokinetics. How long does propylhexedrine stay in the system?
Propylhexedrine penetrates the blood-brain barrier rapidly. In rodent studies, this drug has a large volume of distribution (V d = 19.3 L/kg) after IV administration.
Biotransformation of propylhexedrine involves N – demethylation, N – oxidation, and 4 – hydroxylation with urinary metabolites including norpropylhexedrine, cyclohexylacetoxime, and 4 – hydroxypropylhexedrine. The approximate elimination half-life of propylhexedrine after IV administration to rodents is about 2.5 hours.
Propylhexedrine effects
The ingestion of a hot water extract of a cotton pledget containing 250 mg propylhexedrine produced anxiety, agitation, tremors, nausea, and diaphoresis that resolved without sequelae 4 hours after ingestion. Ingestion of an estimated 375 mg propylhexedrine may cause tremor, muscle spasticity, tachycardia, and insomnia that resolved without sequelae.
Propylhexedrine physical effects
Propylhexedrine can provoke following physical effects:
- Abnormal heartbeat
- Increased blood pressure
- Increased heart rate
- Appetite suppression
- Bronchodilation
- Dehydration
- Frequent urination
- Increased bodily temperature
- Increased perspiration
- Nausea
- Stamina enhancement
- Teeth grinding
- Temporary erectile dysfunction
- Vasoconstriction
Propylhexedrine cognitive effects
Propylhexedrine can provoke following cognitive effects:
- Disinhibition
- Euphoria
- Analysis enhancement
- Anxiety
- Compulsive redosing
- Ego inflation
- Euphoria
- Focus enhancement
- Increased libido
- Increased music appreciation
- Memory enhancement
- Motivation enhancement
- Thought acceleration
- Thought organization
- Wakefulness
Propylhexedrine cognitive effects can be broken down into several components which progressively intensify proportional to dosage. The general head space of propylhexedrine is described by many as one of moderate to extreme mental stimulation, increased focus, and powerful euphoria.
Negative adverse effects are usually mild at low to moderate dosages, they might become very likely to manifest themselves with higher amounts or extended usage. This particularly holds true during the offset of the experience.
Stimulative effects of propylhexedrine
Propylhexedrine is usually considered as extremely energetic and stimulating agent and these effects are extremely similar to that of amphetamine or methamphetamine, and stronger than that of caffeine, modafinil, and methylphenidate. Stimulating effects most commonly encourage physical activities such as dancing, socializing, running, or cleaning.
The particular way of stimulation can be described as pressing or forced and is noticeably more adrenergic and tense on the body. Therefore, at higher dosages, it may become difficult or impossible to keep still as jaw clenching, involuntarily bodily shakes and vibrations become present, resulting in the extreme shaking of the entire body, unsteadiness of the hands, and a general lack of motor control.
Propylhexedrine after effects
The effects which occur during the offset of a stimulant experience generally feel negative and uncomfortable in comparison to the effects which occurred during its peak. This is often referred to as a “comedown” and occurs because of neurotransmitter depletion. Its effects commonly include:
- Anxiety
- Appetite suppression
- Cognitive fatigue
- Depression
- Irritability
- Motivation suppression
- Thought deceleration
- Wakefulness
Propylhexedrine visual effects
Similarly to the amphetamine or methamphetamine, the visual effects of propylhexedrine are usually less consistent and are only mildly noticeable at higher dosages. They are somewhat comparable to the visual effects produced by deliriants and are generally more frequent in darker areas.
Propylhexedrine may provoke following visual disturbances:
- Double vision – This effect is uncommon and will only occur at very high, potentially dangerous or toxic dosages.
- Drifting – the effect is usually subtle and barely noticeable and only occurs at higher dosages or when combined with cannabis. Commonly this consists of level 1-2 drifting.
- Brightness alteration
Propylhexedrine side effects and toxicity
Propylhexedrine is a potent local vasoconstrictor with substantially less (i.e., < 10%) CNS stimulating effect compared with dextroamphetamine sulfate. The acute toxicity of propylhexedrine is an exacerbation of pharmacologic effects of this drug. The psychoactive effects of these compounds result from indirect agonist actions that cause the displacement of catecholamines from binding sites in the storage vesicles.
At least in part, the central effects of propylhexedrine result from alterations in the neurotransmitters, dopamine, and norepinephrine, because of the close structural resemblance of amphetamine compounds to these neurotransmitters. Increasing the synaptic concentration of these neurotransmitters either by direct release from storage vesicles or by inhibition of reuptake is the most likely mechanism of toxicity.
Mild intoxication causes restlessness, irritability, anxiety, mydriasis, diaphoresis, nausea, and tachycardia. More severe effects associated with serious propylhexedrine intoxication include hypertension, tachypnea, cardiac dysrhythmias, hyperpyrexia, seizures, psychosis, and coma.
Autopsies of individuals with a history of IV propylhexedrine abuse usually demonstrate foreign body granulomas in the pulmonary parenchyma containing birefringent crystalline material along with variable amounts of pulmonary hypertension and right ventricular hypertrophy.
Histologic examination reveals diffuse interstitial alveolar fibrosis, pulmonary edema with focal intraalveolar hemorrhage, intimal and medial proliferation with luminal obstruction, medial hypertrophy of the muscular arteries, and focal myocardial fibrosis (usually without clinically significant coronary artery stenosis).
Can Propylhexedrine provoke mental disorders?
Similarly to methamphetamine, propylhexedrine abuse can also result in a stimulant-induced psychotic state that may present with a variety of symptoms including: paranoia, delusions, hallucinations, though likely to a lesser degree.
A case report associated the development of an acute paranoid psychosis with the daily chewing of 10 – 12 propylhexedrine inhalers by 2 amphetamine abusers. The psychosis in both of these patients resolved after the cessation of drug use. Initially, the propylhexedrine abuser can experience a headache, nausea, and double vision followed by euphoria that lasts several hours.
Case reports associate deterioration of chronic paranoid schizophrenia with the chewing of propylhexedrine decongestant inhalants as manifest by the onset of acute delusional mood and paranoid delusions. Other factors (sleep deprivation, predrug personality, idiosyncratic reactions) contribute to the development of the toxic psychosis. Homicidal and suicidal ideations may occur during drug-induced psychosis.
How long Propylhexedrine effects lasts? How long Propylhexedrine stay in the system?
Propylhexedrine effects usually last from 4-10 hours. The onset of action is after 10-30 minutes. The strongest effects can be expected after 2-5 hours. After effects can last 2-12 hours.
Propylhexedrine detection
Analysis of biologic samples for propylhexedrine is similar to the methods used to detect methamphetamine and amphetamine; typically these methods involve gas chromatographic separation with detection by flame ionization (FID), mass spectrometry (MS), or nitrogen –phosphorus detection (NPD).
Challenges associated with inadequate chromatographic resolution or coelution of propylhexedrine with methamphetamine can be resolved by the use of selective ion monitoring (SIM) mode in the mass spectrometer.
Preanalytical derivatization with heptafluorobutyric anhydride or 4 – carbethoxyhexafl uorobutyryl chloride following chromatographic separation and MS detection in SIM mode also effectively distinguishes methamphetamine from propylhexedrine.
Mass spectral fragmentation of methamphetamine includes the tropylium ion (m/z 91) from phenyl ring expansion and benzyl carbon incorporation; the lack of a phenyl ring in propylhexedrine eliminates this ion from its mass spectrum. Additionally, the mass spectrum of propylhexedrine includes a prominent occurrence at m/z 182, which is absent in the mass spectrum of methamphetamine.
High performance liquid chromatography/atmospheric pressure chemical ionization/mass spectrometry does not require preanalytical derivatization necessary for gas chromatographic analysis. This method allows the separation of sympathomimetic amines including propylhexedrine with limits of detection in the range of 0.001 – 0.005 mg/L.
Propylhexedrine legal status in the US
Propylhexedrine is a legal OTC drug. It is designed not to be recreationally used unless the user decides to bypass the manufacturer’s intended design with which it was granted approval to market as a nasal decongestant. The simple consumption of this substance is not technically considered illegal.
Does Propylhexedrine show up on standard drug tests?
Since it is structurally related to metamphetmaine and amphetamines drugs, it can be expected to show up on commercial drug tests.
Propylhexedrine intoxication treatment
The treatment of propylhexedrine intoxication is similar to the treatment of methamphetamine and amphetamine toxicity. Most agitated patients intoxicated with propylhexedrine respond to reassurance, a quiet environment, and IV benzodiazepines (lorazepam, diazepam).
Similar to methamphetamine, the major life threatening complications of acute propylhexedrine intoxication include hyperthermia, hypertension, seizures, cardiovascular instability, and trauma. Coma, shock, acute renal failure, severe hyperthermia (i.e., temperature > 41 o C/105.8 oF ), and seizures are poor prognostic indicators.
Severely intoxicated patients require IV access, cardiac monitoring, pulse oximetry, an ECG, and supplemental oxygen. Respiratory depression does not usually occur during mild to moderate propylhexedrine intoxication, but pulmonary edema (i.e., cardiogenic, acute lung injury) can develop during severe propylhexedrine poisoning. Underlying pulmonary hypertension can occur in habitual IV propylhexedrine users.
Most patients tolerate sinus tachycardia without pharmacologic intervention unless the tachycardia results from right-sided heart failure. Both hypertension and tachycardia often respond to IV benzodiazepines (Adult: lorazepam 2 mg or diazepam 5 mg IV bolus titrated to effect). Core temperatures should be measured in any agitated patient.
Patients with suspected myocardial ischemia should be managed according to the usual guidelines for myocardial ischemia including aspirin, antiplatelet agents, thrombolytics, primary percutaneous coronary intervention, and blood pressure control as needed. Hypotension may respond to fluid challenges, but often a vasopressor is needed.
Shock is a poor prognostic sign that indicates the need for monitoring of cardiac output to determine the most efficacious combination of fluid and vasopressors. There are no specific antidotes for propylhexedrine intoxication or efficacious methods to enhance the elimination of propylhexedrine.
Propylhexedrine systemic intoxication and clinical case-reports
Peripheral vascular complications of IV propylhexedrine abuse include local ischemia with necrosis of the digits requiring amputation, cellulitis, sterile abscess, and a fatal abscess in the neck after an attempted central venous injection of an aqueous extract of propylhexedrine – containing inhalers.
Incidental injection of the propylhexedrine extract into a peripheral artery caused local vasoconstriction, inflammation, infection, and necrosis manifest by intense pain, marked edema, blanching of the extremity, and loss of distal arterial pulses.
A case report of a 22 year old man associated the development of pulmonary edema, hypotension, anterior myocardial infarction on electrocardiogram (ECG), accelerated junctional rhythm, and pericardial effusion with the ingestion of 250 mg of propylhexedrine obtained from a Benzedrex inhaler several hours prior to the onset of symptoms. The patient recovered from intensive cardiorespiratory support. There was no cardiac angiography or echocardiography.
Pulmonary edema is a common finding among habitual IV propylhexedrine abusers presenting to emergency departments with dyspnea and cardiovascular instability; cor pulmonale can develop in these patients. Case reports associated the IV use of propylhexedrine extracts with the development of brainstem dysfunction including transient diplopia, right – internuclear ophthalmoplegia, and focal paralysis of the tongue
Propylhexedrine interactions with other drugs
If combined with other drugs, propylhexedrine may become life-threatening. Although, certain combinations may be safe in low doses of each but still increase the potential risk of death. Propylhexedrine should be avoided in combination with following drugs:
- Tricyclic antidepressants- Propylhexedrine may increase the effects of tricyclic antidepressants to dangerous levels.
- Alcohol- Stimulants decrease the sedative effect of alcohol which is the main factor most people consider when determining their level of intoxication. Once the stimulant wears off, the effects of alcohol will be significantly increased, leading to intensified disinhibition as well as respiratory depression. If combined, one should strictly limit themselves to only drinking a certain amount of alcohol per hour.
- DXM- This combination may cause increased heart rate and panic attacks.
- MXE- Increased heart rate and blood pressure may occur.
- Tramadol- This combination can increase the risk of seizures.
- MDMA- Like with amphetamines, the neurotoxic effects of MDMA may be increased when combined with propylhexedrine.
- MAOIs- This combination may increase the number of neurotransmitters such as dopamine to dangerous or even fatal levels.
- Cocaine- This combination may increase strain on the heart to dangerous levels.
“What is it like to take prolintane?”
“Is Soframycin an antibiotic? What is the use of Soframycin?