Contents
- What is Rivastigmine (Exelon)?
- What is the generic and brand name of the drug?
- What is the source of the drug (natural or synthetic)?
- Why is this medication prescribed?
- Pharmacophore structure: Information about the chemical structure of the drug.
- Chemical information of the drug
- What is the available strength of the drug?
- When should I discontinue, withhold or modify the dose of Rivastigmine?
- What are the pharmacokinetic properties of the drug?
- Which pregnancy category (A; B; C; D; X) has been assigned to Rivastigmine?
- How to use the drug?
- How to store the drug?
- How to dispose the medicine?
- Does Rivastigmine has approval from government / FDA /or any other related agencies?
- Other uses of the drug.
- What special dietary precautions should I follow?
- What special precautions should I follow?/ What should I avoid while using Rivastigmine?
- What are the possible side effects of this drug?
- What should I do in case of overdose?
- What should I do in case of missed a dose?
- Does Rivastigmine have any interaction with diseases?
- Where can I get more information?
- Clinical research and current scenario of the drug.
- References from chemical, biological and toxicological databases.
What is Rivastigmine (Exelon)?
- Rivastigmine is a parasympathomimetic or cholinergic class of drug that is used to treat symptoms of dementia (neurological disorder).
What is the generic and brand name of the drug?
- The drug is available under generic name Rivastigmine and marketed under brand name Exelon
- The drug was originally developed by Marta Weinstock-Rosin of the Department of Pharmacology at the Hebrew University of Jerusalem and sold by Novartis Pharmaceuticals Corporation.
What is the source of the drug (natural or synthetic)?
- Rivastigmine is a semi-synthetic (man-made) pharmaceutical derivative of physostigmine, a parasympathomimetic alkaloid (reversible cholinesterase inhibitor) that is obtained from Calabar bean.
Why is this medication prescribed?
- Rivastigmine is a parasympathomimetic drug also known as cholinergic agent, which stimulates the parasympathetic nervous system.
- The drug is used to prevent or reduce the symptoms of mild to moderate dementia in people with Alzheimer’s disease (a neurodegenerative disease characterized by progressive memory loss and impaired cognitive behaviour).
- Dementia is a medical condition characterized by progressive memory loss (ability to remember and think properly), reduced ability to communicate, and to execute daily activities.
- The drug is also used to treat symptoms of dementia in people with Parkinson’s disease (a neurodegenerative disorder of the central nervous system).
- The drug improves the memory and cognitive functions by increasing the amount of acetylcholine substance in the brain.
Pharmacophore structure: Information about the chemical structure of the drug.
Rivastigmine is a parasympathomimetic or cholinergic agent that is semi synthetic derivative of natural substance physostigmine.
Chemically, Rivastigmine belongs to the class of organic compounds which are known aralkylamines, in which alkyl group is replaced by an aromatic hydrocarbyl group at one carbon atom. The detailed chemical classification of Rivastigmine is described below:
| Kingdom | Organic compounds |
| Super Class | Organonitrogen compounds |
| Class | Amines |
| Sub Class | Aralkylamines |
| Direct Parent | Aralkylamines |
Chemical information of the drug
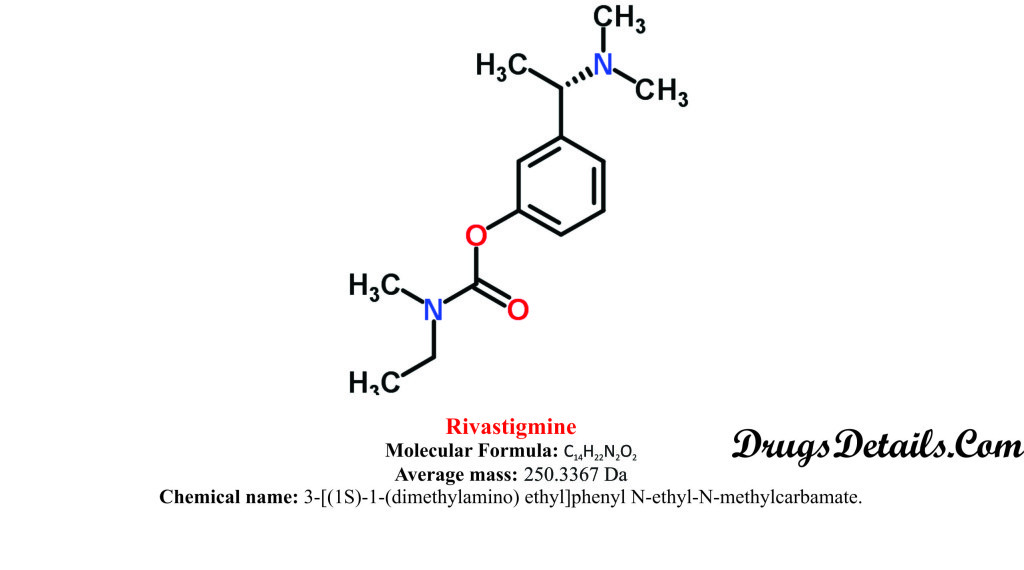
- Rivastigmine is a semi-synthetic pharmaceutical organic compound named as 3-[(1S)-1-(dimethylamino) ethyl]phenyl N-ethyl-N-methylcarbamate.
- The compound has molecular formula C14H22N2O2 and the molecular weight of 3367 Da.
- The melting point of Rivastigmine is 158 °C.
- Rivastigmine is a strong base with a pKa value of 8.89 and have water solubility of 2.04 mg/ml.
- Rivastigmine is available as Rivastigmine tartrate, which is commonly known in pharmacy as SDZ ENA 713 or ENA 713.
- Rivastigmine is almost insoluble in water at neutral pH (pH-7.0), however, solubility is increased dramatically at pH 1.
- Rivastigmine tartrate is chemically represented as C14H22N2O2 C4H6O6 (hydrogen tartrate salt–HTA salt) and has a molecular weight of 400.43.
- Rivastigmine tartrate is fine crystalline powder and white to off-white in colour.
What is the available strength of the drug?
- The medication is available as capsules, liquid oral solution and skin patches.
- Rivastigmine (Exelon) is usually recommended to be taken twice daily with food.
- Capsule and solution forms are for oral administration whereas skin patch is for transdermal application.
- The capsule is available in varying strengths as follows:
- 5 mg capsule is yellow in color and imprinted with “Exelon 1,5 mg” in red on the capsule body.
- 3 mg capsule is orange in color and imprinted with “Exelon 3 mg” in red on the capsule body.
- 5 mg capsule is red in color and imprinted with “Exelon 4,5 mg” in white on the capsule body.
- 6 mg capsule is orange and red in color and imprinted with “Exelon 6 mg” in red on the capsule body.
- Rivastigmine (Exelon) solution is clear yellow and contain Rivastigmine tartrate equivalent to 2 mg/mL of Rivastigmine base.
- Rivastigmine (Exelon) patch is available in three different strengths. Each patch has a backing layer which is labelled as:
- EXELON® PATCH 4.6 mg/24 hours, AMCX
- EXELON® PATCH 9.5 mg/24 hours, BHDI
- EXELON® PATCH 13.3 mg/24 hours, CNFU
- Besides Rivastigmine tartarate, each capsule contains other inactive ingredients such as magnesium stearate, hydroxypropyl methylcellulose, silicon dioxide, and microcrystalline cellulose. The gelatin capsule is comprised of gelatin, titanium dioxide and red and/or yellow iron oxides.
- Inactive ingredients in Rivastigmine (Exelon) solution include D&C yellow #10, purified water, sodium benzoate, citric acid, and sodium citrate.
- The patch consists of four layers namely, backing layer, drug matrix, adhesive matrix, and overlapping release liner (which is discarded before use).
- The constituents present within the formulation in patch include acrylic copolymer, poly (butylmethacrylate, methylmethacrylate), silicone adhesive applied to a flexible polymer backing film, silicone oil, and vitamin E.
How the medicine works (mode of action)?
- Rivastigmine is responsible for improving the mental function such as memory and thinking (cognitive deficits) in dementia associated with Alzheimer’s disease and Parkinson’s disease via
- Increasing the quantity of some natural substance like acetylcholine in the brain.
- Facilitating cholinergic neurotransmission.
- Memory loss and cognitive deficits associated with Alzheimer’s are mainly due to lack of acetylcholine. The deficiency of acetylcholine is principally due to selective loss of cholinergic neurons in the nucleus basalis, cerebral cortex, and hippocampus part of the brain.
- Acetylcholine is a chemical that is responsible for thinking, memory and perception.
- The precise mechanism of action of Rivastigmine is not known but it is postulated that the drug exerts its therapeutic effect by enhancing cholinergic function.
- Rivastigmine belongs to the class of medications known as cholinesterase inhibitors that binds reversibly with cholinesterases (both butyrylcholinesterase and acetylcholinesterase).
- The Rivastigmine interaction with its target enzyme i.e. acetylcholinesterase results in a covalently bound complex that brings about the temporary inactivation of the enzyme.
- As a result, Rivastigmine inactivates cholinesterase (eg. acetylcholinesterase, butyrylcholinesterase), and prevents the hydrolysis of acetycholine. This results in an increased concentration of acetylcholine at cholinergic synapses, which facilitates the cholinergic neurotransmission at the junctions between nerve cells.
- The anti-cholinesterase activity of Rivastigmine is mostly specific for brain acetylcholinesterase and butyrylcholinesterase as compared with those in peripheral tissues.
What are the recommended doses of Rivastigmine?
The doses of Rivastigmine vary depending upon the diseased state, response to treatment, interruption period and specific population.
- Dosing in Alzheimer’s disease: The drug should be taken twice a day (morning and evening) with meals. The recommended dosage of drug as solution and capsules varies from 6 mg to 12 mg per day, administered twice a day.
- Initial Dose: A dose of 1.5 mg twice a day is recommended.
- Dose Titration: The dose of the drug can be increased to 3 mg twice a day only if the drug is well tolerated and after a period of 2 weeks with subsequent increase to 4.5 mg twice a day and 6 mg twice a day only after a minimum of 2 weeks at the previous dose and if well tolerated. The maximum recommended dose is 6 mg twice a day (12 mg once a day).
- Dosing in Parkinson’s disease dementia: The drug should be taken with food in the morning and evening in divided doses. The drug dosage recommended in dementia associated with Parkinson’s disease is 3 mg to 12 mg once a day that is to be administered twice daily (daily doses of 1.5 mg to 6 mg twice a day).
- Initial Dose: A dose of 1.5 mg twice a day is recommended.
- Dose Titration: The dose of the drug can be increased to 3 mg twice a day only if the drug is well tolerated and after a period of 4 weeks with subsequent increase to 4.5 mg twice a day and 6 mg twice a day only after a minimum of 4 weeks at the previous dose and if well tolerated. The maximum recommended dose is 6 mg twice a day (12 mg once a day).
- Interruption of treatment: The treatment is discontinued for several doses (for a period of 3 days or less) in case the patient encounters the side effects such as abdominal pain, vomiting, nausea, loss of appetite etc. The drug treatment is then reinitiated either at the same drug dose or at a lower drug dose level. In case the interruption is for more than 3 days, the treatment should be restarted at 1.5 mg twice daily and titration is carried out as in described for Alzheimer’s disease or Parkinson’s disease.
- Dosing In Specific Populations:
- Dose modifications in case of renal impairment: Patients with moderate or severe renal impairment can tolerate only lower doses.
- Dose modifications in case of hepatic impairment: Patients with mild and moderate hepatic impairment can tolerate only lower doses. No data exists for the use of the drug in case of severe hepatic impairment.
- Dose modifications in case of Low Body Weight: Patients with low body weight of less than 50 kg are prone to toxicities such as vomiting and excessive nausea. Such patients are required to be monitored carefully and their drug dose should be adjusted accordingly.
When should I discontinue, withhold or modify the dose of Rivastigmine?
- The dose of Rivastigmine should be adjusted based on the patient’s clinical status, and medical history.
- The drug is contraindicated in case of pregnancy or breastfeeding.
- The use of the drug is restricted in patients who are hypersensitive to any component of the drug.
- Rivastigmine is contraindicated in case of a previous history of allergic contact dermatitis with Rivastigmine transdermal patch.
- The use of Rivastigmine is contraindicated in case of hepatic impairment and heart problem.
- Safety and effectiveness of the drug has not been evaluated in case of pediatric populations.
- The use of the drug should be under professional monitoring in case of patients with a history of seizures (e.g., epilepsy), inflammation of the pancreas (pancreatitis), stomach or bowel problems, or a urinary blockage (trouble urinating) and lung or breathing problems (e.g., asthma, chronic obstructive pulmonary disease)
What are the pharmacokinetic properties of the drug?
- At lower doses (up to 3 mg twice a day) Rivastigmine shows a linear pharmacokinetics. However, at higher doses (6 mg or higher twice a day) Rivastigmine results in non linear pharmacokinetics with a 3-fold increase in AUC (area under the plasma drug concentration-time curve).
- Following administration Rivastigmine is rapidly and completely absorbed by the body.
- The peak plasma concentrations of Rivastigmine are achieved in approximately 1 hour.
- The absolute bioavailability of Rivastigmine following administration of 3-mg dose is approximately 36%.
- Bioavailability of Rivastigmine is significantly affected by the food. Administration of Rivastigmine with food delays its absorption by the body.
- Following absorption, only a small amount of Rivastigmine is bound to plasma proteins (approximately 40%).
- Rivastigmine readily crosses the blood-brain barrier, reaching CSF (cerebrospinal fluid) peak concentrations in 1.4 to 2.6 hours.
- The apparent volume of distribution of Rivastigmine is in the range of 1.8 to 2.7 L/kg.
- Following absorption, Rivastigmine is rapidly metabolized via cholinesterase-mediated hydrolysis to the decarbamylated metabolite.
- The major route of elimination of Rivastigmine is via the kidney (half life 1.5 hours). It has been observed that out of total Rivastigmine administrated, 97% was eliminated in urine, while 0.4% is eliminated in
Which pregnancy category (A; B; C; D; X) has been assigned to Rivastigmine?
- The Rivastigmine is classified by US FDA pregnancy category: B.
- Due to lack of adequate and well-controlled studies, the use and safety of Rivastigmine in pregnant women is contraindicated and recommended only if clearly needed.
- No adequate data is available on excretion of Rivastigmine into human breast milk. However, the use of drug is not recommended in nursing mothers.
- Due these facts caution should be exercised when taking Rivastigmine during pregnancy.
How to use the drug?
The drug is available as capsule, solution (liquid) and transdermal patches to be taken orally through mouth or used directly to skin. The recommended doses of Rivastigmine is twice a day with meal usually in the morning and evening.
Rivastigmine capsule:
- Follow the instructions carefully as directed on prescription leaflet and take Rivastigmine exactly as directed by your health care professional.
- Take the medication regularly, even if you feel well. Try to take the medicine at the same time every day.
- Do not stop taking the medication abruptly without consulting your doctor.
- Do not change the dose of the drug as prescribed by your doctor. Since, dosage and duration of treatment are based on your medical condition and response to treatment.
- Usually Rivastigmine administration is started with low doses, which gradually increases with time (not more than once every 2 weeks).
- In case if you are using Rivastigmine oral solution, it is recommended to ask your pharmacist for a manufacturer guidelines for proper use of drug.
Rivastigmine solution:
- The oral dose of Rivastigmine should be taken using oral dosing syringe that comes with Rivastigmine oral solution. In case, if you are feeling any confusion ask your doctor or pharmacist.
- The oral solution of Rivastigmine is ready to use and can be taken directly from the syringe or it can be mixed with some liquid (small glass of water, fruit juice or soda) before use.
- Mix properly the Rivastigmine oral solution and do not use any liquid other than the listed. It is recommended to take medicine with in 4 hour if mixed in water, soda or fruit juice.
Following are some instruction to use Rivastigmine oral solution.
- Remove the protective cap from the bottle of Rivastigmine solution.
- Remove the oral dosing syringe from its shielding case (came along with this medication).
- Keep the tip of the oral syringe into the opening on top of the Rivastigmine solution bottle to take the desired amount of the solution.
- Pull up the plunger of the syringe to the graduated mark on the syringe (equal to your prescribed dose).
- Remove the air bubble from the syringe by gently up and down the syringe plunger. Do not be anxious about a few tiny air bubbles.
- Remove the syringe from the bottle and swallow the dose from the syringe directly or mix it with some liquid.
- Clean the corner and outside of the syringe with a clean tissue and put it back into the case for further use.
- Tightly close the protective cap of the Rivastigmine solution bottle and keep it away from children.
Rivastigmine transdermal patches:
- Transdermal patch of Rivastigmine is available for direct application to the skin.
- The recommended dose of transdermal patch is usually once a day.
- Follow the instructions carefully as directed on prescription leaflet and take Rivastigmine patch exactly as directed by your health care professional. Do not apply the dose more or less as prescribed by your doctor and use it at around the same time.
- Your doctor will probably start you on a low dose of Rivastigmine and gradually increase your dose, not more often than once every 4 weeks.
- It is suggested to apply the patch to clean, dry and hair free skin (upper or lower back or upper arm or chest).
- Use of patch is contraindicated in case of an open wound or cut, or skin problem, or rashes or allergic or irritated skin.
- Do not use the patch at the same place and select a different area to avoid skin irritation.
- Remove the previous patch before applying a new one and if patch falls off or loosen replace it with new one.
- Do not expose the patch to direct sunlight for very long.
- During the use of Rivastigmine patch try to avoid excess direct heat by using electric blankets, heat lamps, heating pads, saunas, hot tubs, and heated water beds.
How to store the drug?
- Rivastigmine is stored at room temperature 25°C (77°F).
- Do not store the drug in the freezer.
- Brief excursion period is permitted at temperature 15–30°C (59–86°F).
- The drug should be kept away from excess heat, direct sun light, moisture and reach of children.
- The drug should not be stored in bathroom.
How to dispose the medicine?
- Throw away unused and opened, outdated or no longer used drug.
- Also dispose the old medicine after the expiration date.
- Talk to your pharmacist about the proper disposal of your medication.
- Rivastigmine was originally developed by Marta Weinstock-Rosin (Hebrew University of Jerusalem) and sold to Novartis by Yissum for commercial development.
- The drug itself is a semisynthetic derivative of physostigmine and became available in capsule and liquid formulations since 1997.
- The drug has first received its official approval for the treatment of mild to moderate dementia of the Alzheimer’s type.
- On 27 June 2006, Rivastigmine (Exelon) has received its official approval from US Food and Drug Administration (FDA) for the treatment of mild to moderate dementia (chronic loss or impairment of intellectual capacity) associated with Parkinson’s disease, a disorder of the central nervous system.
- In year 2007, the US Food and Drug Administration has approved an expanded indication for Rivastigmine transdermal patch (first patch treatment) for dementia.
Other uses of the drug.
- Rivastigmine is also sometimes used to prevent Lewy body dementia (a type of dementia characterized by the presence of clumps of alpha-synuclein and ubiquitin protein in neurons and closely associated with Parkinson’s disease).
- Rivastigmine may also be used for other uses not listed here. It is advisable to ask your doctor or pharmacist for more detailed information regarding its use.
What special dietary precautions should I follow?
- It is generally recommended to continue with the normal diet unless and until asked by your doctor.
- Avoid grape juice and alcoholic beverages.
- Drink plenty of water when you take the medicine.
What special precautions should I follow?/ What should I avoid while using Rivastigmine?
- First of all inform your doctor if you are allergic to Rivastigmine or any of the ingredients present in the Rivastigmine product. Ask your pharmacist or check the prescription leaflet carefully for a list of the ingredients.
- It is advisable to discuss with your doctor and pharmacist about what prescription and nonprescription medications, vitamins, and nutritional supplements you are taking or plan to take.
- Do not take Rivastigmine if you are taking another form of Rivastigmine (e.g., capsules, solution or patch) or another cholinesterase inhibitor (e.g., donepezil).
- Do not take Rivastigmine in combination with Beta-adrenergic receptor antagonists (beta-blockers), metoclopramide, aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs) and cholinomimetic and anticholinergic medications.
- Inform your doctor if you are breastfeeding or pregnant or plan to become pregnant.
- Rivastigmine use should be stopped under the supervision of doctor if you have bleeding ulcers, bleeding in the brain or a condition causing excessive bleeding.
- Before taking Rivastigmine tell your doctor about your medical history preferentially if you have any kind of liver or kidney disease, certain heart problems (e.g., slow or irregular heartbeat, sick sinus syndrome), stomach or bowel problems (e.g., an ulcer), inflammation of the pancreas (pancreatitis), seizures (e.g., epilepsy), lung or breathing problems (e.g., asthma, chronic obstructive pulmonary disease [COPD]), or a urinary blockage (trouble urinating).
- It is recommended to avoid consuming alcohol as it may increase the risk of bleeding in stomach or intestines.
- Inform your doctor about the use of Rivastigmine if you are having surgery or other medical procedures with general anesthesia.
- Do not use Rivastigmine if you have tremors or dementia/decreased mental ability not caused by Alzheimer or Parkinson disease.
- Do not use Rivastigmine if you are dehydrated or if your weight is less than 110 pounds (50 kg).
- Inform your doctor about the recent injury and try to avoid cuts or hurting yourself as it may lead to severe bleeding while you are taking Rivastigmine.
- Consult your doctor in case of any query.
What are the possible side effects of this drug?
The active ingredient in Rivastigmine may result in some side effects that require medical attention. Check with your doctor immediately if these side effects persist for a long time and become bothersome. Some commonly occurring side effects include:
- Loss of appetite
- Nausea
- Heartburn or indigestion
- Vomiting
- Weight loss
- Diarrhea
- Increased sweating
- Gas/stomach pain /constipation
- Extreme tiredness and lack of energy
- Weakness, dizziness and headache
- Tremor or worsening of tremor
- Difficulty falling asleep or staying asleep
Most of the people who take this drug do not develop serious side effects. The symptoms can develop any time after you start taking the medicine. If any of following symptoms appears get immediate medical help.
- Rash, hives
- Difficulty breathing or swallowing
- Bloody, black and tarry stools
- Bloody vomit
- Seizures, depression and anxiety
- Aggressive behavior
- Vomiting material that looks like coffee grounds
- Difficulty in urinating/painful urination
- Insomnia
- Hallucinations (hearing voices or seeing things that do not exist)
- Uncontrollable movements and muscle contractions
There can be more side effects of the drug not mentioned here. Contact your doctor or pharmacist if you notice any other unusual effect after taking this medicine.
What should I do in case of overdose?
- Overdose usually occurs when someone by mistake or deliberately takes more than the prescribed limit of this medication.
- In case of overdose, accompanied by severe nausea and vomiting, the use of antiemetics should be considered.
- In case of overdose, contact with your doctor or emergency room immediately.
- Seek emergency medical attention or call the Poison Help line at 1-800-222-1222.
What should I do in case of missed a dose?
- In case of missed dosage, take it as soon as you remember and maintain a regular dosing schedule. Keep in mind to take the missed dose only on an empty stomach.
- Skip the missed dose if it is almost time for your next scheduled dose. Keep in mind to not use a double dose to make up a missed dose.
Does Rivastigmine have any interaction with drugs?
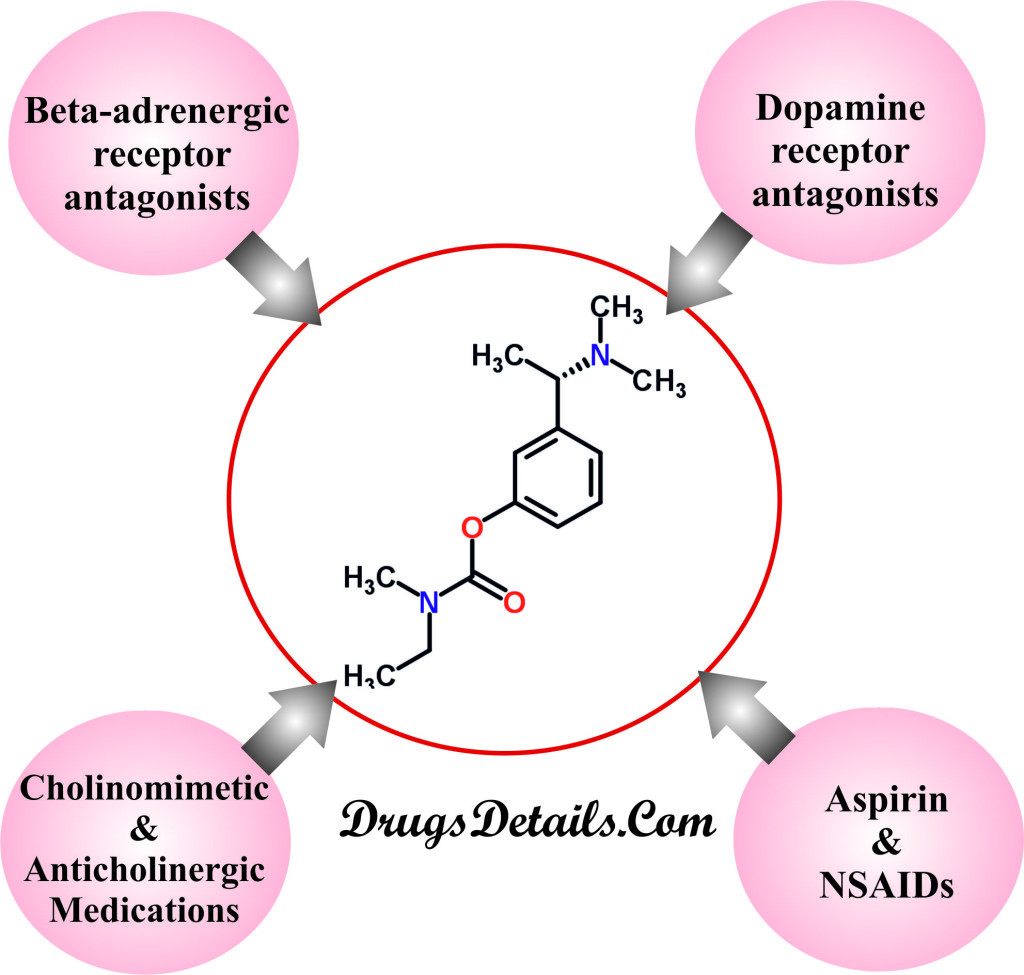
It has been observed that Rivastigmine may interact with or increase or decrease the effect of following drugs. Caution should be taken when co administrating Rivastigmine with one of the following drugs. It is advisable to inform your health care provider if you are taking any of the following medication.
- Beta-adrenergic receptor antagonists (beta-blockers): These are a family of medication that is widely used to treat hypertension, migraine prophylaxis, anxiety, essential tremor, hyperthyroidism, angina pectoris and cardiac arrhythmias. When used in combination with Rivastigmine these drugs have additive bradycardic effects that may increase the heartbeat (especially cardioselective beta-blockers including atenolol). Some of the most common beta blockers which interacts with Rivastigmine are acebutolol, atenolol, labetalol, betaxolol, bisoprolol, nadolol, carvedilol, penbutolol, pindolol, esmolol, metoprolol, nebivolol, propranolol, sotalol, or timolol. Coadministration of Rivastigmine is highly contraindicated and recommended when signs of bradycardia including syncope are present.
- Dopamine-receptor antagonists: These are group of medications that are used to treat stomach and esophageal problems such as nausea and vomiting (especially metoclopramide). Since coadministration of metoclopramide increased the risk of uncontrolled body due to the additive extra-pyramidal adverse reactions.
- Aspirin and Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): The risk of gastrointestinal bleeding (stomach or bowel bleeding) increases when Rivastigmine is coadministered with NSAIDs.
- Cholinomimetic and Anticholinergic Medications: Coadministration of Rivastigmine with other Cholinomimetic medications (e.g., bethanechol, donepezil) potentiate the cholinergic effects and interfere with the activity of anticholinergic medications (e.g., oxybutynin, tolterodine).
This list is not a complete list and there can be many more drugs that can interact with Rivastigmine. It is advisable to ask your health care provider if Rivastigmine may interact with other medicines that you take.
Does Rivastigmine have any interaction with diseases?
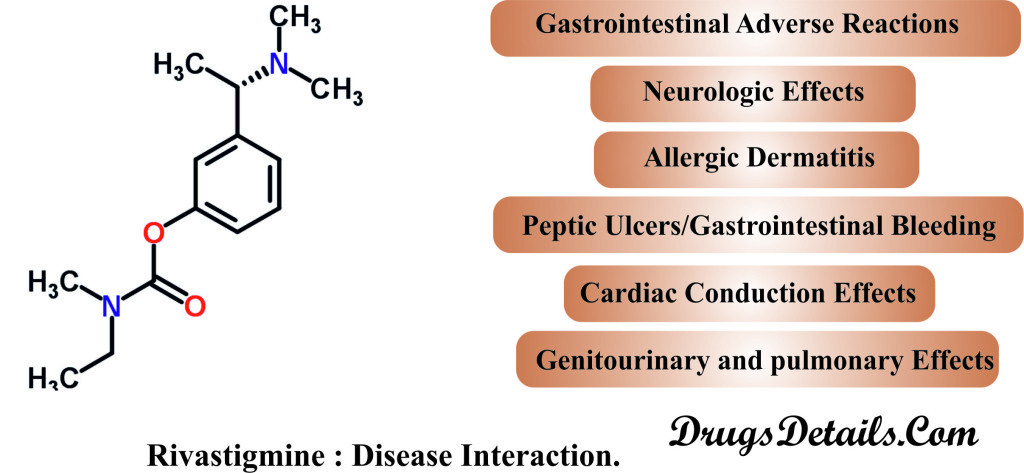
Before you begin to take Rivastigmine, it is necessary to discuss any medical condition or allergies you have or any other significant fact. It has been observed that following medical conditions (disease) may interact with Rivastigmine:
- Gastrointestinal Adverse Reactions: It has been observed that use of Rivastigmine can cause gastrointestinal adverse reactions, including significant nausea, anorexia/decreased appetite, vomiting, diarrhea, and weight loss. Prolonged vomiting or diarrhea can be associated with dehydration and other serious outcomes. The incidence and fatal outcome are associated in dose-dependent fashion.
- Allergic Dermatitis: Post-marketing reports of Rivastigmine use have reported the occurrence of disseminated allergic dermatitis irrespective of the route of administration (oral or transdermal). Therefore, it is advisable to stop the treatment immediately if disseminated allergic dermatitis occurs.
- Neurologic Effects: Rivastigmine is principally a cholinomimetics that may potentiate or induce extrapyramidal symptoms such as tremors and deterioration of parkinsonian symptoms. Apart from this, Rivastigmine use may also exacerbate the incidence of seizures, although, seizure activity may also be due to manifestation of Alzheimer’s disease.
- Peptic Ulcers/Gastrointestinal Bleeding: The use of Rivastigmine is associated with increased gastric acid secretion due to its cholinergic activity. Therefore, it is advisable to monitor the patients (especially who have increased risk for developing ulcers) for symptoms of active or occult gastrointestinal bleeding.
- Cardiac Conduction Effects: Because Rivastigmine increases cholinergic activity, use of Rivastigmine may hyper the excitability of the vagus nerve (Vagotonic effect) that can produce bradycardia, decreased heart output, and faintness. The occurrence of bradycardia is more in patients with sick sinus syndrome or other supraventricular cardiac conduction conditions.
- Genitourinary and Pulmonary Effects: It has been speculated that Rivastigmine cholinergic activity may cause urinary obstruction. Furthermore, Rivastigmine use should be under care in patients with a history of asthma or obstructive pulmonary disease.
Where can I get more information?
Your pharmacist can provide more information about Rivastigmine.
Clinical research and current scenario of the drug.
- Clinical studies performed under both in vitro and in vivo conditions suggested that cholinergic activity of Rivastigmine (inhibition of cholinesterase) is not affected by the concomitant administration of N-methyl-D-aspartate receptor antagonist i.e. memantine.
- Toxicity and metabolic studies using animal model suggested the minimal involvement of major cytochrome P450 isozymes in Rivastigmine metabolism. These studies were in accordance with previous observations in humans that suggested no drug interactions related to cytochrome P450.
- Clinical studies using Rivastigmine in North American trial in patients with mild to moderate Alzheimer disease, showed dose-dependent treatment benefit on cognition, function, and activities of daily living.
- Experimental trials have been performed to evaluate the effect of Rivastigmine for dementia with lewy bodies and Parkinson’s disease dementia, delirium, traumatic brain injury, down’s syndrome, supranuclear palsy, and cocaine dependence.
- Clinical trials conducted to analyze the effect of Rivastigmine therapy in AD patients with hallucinations showed better response to Rivastigmine treatment than those without
- Clinical investigation on Rivastigmine transdermal patch suggested that transdermal patches provide continuous drug delivery and, reduce the fluctuations in plasma and central nervous system drug concentrations.
References from chemical, biological and toxicological databases.
- Rivastigmine: MedlinePlus Drug Information. http://www.nlm.nih.gov/medlineplus/druginfo/meds/a602009.html
- Rivastigmine – Wikipedia, the free encyclopedia. https://en.wikipedia.org/wiki/Rivastigmine
- DrugBank: Rivastigmine (DB00989). http://www.drugbank.ca/drugs/DB00989
- Rivastigmine | C14H22N2O2 | ChemSpider. www.chemspider.com/Chemical-Structure.70377.html
- Rivastigmine | C14H22N2O2 – PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/rivastigmine
- Rivastigmine in the treatment of Alzheimer’s disease: an update. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2684084/
- Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: a systematic review and meta-analysis. http://www.ncbi.nlm.nih.gov/pubmed/18686744
- PubHealth. http://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0001593/
- Pharmacokinetics of a novel transdermal rivastigmine patch for the treatment of Alzheimer’s disease: a review. http://www.ncbi.nlm.nih.gov/pubmed/19392927
- Rivastigmine for Alzheimer’s disease. http://www.ncbi.nlm.nih.gov/pubmed/25858345
Read about,
“Darunavir, drug class, uses, strength, side effects, mechanism of action”
“Enoxaparin, drug class, uses, strength, side effects, mechanism of action“
